-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Research in Zoology
p-ISSN: 2325-002X e-ISSN: 2325-0038
2013; 3(3): 67-74
doi:10.5923/j.zoology.20130303.01
Preliminary Zooplankton Study of Six “Mercedinas” Lakes (San Luis, Argentina)
Cabrera Gabriela1, Vignatti Alicia1, Salinas Víctor2, Echaniz Santiago1, Mancini Miguel2
1Facultad de Ciencias Exactas y Naturales. Universidad Nacional de La Pampa. Avda. Uruguay 151. Código Postal 6300. Santa Rosa, La Pampa
2Ecología y Acuicultura. Facultad de Agronomía y Veterinaria. Universidad Nacional de Río Cuarto. Ruta Nac. 36 - Km. 601. Código Postal X5804BYA. Río Cuarto, Córdoba
Correspondence to: Cabrera Gabriela, Facultad de Ciencias Exactas y Naturales. Universidad Nacional de La Pampa. Avda. Uruguay 151. Código Postal 6300. Santa Rosa, La Pampa.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
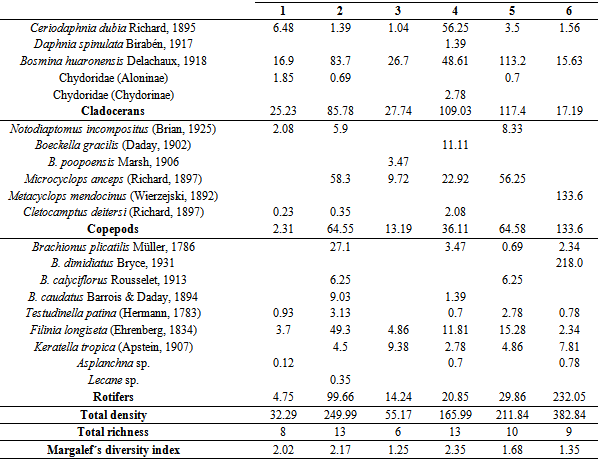
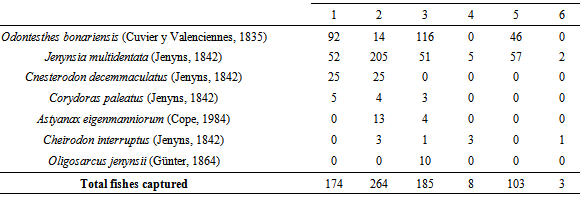
The south of the province of San Luis (Argentina) has more than 100 lakes located among dunes and grasslands, known as mercedinas due to their proximity to the city of Villa Mercedes. Although there are several reports on the ecology of zooplankton in other regions of the country, very little is known about the aquatic ecosystems of this extensive area. The aim of the study was to determine the taxonomic composition and the zooplankton density of six “mercedinas” lakes and analyze the relationship between this community and limnological parameters and fish fauna. The samplings were carried out in October 2007. The average depth of the six lakes ranged between 2.3 and 3.6 m. Salinity ranged between 1.3 and 3.3 g.L-1, so they can be classified as subsaline to slightly hyposaline. Water transparency varied widely (0.3-2.3 m). Macrophytes were present in the six lakes and fish richness was represented by seven species, with predominance of Jenynsia multidentata and Odontesthes bonariensis. Zooplankton richness included 20 taxa: five cladocerans, six copepods and nine rotifers. The species common to the six lakes were Bosmina huaronensis, Ceriodaphnia dubia and Filinia longiseta. We found a situation that does not match that proposed by the model of alternative states of shallow lakes: the lakes where we did not find O. bonariensis, a main predator of zooplankton, showed the lowest water transparency, although a large filtering cladoceran such as Daphnia spinulata was present in one of them. These preliminary results make it necessary to carry out further research to see if this situation is maintained over time or if this was a specific situation.
Keywords: Zooplankton, Bosmina Huaronensis, Shallow Lakes, “Lagunas Mercedinas”
Cite this paper: Cabrera Gabriela, Vignatti Alicia, Salinas Víctor, Echaniz Santiago, Mancini Miguel, Preliminary Zooplankton Study of Six “Mercedinas” Lakes (San Luis, Argentina), Research in Zoology , Vol. 3 No. 3, 2013, pp. 67-74. doi: 10.5923/j.zoology.20130303.01.
Article Outline
1. Introduction
- In aquatic ecosystems, zooplankton is a key component because of it grazes on phytoplankton, recycles nutrients through excretion, and serves as food to predators. Its taxonomic composition and density depend on abiotic factors such as salinity, temperature, environmental heterogeneity, or land use in the basin[1, 2, 3, 4, 5, 6] and biotic factors such as food availability, interspecific competition and predation by vertebrates and invertebrates[7, 8, 9, 10, 11]. The predation by fish has strong structuring effects on the diversity and size of the zooplankton community[12], which in turn cause marked changes in the ecology of water bodies by the effect of the trophic cascade[10, 11, 13, 14]. The distribution of zooplankton rotifers and crustaceans is relatively well known in various regions of Argentina. The taxonomic composition of the zooplankton has been studied both in lentic[15, 16, 17, 18, 19] and lotic environments[20, 21, 22] of the province of Buenos Aires. The zooplankton of the basin of the Paraná River[23, 24], as well as that of the saline lakes of the Northwest, both of the Puna[25, 26] and of the Yungas[27], are also relatively known. There is also information about the zooplankton of some aquatic ecosystems of Patagonia[28, 29, 30] and even of some lakes of Antarctica[31, 32]. Other publications have provided information on the distribution of rotifers[33, 34, 35], cladocerans[36, 37, 38] and copepods[39, 40, 41, 42, 43]. However, knowledge of the zooplankton of the water bodies of the central semiarid region of Argentina, which are usually temporary and shallow lakes, is more recent and referred primarily to environments of the provinces of La Pampa[44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57] and Córdoba[58]. Since the zooplankton diversity of many water bodies in other provinces of the same region still remains unknown, some authors have proposed that some species could have discontinuous distributions between the north of Argentina and Patagonia[59]. In some cases, this perceived discontinuity was the result of lack of information, such as was the case of Daphnia menucoensis, whose distribution was found to be relatively continuous between the two regions[54]. In central Argentina, there are numerous shallow lakes with a wide range of salinities and it is common that they are either eutrophic or hypertrophic[49, 55, 60, 61]. Many have suffered alterations due to the human activities developed in their watersheds, such as piping, animal husbandry, cultivation of grains and oilseeds, and urbanizations, or due to the introduction of fish, particularly the zooplanktivorous Odontesthes bonariensis Cuvier y Valenciennes, 1835 due to its sport and economic value[50, 62, 63, 64]. The aim of this work was to study the taxonomic composition of the zooplankton community, the total density and the density per species of shallow lakes of the province of San Luis, an area for which there are no previous reports on this issue, and to relate them to the composition of the fish assemblages and main limnological parameters.
2. Materials and Method
2.1. Study Area
- The six lakes studied (lakes 1 to 6) are located in the General Pedernera Department (34°00´S, 65°24´W), province of San Luis, in the region known as "area of plains and lagoons", in the geomorphological system of the pampas of eolic sands[60] (Figure 1). From the phytogeographic point of view, the lakes are located in the province of Thorny Forest, characterized by the presence of deciduous forests with a predominance of the genus Prosopis and psammophilous pastures[65]. The average annual rainfall ranges from 500 to 600 mm and the average temperature from 7.3 to 22.9ºC for the months of January and July respectively.
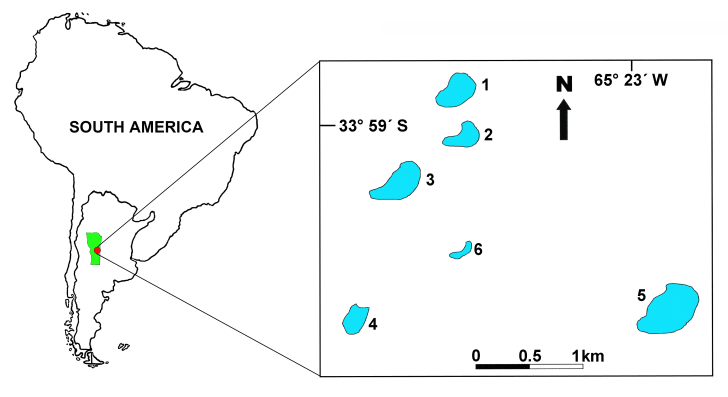 | Figure 1. Geographical location of the studied “mercedinas” lakes |
|
2.2. Field Work and Laboratory
- The samplings were carried out in October 2007. In situ, we determined transparency with a Secchi disk, the concentration of dissolved oxygen, and pH and water temperature with a Consort C535 digital Analyzer. Also, water samples from the center of each lake were taken to perform physicochemical analysis.Quantitative subsurface zooplankton samples were collected at three sampling stations in the limnetic zone along the longitudinal axis of each lake. In each station, we filtered 36 L of water with a 90-µm-mesh plankton net. Samples were fixed with 4% formalin.Fish were captured by trotlines, drag nets and drift nets, and species were identified according to Rosso, 2007[62], Haro and Bistoni, 2007[66] and López et al., 2003[67].Hardness, alkalinity and concentration of total dissolved solids in the water[68] were determined in the laboratory.Abundances of macro- and microzooplankton[14] were determined by the count of subsamples in Bogorov and Sedgwick Rafter chambers respectively. Aliquots were taken with 5-ml Russell subsamplers and 1-ml micropipettes. The taxonomic identification of zooplankton were according to Ringuelet, 1958[39], Olivier, 1962 and 1965[37, 69], Ruttner-Kolisko, 1974[70], Koste, 1978[71], Reid, 1985[72], Dusart and Frutos, 1986[73], Bayly, 1992[74] and Paggi, 1995[36].The ratio between the mean depth and the depth of the photic area (Zm/Zphot) was calculated to classify the lakes according to their transparency[75]. To compare the diversity of water bodies, we calculated the Margalef and dominance indices, and to determine replacement of species between environments, we calculated Whittaker’s beta diversity index[76, 77, 78, 79]. The similarity of the lakes was determined through cluster analysis based on the similarity of Bray-Curtis. Past software was used[80].
3. Results
|
|
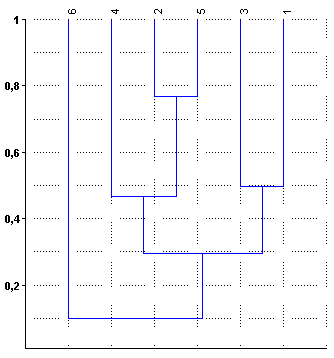 | Figure 2. Clustering of lakes based on zooplankton species |
4. Discussion
- The lakes studied were shallow and had a relatively reduced extension. Although the concentration of dissolved solids was not determined in all cases, four of them can be characterized as subsaline to slightly hyposaline[81]. The latter is common in aquatic environments located in dune landscapes, in watersheds caused by wind deflation, fed mainly by rainfall and phreatic contributions. Water transparency covered a wide range, from very turbid to very clear lakes, such as the case of lake 1. The values of the ratio between the depths of the lake and the photic zone were also different and allowed categorizing lakes 1, 3 and 5 as clear, a state that would be favored by the presence of macrophytes in the littoral zone and the lower impact of agricultural activities developed in its surroundings[60]. In contrast, lakes 2 and 4 can be categorized as turbid. In this case, the dominance of cyanophytes and the agricultural activity developed in its basins would contribute to the maintenance of the turbid state of its waters[60].Total zooplankton richness was relatively high and the beta diversity index showed a relatively low species replacement between lakes, which is natural because these lakes are located in a region with little expansion and have relatively similar characteristics.Most of the species found are typical of the zooplankton association of lakes with intermediate salinity in the central region of the country and the presence of the cladocerans Ceriodaphnia dubia and Bosmina huaronensis makes the “mercedinas” lakes similar to the relatively eutrophic reservoirs of the Northwest Center of Argentina[38]. However, a discordant trait was the presence in lake 3 of Boeckella poopoensis, a widely extended halotolerant calanoid, typical of saline lakes, between 5 and 90 g.L-1, whose distribution is considered to be linked with the migratory route of the southern flamingo Phoenicopterus chilensis Molina, 1782[82], since it includes B. poopoensis in its feeding and has a wide distribution in Argentina[83]. The presence of B. poopoensis in only one lake and its very low density could be due to an occasional event, related to the passing of flamingos during their migration, since, in the same lake, we recorded B. huaronensis and C. dubia, zooplankton species typical of low salinity lakes, with which the coexistence of B. poopoensis has not been reported.Lake 4, which had very low transparency, showed the presence of Daphnia spinulata, a cladoceran of considerable size, which is relatively common when there are no predators[64, 84]. This was the case of this lake because O. bonariensis, the main zooplanktivorous fish, was absent. Most of the remaining lakes showed higher water transparency and presence of O. bonariensis, and the crustaceans recorded were of small size. The latter raises a particular situation regarding the “mercedinas” lakes, because it does not coincide with that proposed by the model of the alternative states of shallow lakes (lakes less than 3 m deep). This theory, which takes into account changes in the characteristics of the lakes caused by changes in the zooplankton grazing pressure, considers that the lakes can alternate between two states. In the absence of predation, zooplankton dominated by species of large size and high efficiency of filtration of phytoplankton (especially cladocerans of the genus Daphnia) frequently develop, which leads to a clear state, with high water transparency[14, 47, 85, 86, 87, 88]. On the other hand, in the presence of zooplanktivorous fish,the zooplankton community is reflected in the dominance of taxa of reduced size and hence of low filtration efficiency, which results in the establishment of the turbid state[14, 64, 85, 86, 87, 88]. However, in the lakes studied, we verified that the lowest water transparencies were found in lakes where O. bonariensis was absent, despite the presence of D. spinulata in one of them.Although the results here presented are preliminary, they allow characterizing the diversity and density of the zooplankton of six “mercedinas” lakes, located in a region of Argentina where there was no information. Future systemized and long-term studies will help determine the seasonal dynamics of zooplankton and its relationship with the fish fauna and verify whether there is alternation between the clear and turbid states that characterize shallow lakes, or if (as in some lakes of Central Argentina) one state or another dominates[48, 51, , 56, 57, 64, 89].
References
| [1] | Dumont, H. and H. Segers, 1996. Estimating lacustrine zooplankton species richness and complementarity. Hydrobiologia 341: 125-132. |
| [2] | Kobayashi, T., 1997. Associations between environmental variables and zooplancton body masses in a regulated Australian river. Marine and Freshwater Research 48: 523 - 529. |
| [3] | Hobæk, A., M. Manca and T. Andersen, 2002. Factors influencing species richness in lacustrine zooplankton. Acta Oecologica 23: 155 - 163. |
| [4] | Hall, C. and C. Burns, 2003. Responses of crustacean zooplankton to seasonal and tidal salinity changes in the coastal Lake Waihola, New Zealand. New Zealand Journal of Marine and Freshwater Research 37: 31- 43. |
| [5] | Dodson, S. I., R. A. Lillie, and S. Will-Wolf, 2005. Land use, water chemistry, aquatic vegetation, and zooplankton community structure of shallow lakes. Ecological applications. 15: 1191-1198. |
| [6] | Dodson, S., W. Everhart, A. Jandl and S. Krauskopf, 2007. Effect of watershed land use and lake age on zooplankton species richness. Hydrobiologia 579: 393 - 399. |
| [7] | Lampert, W. and U. Sommer, 1997. Limnoecology. The ecology of lakes and streams. Oxford University Press, London. |
| [8] | Khan, T., M. Wilson and M. Khan, 2003. Evidence for invasive carp mediated trophic cascade in shallow lakes of western Victoria, Australia. Hydrobiologia 506 - 509: 465 - 472. |
| [9] | Chang, K., T. Nagata and T. Hanazato, 2004. Direct and indirect impacts of predation by fish on the zooplankton community: an experimental analysis using tanks. Limnology 5: 121 - 124. |
| [10] | [Boveri, M. and R. Quirós, 2007. Cascading trophic effects in pampean shallow lakes: results of a mesocosm experiment using two coexisting fish species with different feeding strategies. Hydrobiologia 584: 215 - 222. |
| [11] | Manca, M., J. Vijverberg, L. Polishchuk and D. Voronov, 2008. Daphnia body size and population dynamics under predation by invertebrate and fish predators in Lago Maggiore: an approach based on contribution analysis. Journal of Limnology 67(1): 15 - 21. |
| [12] | Brooks, J.L. and S.I. Dodson, 1965. Predation, body size, and composition of plankton. Science, 150: 28-35. |
| [13] | Bertolo, A., G. Lacroix, F. Lescher-Moutoué and C. Cardinal-Legrand, 2000. Plankton dynamics in planktivore and piscivore-dominated mesocosms. Archiv für Hydrobiologie. 147: 327-349. |
| [14] | Kalff, J., 2002. Limnology. Inland Water System. Prentice Hall, New Jersey. |
| [15] | Ringuelet, 1962. Ecología acuática continental. EUDEBA, Buenos Aires. |
| [16] | Ringuelet, R., 1972. Ecología y Biocenología del hábitat lagunar o lago de tercer orden de la región neotrópica templada (Pampasia Sudoriental de la Argentina). Physis 31 (82): 55 - 76. |
| [17] | Ringuelet, R., I. Moreno and E. Feldman, 1967. El zooplancton de las lagunas de la Pampa Deprimida y otras aguas superficiales de la llanura bonaerense (Argentina). Physis 27 (74): 187 - 200. |
| [18] | Claps, M., N. Gabellone and H. Benítez, 2004. Zooplankton biomass in an eutrophic shallow lake (Buenos Aires, Argentina): spatio – temporal variations. Annales de Limnologie - International Journal of Limnology 40 (3): 201 - 210. |
| [19] | Ardohain, D., H. Benítez, M. Claps and N. Gabellone, 2005. Estructura y dinámica de rotíferos planctónicos en dos lagunas pampásicas: similitudes y diferencias. Biología Acuática 22: 7 - 18. |
| [20] | Modenutti, B., 1998. Planktonic rotifers of Samborombón River Basin (Argentina). Hydrobiologia 387/388: 259 - 265. |
| [21] | Neschuk, N., N. Gabellone and L. Solari, 2002. Plankton characterisation of a lowland river (Salado River, Argentina). Verhandlungen des Internationalen Verein Limnologie 28: 1336 - 1339. |
| [22] | Neschuk, N., M. Claps and N. Gabellone, 2002. Planktonic rotifers of a salty lowland river (Salado River, Argentina). Annales de Limnologie- International Journal of Limnology 38: 191 - 198. |
| [23] | Paggi, J.C. and S. José de Paggi, 1990. Zooplankton of lotic and lenitic environments of the Middle Paraná River. Acta Limnologica Brasiliensis 3: 685-719. |
| [24] | José De Paggi S. and J. Paggi, 1998. Zooplancton de ambientes acuáticos con diferentes estados tróficos y salinidad. Neotropica. 44(1): 95 - 106. |
| [25] | Bayly, I. A. E., 1993. The fauna of athalassic saline waters in Australia and the Altiplano of South America: comparisons and historical perspectives. Hydrobiologia 267: 225 - 231. |
| [26] | Locascio de Mitrovich, C., A. Villagra de Gamundi, J. Juárez and M. Ceraolo, 2005. Características limnológicas y zooplancton de cinco lagunas de la Puna – Argentina. Ecología en Bolivia 40(1): 10 - 24. |
| [27] | Villagra de Gamundi, A., C. Locascio de Mitrovich, J. Juárez and G. Ferrer, 2008. Consideraciones sobre el zooplancton de las lagunas de Yala (Jujuy, Argentina). Ecología en Bolivia 43(2) 1 - 16. |
| [28] | Modenutti, B., E. Balseiro, M. Diéguez, C. Queimaliños and R. Albariño, 1998. Heterogeneity of fresh-water Patagonia ecosystems. Ecologia Austral 8: 155 - 165. |
| [29] | Modenutti, B., R. Albariño, M. Bastidas Navarro, V. Díaz Villanueva, M. Souza, C. Trochine, C. Laspoumaderes, F. Cuassolo, G. Mariluán, L. Buria and E. Balseiro, 2010. Structure and dynamic of food webs in Andean North Patagonian freshwater systems: organic matter, light and nutrient relationships. Ecología Austral 20(2): 95-114. |
| [30] | Adamowicz, S., P. Hebert and M. C. Marinone, 2004. Species diversity and endemism in the Daphnia of Argentina: a genetic investigation. Zoological Journal of the Linnean Society. 140: 171-205. |
| [31] | Paggi, J., 1987. Limnological studies in Potter Peninsula, 25 de Mayo Island, South Shetland Islands: Biomass and spatial distribution of zooplankton. BIOMASS Scientific Series 7: 175 - 191. |
| [32] | Izaguirre, I., L. Allende and M. C. Marinone, 2003. Comparative study of the planktonic communities of three lakes of contrasting trophic status at Hope Bay (Antarctic Peninsula). Journal of Plankton Research. 25(9): 1079-1097. |
| [33] | Olivier, S., 1965. Rotíferos planctónicos de Argentina. Revista del Museo de La Plata (NS) Sección Zoología 8 (63):177 - 260. |
| [34] | Kuckzinsy, D., 1987. The rotifer fauna of Argentine Patagonia as a potential limnological indicator. Hydrobiologia 150: 3-10. |
| [35] | José de Paggi, S., 1989. Rotíferos de algunas provincias del Noroeste de Argentina. Rev. Hydrobiol. Trop. 22: 223-238. |
| [36] | Paggi, J., 1995. Cladocera. Pp. 909 - 951. En: Lopretto E. y G. Tell (eds) Ecosistemas de aguas contientales. Metodologías para su estudio. Ediciones Sur, La Plata. |
| [37] | Olivier, S., 1962. Los cladóceros argentinos, con clave de las especies, notas biológicas y distribución geográfica. Revista del Museo de La Plata (NS) Sección Zoología 7: 173 - 269. |
| [38] | Paggi, J., 1998. Cladocera (Anomopoda y Ctenopoda). Pp. 507-518. En: S. Coscarón & J. J. Morrone (eds), Biodiversidad de Artrópodos Argentinos. Ediciones Sur, La Plata. |
| [39] | Ringuelet, R., 1958. Los Crustáceos Copépodos de las aguas continentales de la República Argentina. Sinopsis sistemática. Contribuciones Científicas de la Facultad de Ciencias Exactas y Naturales UBA, Zoología 1: 35 - 126. |
| [40] | Dussart, B. and S. Frutos, 1986. Sur quelques Copépodes d’Argentine. 2. Copépodes du Paraná medio. Revue d’ Hydrobiologie Tropicale 19 (3-4): 241 - 262. |
| [41] | Battistoni, P. A., 1998. Capítulo 51: "Copepoda”. Pp. 519-530. En: S. Coscarón & J. J. Morrone (eds), Biodiversidad de Artrópodos Argentinos. Ediciones Sur, La Plata. |
| [42] | Menu-Marque, S. and C. Locascio de Mitrovich, 1998. Distribución geográfica de las especies del género Boeckella (Copepoda, Calanoida, Centropagidae) en la República Argentina. Physis, B, 56: 1 - 10. |
| [43] | Menu-Marque, S., J. Morrone and C. Locascio de Mitrovich, 2000. Distributional patterns of the south american species of Boeckella (Copepoda: Centropagidae): a track analysis. Journal of Crustacean Biology 20 (2): 262 - 272. |
| [44] | Echaniz, S. and A. Vignatti, 1996. Cladóceros limnéticos de la provincia de La Pampa (Argentina). Revista de la Facultad de Agronomía. UNLPam 9 (1): 65 - 80. |
| [45] | Echaniz, S. and A. Vignatti, 2002. Variación anual de la taxocenosis de cladóceros planctónicos (Crustacea: Branchiopoda) de una laguna de elevada salinidad (La Pampa, Argentina). Neotrópica 48: 11 - 17. |
| [46] | Echaniz, S. and A. Vignatti, 2002. Caracterización limnológica y variación anual de la taxocenosis de cladóceros de la laguna Quetré Huitrú (pcia. de La Pampa, Argentina). Pág. 40, resúmenes II Jornadas de Ecología y Manejo de Ecosistemas Acuáticos Pampeanos. La Plata, 13 al 15 de noviembre de 2002. |
| [47] | Echaniz, S. A. and A. M. Vignatti, 2010. Diversity and changes in the horizontal distribution of crustaceans and rotifers in an episodic wetland of the central region of Argentina. Biota Neotropica 10 (3): 133-141. |
| [48] | Echaniz, S. A. and A. M. Vignatti, 2011. Seasonal variation and influence of turbidity and salinity on the zooplankton of a saline lake in central Argentina. Latin American Journal of Aquatic Research 39 (2): 306-315. |
| [49] | Echaniz, S., A. Vignatti, S. José de Paggi, J. Paggi and A. Pilati, 2006. Zooplankton seasonal abundance of south american saline shallow lakes. International Review of Hydrobiology (91): 86 - 100. |
| [50] | Echaniz, S., A. M. Vignatti and P. C. Bunino, 2008. El zooplancton de un lago somero hipereutrófico de la región central de Argentina. Cambios después de una década. Biota Neotropica. 8(4): 63 - 71. |
| [51] | Echaniz, S. A., A. M. Vignatti, G. C. Cabrera, and S. B. José de Paggi, 2012. Zooplankton richness, abundance and biomass of two hypertrophic shallow lakes with different salinity. Biota Neotropica 12(2): 37-44. |
| [52] | Pilati, A., 1997. Copépodos calanoideos de la provincia de La Pampa. Revista de la Facultad de Agronomía UNLPam 9 (2): 57 - 67. |
| [53] | Pilati, A., 1999. Copépodos ciclopoideos en la provincia de La Pampa (Argentina). Revista de la Facultad de Agronomía UNLPam 10 (1): 29 - 44. |
| [54] | Vignatti, A. M. and S. A. Echaniz, 1999. Presencia de Daphnia (Ctenodaphnia) menucoensis Paggi, 1996 en la provincia de La Pampa (Argentina). Rev. Fac. Agronomía. UNLPam. 10 (1): 21-27. |
| [55] | Vignatti, A., S. Echaniz and M. Martín, 2007. El zooplancton de lagos someros de diferente salinidad y estado trófico en la región semiárida pampeana (La Pampa, Argentina). Gayana 71 (1): 38 - 48. |
| [56] | Vignatti, A., G. Cabrera, and S. Echaniz, 2012. Changes in the zooplankton and limnological variables of a temporary hypo-mesosaline wetland of the central region of Argentina during the drying. Pan American Journal of Aquatic Sciences 7(2): 93-106. |
| [57] | Vignatti, A. M., J. C. Paggi, G. C. Cabrera and S. A. Echaniz, 2012. Zooplankton diversity and its relationship with environmental changes after the filling of a temporary saline lake in the semi-arid region of La Pampa (Argentina). Latin American Journal of Aquatic Research 40(4): 1005-1016. |
| [58] | Mancini, M., A. Bethular, A. Vignatti, S. Echaniz, M. Bonansea, V. Salinas and C. Rodríguez, 2011. Calidad del agua y zooplancton del embalse San Roque (Córdoba, Argentina). Ciencia (UNCa) 6 (21): 69-80. |
| [59] | Paggi, J., 1996. Daphnia (Ctenodaphnia) menucoensis (Anomopoda; Daphniidae) a new species from athalassic saline waters in Argentina. Hydrobiologia 319: 137 -147. |
| [60] | Mancini, M., V. Salinas, J.G. Haro, 2009. Diferentes aspectos ecológicos e ictiofauna de seis lagunas mercedinas (San Luis, Argentina). Biología Acuática 26: 151-165. |
| [61] | Echaniz, S., 2010. Composición y abundancia del zooplancton en lagunas de diferente composición iónica de la provincia de La Pampa. Tesis Doctoral. Universidad de Río Cuarto, Facultad de Ciencias Exactas, Físico-Químicas y Naturales. |
| [62] | Rosso, J., 2007. Peces pampeanos. Guía y Ecología. LOLA, Literatura of Latin America, Buenos Aires, 221 pp. |
| [63] | Echaniz, S., A. Vignatti and G. Cabrera, 2009. Características limnológicas de una laguna turbia orgánica de la provincia de La Pampa y variación estacional del zooplancton. Biología Acuática 26: 71-82. |
| [64] | [64]Echaniz, S.; A. Vignatti; S. José de Paggi and G. Cabrera, 2010. El modelo de estados alternativos de lagos someros en La Pampa: comparación de Bajo de Giuliani y El Carancho. Libro de Trabajos del 3º Congreso Pampeano del Agua. 45-53. |
| [65] | Cabrera, A., 1976. Regiones fitogeográficas argentinas. Fascículo 1, Enciclopedia Argentina de agricultura y jardinería. Ed. Acme. Buenos Aires. |
| [66] | Haro, J. and M. Bistoni, 2007. Peces de Córdoba. Editorial de la Universidad Nacional de Córdoba. Córdoba. |
| [67] | López H., A. Miquelarena and R. Menni, 2003. Lista comentada de los peces continentales de Argentina. ProBiota, Serie Técnica y Didáctica N° 5. La Plata. |
| [68] | APHA, 1992. Standard Methods for the Examination of Water and Wastewater. 18th edition. American Public Health Association (APHA), American Water Works Association (AWWA) and Water Pollution Control Federation (WPCF), Washington, DC. |
| [69] | Olivier, S., 1965. Rotíferos planctónicos de Argentina. Revista del Museo de La Plata (NS) Sección Zoología 8 (63):177 - 260. |
| [70] | Ruttner-Kolisko, A., 1974. Plankton rotifers; Biology and taxonomy. Die Binnengewässer 26 (1), Stuttgart. |
| [71] | Koste, W., 1978. Rotatoria. Die Rädertiere Mitteleuropas. Borntraeger, Berlin-Stuttgart. |
| [72] | Reid, J., 1985. Chave de identificação e lista de referências bibliográficas para as espécies continentais sulamericanas de vida livre da ordem Cyclopoida (Crustacea, Copepoda). Boletim de Zoologia, Universidade de Säo Paulo 9: 17 - 143. |
| [73] | Dussart, B. and S. Frutos, 1986. Sur quelques Copépodes d’Argentine. 2. Copépodes du Paraná medio. Revue d’ Hydrobiologie Tropicale 19 (3-4): 241 - 262. |
| [74] | Bayly, I. A., 1992. Fusion of the genera Boeckella and Pseudoboeckella (Copepoda) and revision of their species from South America and sub-Antarctic islands. Revista Chilena de Historia Natural 65: 17 - 63. |
| [75] | Quirós, R., A. Rennella, M. Boveri, J. Rosso and A. Sosnovsky, 2002. Factores que afectan la estructura y el funcionamiento de las lagunas pampeanas. Ecología Austral 12: 175-185. |
| [76] | Moreno, C., 2001. Métodos para medir la biodiversidad. M. & T. Manuales y Tesis SEA, Vol 1. Zaragoza. |
| [77] | Magurran, A., 2004. Measuring Biological Diversity. Blackwell Science Ltd., Victoria. |
| [78] | Halffter, G. and C. Moreno, 2005. Significado biológico de las diversidades alfa, beta y gamma. Pp 5-18. En Halffter, G., J. Soberón, P, Koleff & A. Melic (eds.), Sobre diversidad biológica: significado biológico de las diversidades alfa, beta y gamma. Serie Monografías del Tercer Milenio vol. 4. Sociedad Entomológica Aragonesa, Zaragoza. |
| [79] | Koleff, P., 2005. Conceptos y medidas de la diversidad beta. Pp 19-40. En Halffter, G., J. Soberón, P, Koleff & A. Melic (eds.), Sobre diversidad biológica: significado biológico de las diversidades alfa, beta y gamma. Serie Monografías del Tercer Milenio vol. 4. Sociedad Entomológica Aragonesa, Zaragoza. |
| [80] | Hammer, Ø., D. Harper and P. Ryan, 2001. PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontologia Electronica 4(1): 1-9. |
| [81] | Hammer, U., 1986. Saline Lake Ecosystems of the World. Monographiae Biologicae 59. Dr. W. Junk Publishers, Dordrecht. |
| [82] | De los Ríos-Escalante, P., 2011. Morphological variations in Boeckella poopoensis (Marsh, 1906) (Copepoda, Calanoida) in two shallow saline ponds (Chile) and potential relation to salinity gradient. International Journal of Aquatic Science. 2(1): 80-86. |
| [83] | Narosky, T. and D. Yzurieta, 2003. Guía para la Identificación de las Aves de Argentina y Uruguay. Edición de Oro. Vázquez Mazzini Editores. Buenos Aires. |
| [84] | Muylaert, K., S. Declerck, J. Van Wichelen, L. De Meester and W. Vyverman, 2006. An evaluation of the role of daphnids in controlling phytoplankton biomass in clear water versus turbid shallow lakes. Limnologica 36: 69 - 78. |
| [85] | Scheffer, M., S. Hosper, M. Meijer, B. Moss and E. Jeppesen, 1993. Alternative equilibria in shallow lakes. Trends in Ecology and Evolution 8: 275 - 279. |
| [86] | Scheffer, M., 1998. Ecology of shallow lakes. Chapman & Hall, London. |
| [87] | Jeppesen, E., M. Søndergaard, A. Pedersen, K. Jürgens, A. Strzelczak, T. Lauridsen, and L. Johansson, 2007. Salinity Induced Regime Shift in Shallow Brackish Lagoons. Ecosystems (10): 47 - 57. |
| [88] | Jeppesen, E., M. Søndergaard, M. Meerhoff, T. L. Lauridsen and J. P. Jensen, 2007. Shallow lake restoration by nutrient loading reduction- some recent findings and challenges ahead. Hydrobiologia 584: 239–252. |
| [89] | Echaniz, S., G. Cabrera, C. Rodríguez and A. Vignatti, 2013. Do temporary lakes vary from year to year? A comparison of limnological parameters and zooplankton from two consecutive annual cycles in an Argentine temporary saline lake. International Journal of Aquatic Science. 4(1): 44-61. |
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML