-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Science and Technology
p-ISSN: 2163-2669 e-ISSN: 2163-2677
2016; 6(1): 8-14
doi:10.5923/j.scit.20160601.02

Repetitive Use of Vegetable Cooking Oil and Effects on Physico-Chemical Properties – Case of Frying with Redfish (Lutjanus fulgens)
Nancy Idun-Acquah1, George Y. Obeng2, Ebenezer Mensah1
1Department of Agricultural Engineering, College of Engineering, Kwame Nkrumah University of Science and Technology (KNUST), Kumasi, Ghana
2Technology Consultancy Centre, College of Engineering, Kwame Nkrumah University of Science and Technology (KNUST), Kumasi, Ghana
Correspondence to: George Y. Obeng, Technology Consultancy Centre, College of Engineering, Kwame Nkrumah University of Science and Technology (KNUST), Kumasi, Ghana.
| Email: |  |
Copyright © 2016 Scientific & Academic Publishing. All Rights Reserved.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

We investigated the effect of repetitive use of vegetable cooking oil using redfish (Lutjanus fulgens) as the food item that was fried with frytol cooking oil for 5 days in 6 batches of frying/day. The study results showed free fatty acid values of 2.407±0.32% and acid values of 4.815±0.605 mg KOHg-1, which were significantly higher compared to the Codex Alimentarius Commission standards. Peroxide values of 14.70 to 25.250 ± 2.179 meq O2/kg deviated from the Codex standard value of 10 meq O2/kg. The mean specific gravity of the oil also decreased from 0.901 (pure oil) to 0.887. The results showed degradation in the physico-chemical properties of the vegetable cooking oil after repetitive frying. We conclude that repetitive use of vegetable oilsafter 1-2 times of batch frying up to 250kg/batch should be avoided since changes in physico-chemical properties of the oil affect oil quality for human consumption. Oils not suitable for human consumption would be suitable for alternate uses including biodiesel production.
Keywords: Frying, Vegetable oil, Physico-Chemical properties, Oil quality, Ghana
Cite this paper: Nancy Idun-Acquah, George Y. Obeng, Ebenezer Mensah, Repetitive Use of Vegetable Cooking Oil and Effects on Physico-Chemical Properties – Case of Frying with Redfish (Lutjanus fulgens), Science and Technology, Vol. 6 No. 1, 2016, pp. 8-14. doi: 10.5923/j.scit.20160601.02.
Article Outline
1. Introduction
- Frying is an intense process that induces a multitude of chemical reactions in the frying medium and generates a plethora of chemical compounds [1]. In frying, which ever type the oil is, it is usually heated to about 170oC to 220oC. When the oil is heated to these temperatures in the presence of oxygen (air), the oil undergoes thermal, physical and chemical degradation by reaction [2]. There are potential health hazards generated by the consumption of oxidized products resulting from oils used repeatedly in frying processes [3].Degraded products may include free fatty acids, hydro-peroxides and polymerized triglycerides [4]. The amount of degraded products increases with the duration of heating at high temperature. Thus, the more the oil is heated, the more degradation takes place and the more toxic compounds are formed in the oil [5]. The degree of changes in the physical and chemical properties of the oil dictates the quality of the oil for human consumption.In Ghana, food items that are cooked by frying include fish, meat, chicken, ripe plantain, yam, cocoyam, pastries etc. Unfortunately, most people use vegetable cooking oil numerous times for frying before discarding so as to cut down on cost. Research studies have indicated that vegetable cooking oils are used 3-6 times before being discarded as waste [6]. Repetitive use of vegetable oil lowers the smoke point, which is the temperature at which the oil breaks down creating acrolein, an obnoxious smelling compound; and visible fumes are given off [7]. This makes the oil smoke at a lower temperature in heating [8]. Exposure of cooking oil to high temperatures for long period of time generates lipid peroxidation products that may be harmful to human health [9]. The presence of excess polar compounds in repeatedly used frying oil has been associated with increased risk of developing hypertension [10]. Consumption of repeatedly heated cooking oil might increase the risk of developing atherosclerosis. Lipid peroxidation products induce oxidative stress in endothelial cells, resulting in endothelial dysfunction that could eventually lead to the formation of atherosclerosis [11]. Consumption of repeatedly heated cooking oil is also associated with increased total serum lipid and low density lipoprotein (LDL) levels [12]. Moreover, thermally oxidized lipids enhance peroxidation of membrane macromolecules, contributing to their mutagenicity and genotoxicity which could potentially lead to carcinogenesis [13].
1.1. Objectives
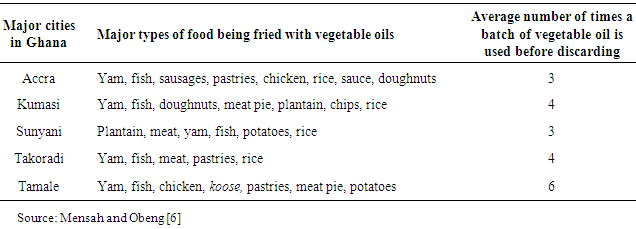
- The objectives of this study were to: (1) determine the effect of repetitive use of vegetable oil on its quality by determining the physico-chemical properties of the oil before and after frying redfish which is among the common food items fried by households and commercial caterers; and (2) compare the test results with internationally accepted standards - Codex Alimentarius Commission Standards.The justification for this study is based on previous research carried out by Obeng and Mensah [6]. The research results as shown in Table 1 indicated that in Ghana the average number of times a batch of vegetable cooking oil is used before being discarded ranged between 3 and 6. The continuous use of vegetable cooking oil results in decomposition, which affects the physical and chemical properties of oil quality for human consumption. Therefore, there was the need to analyse and understand the effect of repetitive use of vegetable oils on physicochemical properties.
|
2. Materials and Methods
- Eight (8) litres of vegetable cooking oil (Frytol brand of palm oil parent stock) and 7.5 kg of redfish (Lutjanus fulgens) popularly known as (Golden African Snapper) were obtained from the central market in Kumasi, Ghana. All reagents used for analysis were obtained from Pakus Ventures in Kumasi. The experiment was carried out at the project laboratory of Department of Chemistry in Kwame Nkrumah University of Science and Technology (KNUST), Kumasi, Ghana.
2.1. Frying Condition and Experiment
- 8 litres of Frytol oil was used for the experiment. The oil was heated to 180°C to fry 250g of redfish per batch per day. Six batches of 250 g of fish per batch were fried in each day; thus the oil was used six times for frying each day. This means that for the five days of frying, 30 batches of fish were used. During the frying experiment, there was no topping up of oil. Samples of oil were taken after frying each batch of fish. The oil samples were frozen at a temperature of -4ºC for 7 days before they were analyzed. The following physico-chemical parameters were focused for analysis: temperature and smoke point, acid value, percentage free fatty acid, peroxide value and specific gravity.
|
2.2. Determination of Acid Value of the Oil
- The acid value of the oil samples were determined by titration method. Two (2) grams of the oil was weighed into a 250 ml conical flask. 50 ml of neutralized ethyl alcohol was added to the oil sample. The mixture was then heated in a water bath. The solution was titrated against 0.1 M KOH using phenolphthalein as indicator. The acid value was calculated using the following formula:
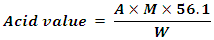 Where,
Where,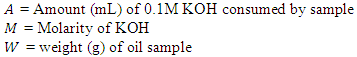 Source: WHO -The International Pharmacopoeia [14]
Source: WHO -The International Pharmacopoeia [14]2.3. Determination of Percent Free Fatty Acid
- The percent free fatty acid of the oil samples was obtained from the acid values of the oil samples as follows:
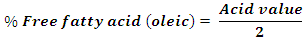 Source: Da Tech [15]
Source: Da Tech [15]2.4. Determination of Specific Gravity of the Oil by ASTM D854
- The specific gravity of the oil samples was determined using a 25 ml specific gravity bottle. The specific gravity bottle was weighed empty and then filled with the oil sample up to the mark on the bottle. The bottle with oil sample was weighed again. The weight of the empty bottle was subtracted from the total weight of the bottle and oil sample. The weight of the empty bottle was divided by the total weight of the bottle and oil to obtain the weight of the oil sample. The weight of the oil sample was then divided by the weight of an equal volume of water to get the specific gravity of the oil sample.
2.5. Determination of the Peroxide Value of Oil Samples
- The peroxide values of the oil samples were determined by titration method. One gram of oil and one gram of potassium iodide were weighed into a clean dry boiling tube.20 ml of solvent mixture (two volumes of glacial acetic acid with one volume of chloroform) was added to the sample. The boiling tube was placed in a water bath and the liquid boiled for 30 seconds. The content of the tube was quickly transferred into a conical flask containing 20 ml of 5% potassium iodide solution. The tube was washed twice with 25 ml of distilled water and collected into the conical flask. The content of the conical flask was titrated against 2 X 10-3 N Sodium thiosulphate solution until yellow color of the sample almost disappeared. 0.5ml of starch solution was added to the sample, shaken vigorously and titrated against the 2 X 10-3N Sodium thiosulphate solution until the blue colour disappeared. A blank was also set at the same time. The peroxide values of the oil samples were calculated using the following formula:
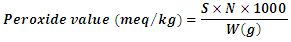 Where,
Where,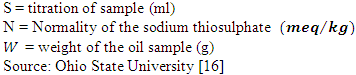
2.6. Statistical Analyses
- The results obtained from the study were statistically analysed using Microsoft excel 2010 with focus on the physico-chemical properties mentioned above. A significance level of 5% (p<0.05) was used for all analyses.
3. Results and Discussion
3.1. Temperature and Smoke Point of the Oil
3.1.1. Temperature
- From the graphs in Figure 1, it is observed that the temperature of the vegetable cooking oil increased with time in all the days of the frying experiment. Overall, it took 80-105 minutes to attain oil temperatures of 160-190°C. Relative to the other experimental days, on the fifth day smoke was observed after about 30 minutes of frying. After 30-50 minutes the oil temperatures gradually decreased and then increased until the end of the experiments.
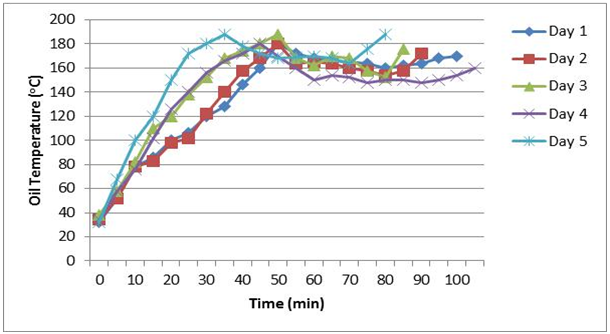 | Figure 1. Oil temperature and time used for frying |
3.1.2. Smoke Point of the Oil
- Figure 2 shows a graph of oil temperature and time for frying, which was recorded in every 5 minutes over a frying period of 100 minutes. In the first 20 minutes, the oil temperature increased from 32°C to 150°C. Thereafter the oil temperature continued to increase until it reached its smoke point of 190°C 30 minutes after frying. Most vegetable cooking oils have smoke points between 165°C and 260°C [17, 18]. The smoke point of any oil is the temperature at which that particular oil begins to burn and produce smoke [18]. This is the temperature the oil begins to break down to glycerol and free fatty acids [19]. The glycerol is then further broken down to acrolein which is a component of the smoke. It is the presence of the acrolein that causes the smoke to be extremely irritating to the eyes and throat. The smoke point also marks the beginning of both flavour and nutritional degradation [19].
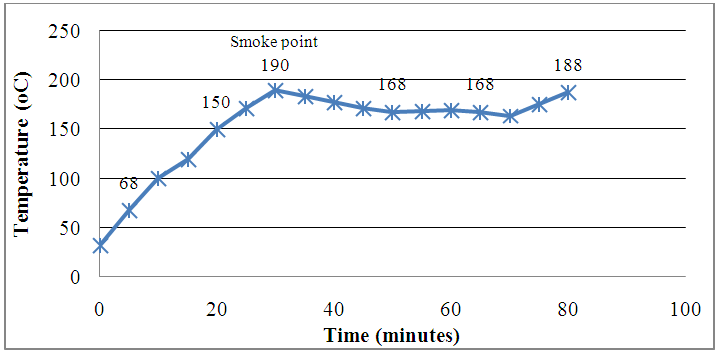 | Figure 2. Oil temperature, smoke point and time for frying |
3.2. Acid Value of the Oil
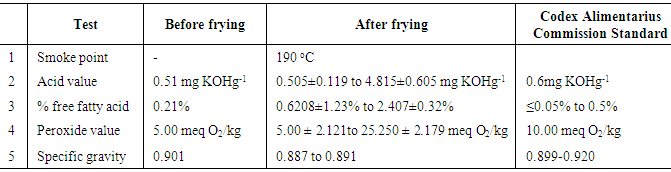
- The acid value of the oil increased with time from 1.24 mg KOHg-1 to 4.82 mg KOHg-1 as shown in Figure 3. The mean acid value measured after the frying experiment in Day 1 was 1.24 mg KOHg-1. This value is higher than the acid value of 0.51 mg KOHg-1 for the pure oil sample (Figure 3). The highest mean acid value of 4.82 mg KOHg-1 was recorded on the fifth day of frying. The acid values obtained from the oil samples ranged from 0.505±0.119 mg KOHg-1 to 4.815±0.605 mg KOHg-1. With the exception of the pure oil sample, the oil samples exhibited significant deviation from the standard set by Codex Alimentarius Commission (0.6mgKOHg-1) for refined oils for human consumption [20, 21]. Studies have shown that high acid values translate to high free fatty acid. These high free fatty acids have the tendency to increase the risk of coronary heart disease by raising cholesterol levels [22].
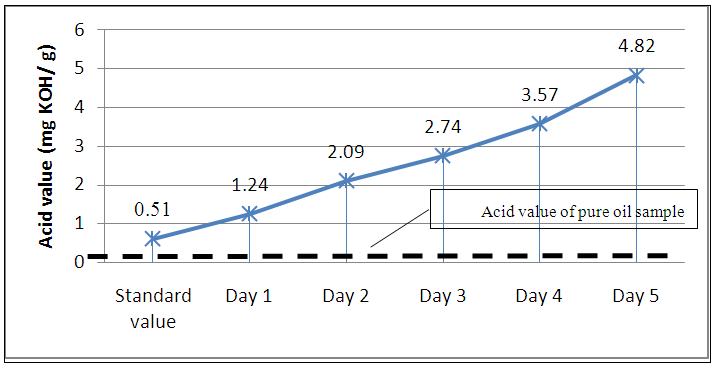 | Figure 3. Acid value of oil and time for frying |
3.3. Percent Free Fatty Acid of the Oil
- The percent free fatty acid (% FFA) of the oil before the commencement of the frying experiment was measured to be 0.21%. A study conducted by Al-Fatlawi and Abbas [23] revealed similar results. The study results indicated that %FFA ranged from 0.6208±1.23% to 2.407±0.32%. The %FFA value of 0.6208 ± 1.23% is higher than the standard value of ≤ 0.05% to 0.5%. The deviation in the values may be attributed to the high temperatures attained by the oil as well as the water from the fresh fish causing the natural fatty acids in the oil to get hydrolyzed into free fatty acids.
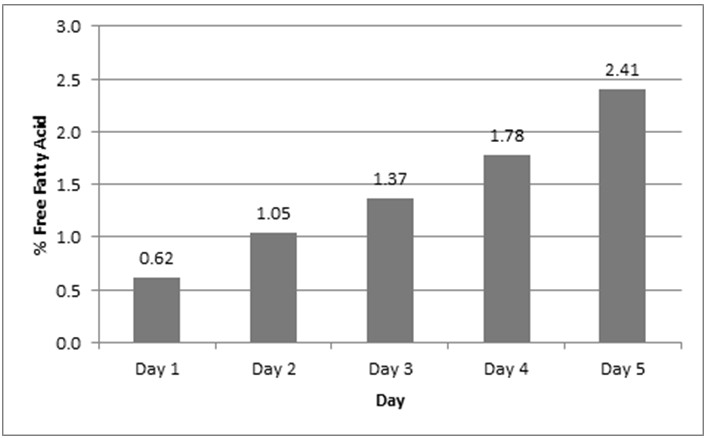 | Figure 4. Free fatty acid of oil and time for frying |
3.4. Peroxide Value of the Oil
- In Figure 5 the peroxide values obtained ranged from 10.00 ± 2.121 meq O2/kg to 25.250 ± 2.179 meq O2/kg. The average peroxide value for the pure oil was 5meq/kg O2/kg and the value measured after the first day of frying was 10 meq O2/kg. The remaining values obtained from Day 2 to 5 deviated significantly from the standard value of 10 meq O2/kg [20, 21]. The deviations from the standard value could possibly be due to the continuous exposure of the oil to light, high temperatures and atmospheric oxygen, which reacts with the oil to form peroxides.
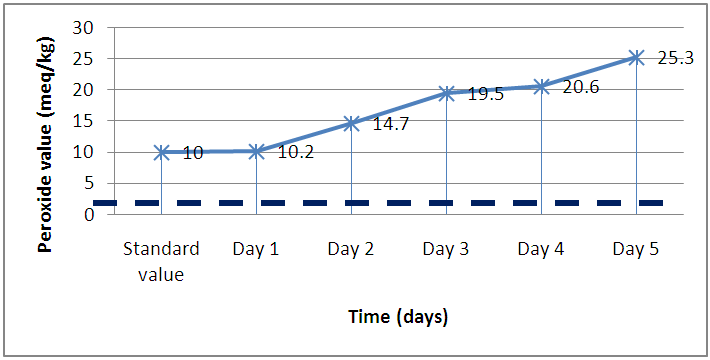 | Figure 5. Peroxide value of oil and time for frying |
3.5. Specific Gravity of the Oil
- Specific gravity is the heaviness of a substance compared to that of water, and it is expressed without units. The results in Figure 6 indicated that the mean specific gravity of the frytol oil decreased from 0.899 to 0.887. Overall the graph shows that the specific gravity decreased as the number of Trials and days increased, particularly in Day 1.
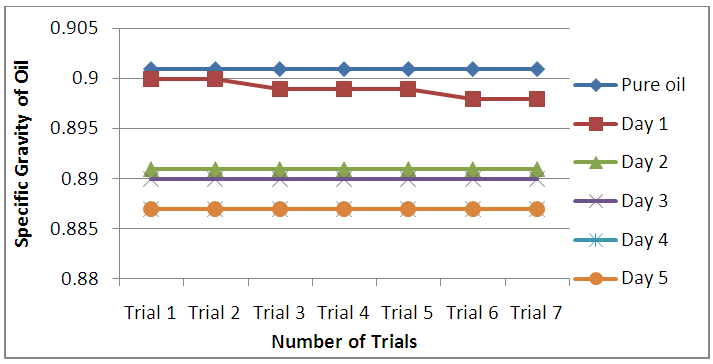 | Figure 6. Specific gravity of the oil |
4. Summary and Conclusions
4.1. Summary of Test Results
- Table 3 is a summary of the tests carried out and the various results before and after frying. The results obtained were also compared to the Codex Alimentarius Commission [20, 21] standard for quality validation.
|
4.2. Conclusions
- The physico-chemical properties of frytol cooking oil before and after frying with redfish were determined. The results obtained were compared with international standards - Codex Alimentarius Commission Standards. The acid values, percent free fatty acid, peroxide values and specific gravity values obtained after the second batch frying deviated from the Codex Alimentarius Commission Standards.Overall, the study results showed degradation in the physico-chemical properties of the vegetable cooking oil after repetitive use. Of significance is the fact that repetitive use of vegetable oils, particularly after 1-2 times of batch frying up to 250kg/batch should be avoided since changes in physico-chemical properties of the oil affect oil quality for human consumption. Oils not suitable for human consumption would be suitable for alternate uses including biodiesel production.
ACKNOWLEDGEMENTS
- The authors wish to acknowledge the Departments of Chemistry, Agricultural Engineering and Technology Consultancy Centre (TCC) all of KNUST, Kumasi, Ghana for their support.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML