-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Resources and Environment
p-ISSN: 2163-2618 e-ISSN: 2163-2634
2015; 5(2): 66-71
doi:10.5923/j.re.20150502.03
Isolation and Characterization of Isolate Thermophilic Bacteria from Water and Solid Sediment of Lapindo Mud
Emad Eldin Amar Dagdag1, Sukoso2, 3, Asthervina WP3
1Doctoral Program of Environmental Studies, Graduate School, University of Brawijaya, Malang, Indonesia
2Graduate School, University of Brawijaya, Malang, Indonesia
3Faculty of Fisheries and Marine Sciences, University of Brawijaya, Malang, Indonesia
Correspondence to: Sukoso, Graduate School, University of Brawijaya, Malang, Indonesia.
| Email: |  |
Copyright © 2015 Scientific & Academic Publishing. All Rights Reserved.
The purpose of this study was to investigate the characteristics of two isolates thermophilic bacteria A1 and S1 isolated from mud water and solid sediment of Lapindo Sidoarjo in term of morphological and biochemical properties and to suspect A1 and S1 isolates based on biochemical tests (microbact identification kits) which were compared to the 16S rDNA test in Kagoshima University laboratory. The method used is exploratory method. The research was conducted by the isolation of dominant bacteria from water samples Lapindo mud and sediment solid as isolates A1 and S1, then characterized by morphological and biochemical properties by using multiple test, i.e. a bacteria colony morphology and cell test, and biochemical tests which were compared to the test results of 16S rDNA with the physical and chemical environment parameters of the Lapindo mudflow in Sidoarjo. Isolation results of A1 and S1 isolates show that the bacteria belong to the group of thermophilic bacteria being able to grow at temperatures over 45°C which are known to have a thermo-stable enzyme which is not easily denatured at high temperature. From the A1isolate, Marinobacter lutaoensis was found based on the test results of 16S rDNA, while the biochemical properties similar to Enterobacter gergoviae referring to the bacterial identification guidelines (Bergey's Manual of Determinative Bacteriology). Moreover, from the S1 isolate, Marinobacter hydrocarbonoclasticus was obtained based on the test results of 16S rDNA and the biochemical properties similar to Klebsiella rhinoscleromatis referring to the bacterial identification guidelines (Bergey's Manual of Determinative Bacteriology). This result indicated that molecular approach more sensitive compare with the biochemical test, because the molecular approach use the gene area while the biochemical test use cell or individual as the object to evaluate the reaction test.
Keywords: Thermophilic bacteria, Mud Water and Solid Sediment, Lapindo Sidoarjo
Cite this paper: Emad Eldin Amar Dagdag, Sukoso, Asthervina WP, Isolation and Characterization of Isolate Thermophilic Bacteria from Water and Solid Sediment of Lapindo Mud, Resources and Environment, Vol. 5 No. 2, 2015, pp. 66-71. doi: 10.5923/j.re.20150502.03.
Article Outline
1. Introduction
- Bacteria are unicellular microbes. In general, bacteria do not have chlorophyll. There are some that are photosynthetic and reproductive asexual cleavage. Bacteria are widely distributed in nature, in the ground, in the atmosphere, in the deposits of mud, in the sea mud, in the water, in the hot springs, in the Antarctic region and in animals, humans and plants. The number of bacteria depends on the circumstances. For example, the number of bacteria in the soil depends on the type of soil fertility [1].Thermophilic microbes are microbes that can grow optimally in extreme heat environments, namely geothermal areas on land or in the ocean. Based on their growth temperature range, microbes are grouped into psychrophilic microbes that grow at -3°C to 20°C; mesophilic microbes which grow at 13°C to 45°C and thermophilic microbes which grow at 42°C to over 100°C [2]. Indonesia is a country that has a geographical situation possible to contain thermophilic bacteria because Indonesia has many volcanoes and hot springs [3].Thermophilic bacteria can be used in the field of biotechnology because it has a high efficiency in the ambient temperature. The higher the temperature, the higher the rate of diffusion is. In addition, the enzyme is also able to catalyze the thermophilic bacterium biochemical reactions at high temperatures and is generally more stable than mesophilic bacteria. Enzymes found in thermophilic bacteria can also be used in industry, among others, the enzyme amylase, cellulose, xylonite, chitins and protease [4]. Thermophilic microorganisms have the ability to withstand high temperatures because of the thermostable enzyme [5]. In addition, the proteins found in cells of thermophilic microorganisms have very strong hydrophobic and ionic bonds. The composition of the cell membrane is composed of thermophilic bacteria in saturated fat so it can be stable at high temperatures [6].Microorganisms are the source of most widely used enzyme compared to plants and animals. As a source of enzymes, microorganisms more profitable because of their rapid growth, ability to grow on cheap substrates, easier improved results by setting the growth conditions and genetic engineering, and capability of producing extreme enzymes [7].Lapindo hot mudflow is located in Sidoarjo, East Java Province. This geological event happened in the oil drilling site owned by PT Lapindo Brantas. The overflow of hot mudflow until now cannot be stopped and Lapindo Sidoarjo mud flow is still channelled directly to the Porong River. The overflow of the Lapindo mudflow was assumed to contain thermophilic bacteria that are capable of living in extreme environmental conditions of Lapindo mud. The thermophlic bacteria are able to produce thermostable enzyme that can be used in the fishery product processing industry, especially in the processes that require high temperature.In this study, there are three objectives, namely, to determine the characteristics of the bacterial isolates A1 and S1 isolated from mud water and solid sediment of Lapindo Sidoarjo, both morphological and biochemical properties; to suspect the dominant bacterial species (A1 and S1) which able to live in conditions of muddy water and solid sediment of Lapindo Sidoarjo using biochemical tests (microbact identification kits) and to compare the result with the 16S rDNA test conducted by Sukoso in Kagoshima University laboratory as well as preliminary information to determine the role of thermophilic bacteria to the processing of fishery products.
2. Research Methods
2.1. Materials and Equipments
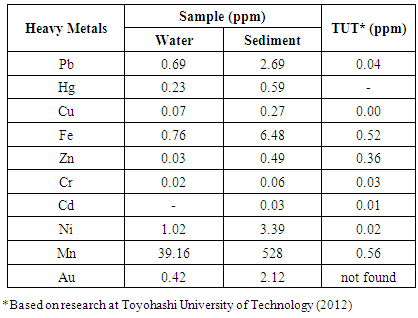
- The main materials used were Lapindo mud water and sediments obtained from the Lapindo mudflow in Sidoarjo. The grower media to indentify dominant bacteria in Lapindo mudflow in consisted of NA merck (Nutrient Agar), NB merck (Nutrient Broth). The materials in bacterial biochemical testing included: TSIA Agar (Triple Sugar Iron Agar), MRVP Agar (Methyl Red and Voges Proskauer), Nitrat Agar, Urea Broth, Simmon Sitrat, SIM Agar (Sulfide Indol Motillity), KOH 40%, -naftol, Methyl Red, Kovac, and distilled water. Materials in gram staining, among others included: Purple Crystal, Acetone, Iodine, Safranin, Yellow Tip, and Blue Tip. The materials in the testing of heavy metals in the sample included: HCl + HNO3 (Aqua reagent), HNO3 (Nitric Acid), ((NH4) 2S2O8) Ammonium per sulphate, H3 PO4 (Phosphorus Acid), NaIO4 (Sodium Periodate), [(C6H6) NHNH] 2CO (Diphenylcarbazide), (CH3CNOH) 2 (Dimethylglyoxime), Iodine, and Ammonium Citrate. In addition, the supplementary materials included filter paper, Whattman paper no. 42, alcohol 70%, spirits, masks and aluminum foil gloves, plastic wrap, cotton, and wipes.The equipment used in this study consisted of Naire Laminar Air Flow (LAF), Pyrex scott bottle, Pyrex test tube, test tube rack, Pyrex brand petri dish, Pyrex pipette with the volume of 10 ml, bulb suction, Pyrex Erlenmeyer glass with volumes of 250 ml, 500 ml and 1000 ml, Pyrex measuring cup100 ml, Pyrex funnel, Pyrex beaker glass 500 ml and 1000 ml, spatula, Sanyo autoclave, digital scales brand Mettler Toledo, trays, SI-600R incubator shaker, loop needle, ose needles, stove, sprayer, Avi-Teck micropipette, a Luton YK-2001PH pH meter, Oxoid thermometer, pans, Bunsen, cooling box, Oxoid microbact identification kits, Shimadzu Atomic Absorption Spectrophotometer, Thermo Spectronic spectrophotometer, Memmert incubator, cruisable Tunk, washing bottle, Olympus microscope, Toshiba refrigerator, object glass, cover glass, Bunsen, Olympus camera, and refract meter.
2.2. Sampling
- Samples were taken from the hot mudflow in Sidoarjo and the laboratory test was conducted at the University of Brawijaya, Malang, in the Laboratory of Microbiology, Faculty of Medical. The entire research activities were carried out in June 2014 to December 2014.
2.3. Data Analysis
- The method used in this research is exploratory. The first phase starts with sampling around the Lapindo mudflow in Sidoarjo by taking two samples, e.g. water and solid sediment of Lapindo mud. The second phase is measuring the physical and chemical parameters of the Lapindo mudflow environment with four parameters: temperature, pH, salinity, and levels of heavy metals. The third stage is mass bacterial cultures in which taken water samples were filtered with paper sludge whattman 42 and the solid sediment. The fourth stage is to isolate the target bacteria in each sample by selecting the dominant bacteria grown and planted with cast method in duplicate in NB medium (Nutrient Broth). The fifth stage is the bacteria characteristic tests which include bacterial colony morphology test (colony form, colony color, colony edge, and colony elevation), bacteria morphology cell test (Gram stain and bacterial cell shape) with a microscope with a magnification of 1000x, biochemical manual test and biochemical tests with Microbact Identification Kits. The molecular approach was conducted by isolating 16S rDNA and sequencing. The description of 16S rDNA nucleotides was done by using BLASTN 2.2,27+ program.
3. Results and Discussion
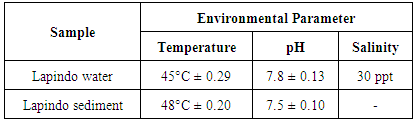
3.1. Environmental conditions of Lapindo Mudflow, Sidoarjo
- Table 1 indicates that the temperature of the mud water and solid sediment of Lapindo is high temperatures due to the Lapindo mudflow originating from the earth. This is in accordance with Koesoemadinata [8] mentioned that many experts stated Lapindo mudflow as an analogy of natural phenomena called mountain/volcano mud that is widely spread in Indonesia. Mud volcano is a volcano of cone-shaped mud clay and small mud volcanoes, generally less than 1-2 m in height. Small mud volcanoes are formed from a mixture of hot water and fine sediments (clay and silt) where there is (1) a slow flow from a hole like a stream of molten lava; or (2) sprayed into the air like a fountain of lava that releases boiling water and volcanic gases.
|
|
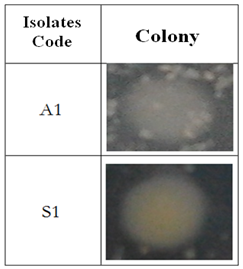
3.2. Colony Morphology of Dominant Bacteria in Lapindo Mudflow, Sidoarjo
- In Table 3, morphology of colony isolates A1 shows a rounded shape, white thin and flat edges with a flat cell elevation. While the colony morphology of bacterial isolates S1 has a smoothly rounded shape, elevation of cells arises, white to soft yellow colour; in which the yellow colour is inside and white colours is found in the outer layer of bacteria; thus the bacterial cell edge layered looks flat because there are two layers of colour.
|
3.3. Characteristics of Dominant Bacteria
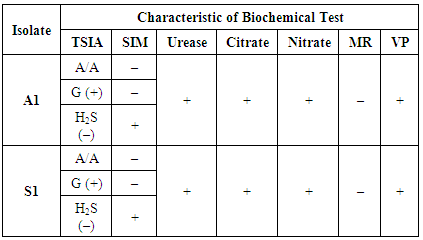
3.3.1. Manual Biochemical Test
- Isolates with code A1 and S1 has similar biochemical properties as showed in Table 4. TSIA test showed that A1 and S1 isolates are able to ferment glucose, lactose, and sucrose with or accompanied by the formation of acid and gas which is characterized by the formation of yellow colour at the bottom of the "butt" and the slope of the "slant" on TSIA media, but the bacteria cannot form sulphur or H2S gas from the hydrolysed amino acids because there is no blackish coloration on the media. SIM test showed that A1 and S1 isolate is not able to break down the amino acid tryptophan because it is not formed on the surface of the red ring of SIM medium, and the A1 isolate is not capable of forming sulphur compounds. It is proved because there is not any black colour on the SIM media, but there is movement or motility in the media characterized by a colour change in the puncture medium showing cloudy marks. In urea test, it is found that A1 and S1 isolate is able to produce the urea enzyme, thus ammonia compounds are formed marked by the discoloration to pink on the urea media. In the citrate test showed that A1 and S1 isolates is able to use citrate as sole carbon source characterized by medium colour change from green to blue. On the nitrate test, it shows that A1 and S1isolate is able to reduce nitrate to nitrite characterized by the occurrence of turbidity changes in nitrate broth media. The MR test result shows that A1 and S1 isolate is not capable of oxidizing glucose to produce high acid concentrations which is characterized by the absence of colour change in the MRVP media. The VP test moreover describes the result that A1 and S1 isolate is capable of forming acetyl carbine as a result of carbohydrate metabolism characterized by the red colour on the MRVP media.
|
3.3.2. Microbact Identification Kits
- From the A1 isolate, the obtained results include: lysine test (+), ornithine test (+), H2S test (-), glucose test (+), mannitol test (+), xylose test (+), ONPG test (+), test indole (-), urea test (+), VP test (+), citrate test (+), and TDA test (-).From S1 isolates, the obtained results are: test lysine (-), ornithine test (-), H2S test (-), glucose test (+), mannitol test (+), xylose test (+), ONPG test (-), test indole (-), urea test (-), VP test (-), test citrate (-), and TDA test (-).
3.4. Estimation of Dominant Bacteria Type
- Table 5 indicates that when using a biochemical test (Microbact Identification Kits) which refers to the bacterial identification guidelines (Bergey's Manual of Determinative Bacteriology), the results show that the suspected bacterial isolates code A1 is Enterobacter gergoviae and compared with the results obtained from 16S rDNA test, it is found Marinobacter lutaoensis. Meanwhile, by using biochemical tests (Microbact Identification Kits) which refers to the guidelines for the identification of bacteria (Bergey's Manual of Determinative Bacteriology), the results show that species of Klebsiella rhinoseleromatis bacteria are obtained from the bacterial isolates with code S1 and compared with the results of the 16S rDNA test, the results species of the bacterial isolates were Marinobacter hydrocarbonoclasticus.
|
3.5. Role of Bacteria Thermopile on Fishery Products
- According to the research, the water and sediment of Lapindo Sidoarjo mud contain the dominant bacterium groups which are thermophilic because both A1 and S1isolates are able to live above 45°C. Thermopile bacteria are known to have a thermo-stable enzyme, an enzyme that is able to survive or stable at high temperatures (6-120°C).According Girindra [15], in a small number, enzymes can regulate certain reactions so that in normal circumstances there is no deviation of final reaction results. This enzyme will lose activity due to heat, acids or strong bases, organic solvents, or anything that causes protein de-naturalization, since all enzymes are proteins. Therefore, in the fishing industry, thermo-stable enzyme has a good role in the processing of food stuffs in the fishery, especially the use of high temperatures in the processing, for example in the manufacture of chitin and chitosan.Chitin is a complex molecule that is normally found in animals of the group of crustaceans such as shrimp, lobster and crab. Chitin can be obtained by grinding the shrimp shells and adding NaOH to remove proteins and HCl to remove minerals (demineralization), after that the drying process is carried out [16]. Until now there are two waste treatment ways to obtain chitin: 1) chemical processing by means of demineralization and de-proteinization through the addition of strong acids or bases, and 2) biochemical processing by the addition of proteolytic enzymes to de-proteinize and involves chitins to degrade chitin waste [17].In addition, the role of thermostable enzymes can also be applied in the manufacture of chitosan from chitin in shrimp or crab shells (Crutacea). In the process of making chitosan, chitin deacetylase enzyme plays a role in the changing process from chitin into chitosan. When chitin is proceeded into chitosan, deacetylation process of chitin should be done, when the process of deacetylation is being conducted, acetyl group needs to be eliminated while still maintaining its amine groups. The process can be done chemically or enzymatically. chemical processes are carried out by soaking the deacetylation of chitin in NaOH 50% whose weight is 10 times the weight of the sample and then heating to a temperature of 116-120°C for 2 hours [18]. On the other hand, the enzymatic deacetylation process can be done by using deacetylation of chitin enzyme (EC 3.5.1.41) derived from microorganisms such as Mucor rouxii [19], E. coli and Colletotrichum lindemuthianum [20]. Moreover, Yamasaki et al. [21] added that some microorganisms can produce enzymes with chitinolytic activity (chitin deacetylase), such examples that have been isolated are from Enterobacter sp.Based on the description, it is found that thermophilic bacteria enzymatically in the process of deacetylated chitin chemical substitutes for chemical deacetylation process has the disadvantage that is not environmentally friendly and chitosan final results are not uniform, it is different from the results of the enzymatic deacetylation if done with bacteria Thermopile, because by using less enzyme deacetylation of chitin deacetylase then the process can take place in addition to the already environmentally friendly and chitosan end result is more uniform due to the nature of specific chitinase enzyme in severing ties acetyl group.
4. Conclusions
- A1 and S1 Isolates belong to the group of thermophilic bacteria because it is able to grow at temperatures over 45°C and is known to have a thermostable enzyme which is not easily denatured at high temperatures, which can be used in the processing of fishery products such as the manufacture of chitin and chitosan.A1 isolates contains Marinobacter lutaoensis which was obtained by 16S rDNA test results conducted by Prof. Ir. Sukoso M.Sc, Ph.D and has biochemical properties similar to Enterobacter gergoviae which refers to the bacterial identification guidelines (Bergey's Manual of Determinative Bacteriology). Otherwise, S1 isolates was found as Klebsiella rhinoseleromatis compared with the results of the 16S rDNA as Marinobacter hydrocarbonoclasticus.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML