-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Plant Research
p-ISSN: 2163-2596 e-ISSN: 2163-260X
2012; 2(5): 146-150
doi: 10.5923/j.plant.20120205.02
Proximate, Mineral and Anti-nutrient Composition of Pumpkin (Cucurbitapepo L) Seeds Extract
Elinge C. M. 1, Muhammad A. 1, Atiku F. A. 1, Itodo A. U. 1, Peni I. J. 2, Sanni O. M. 1, Mbongo A. N. 3
1Department of Pure and Applied Chemistry Kebbi State University of Science and Technology Aliero Nigeria.
2Department of Animal Science Kebbi State University of Science and Technology Aliero
3Department of Microbiology and Biochemistry, University of Buea, Cameroon
Correspondence to: Elinge C. M. , Department of Pure and Applied Chemistry Kebbi State University of Science and Technology Aliero Nigeria..
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
Pumpkin seeds were analysed for their nutritional and anti-nutritional composition, the results obtained were; moisture content (5.00%), ash (5.50%), crude lipid (38.00%, crude fibre (1.00%), crude protein (27.48%), Available carbohydrate (28.03%) and calorific value (564kcal/100g). Elemental analysis shows that potassium is the most abundant element in the sample (273mg/100g) and manganese is least (0.06mg/100g).The anti-nutritional parameters analysed are; phytate (35.06 mg/100g), oxalate (0.02±0.10mg/100g), hydrocyanic acid content (0.22±0.04mg/100g) and nitrate (2.27±002mg/100g). The result shows that the pumpkin seeds if properly utilized can serve as good source of minerals.
Keywords: Pumpkin, Cucurbita Pepo L, Seeds, Proximate, Mineral, Anti-Nutritional, Nutritional
Article Outline
1. Introduction
- From the family Cucurbitaceae, is Cucurbita pepo, (Common name: Pumpkin; Yoruba: Elegede), a medium sized plant grown for its Fruits and edible seeds. Hence, it is known to be used as food and in herbal formulation in Nigeria.The ways of expanding the use of available local food sources are increasingly pursued, some of these local food sources contain seeds, many reports on some lesser known seeds and fruits indicated that they could be good sources of nutrients for both man and livestock[1].Seeds are good sources of protein, fats, carbohydrate and minerals[2].Researches have shown that seeds not only contain nutritionally important bio- compounds but are also sources of other phyto-compounds which at certain critical levels have significant anti-nutritional effects[3]. These compounds include oxalate, phytate, nitrate, cyanide etc.Pumpkin has received considerable attention in recent years because of the nutritional and health protective values of the seeds .Pumpkin seeds are consume directly as snack food in many cultures throughout the world[4].Pumpkin plant is an annual plant with leafy green vegetable; it has a climbing stem of up to 12m long and fruit with a round fibrous flesh[4]. Pumpkin fruits are variable in size, colour, shape and weight; they have a moderately hard flesh with a thick edible flesh below and a central cavity containing the seeds. The seeds are covered with a testa which serves as a protectant around it.More analysis on Pumpkin seeds will only increase the available data on their nutritional and anti-nutritional composition. However, little work has been reported on the mineral composition of pumpkin seeds varieties from Tudunwada area, Kaduna Nigeria, an area known for highly condensed industries including petrochemicals. Therefore, the main objective of the present study was to evaluate the proximate, minerals, nutritional and anti-nutritional composition of Pumpkin seeds as potential source of minerals as well as their calorific value and their health implications.
2. Materials and Methods
- Ripped fruits of cucurbita pepo L were obtained from different farm locations in Tudunwada area, Kaduna Nigeria. The samples were mixed together andrepresentative samples were picked at random[5].The fresh fruits were authenticated at the Herbarium unitof the department of Biological Science of Usman Danfodio University Sokoto, Nigeria. The sample fruits were clean with water and rinsed with distilled water to avoid surface contamination[6]. The flesh of the fruit and the seeds were separated using plastic spoon after dividing the fruit into two. The sample was dried at room temperature, crushed to a fine powder using mortar and pestle, sieved through 20-mesh and stored in an air tight plastic container for analysis.
2.1. Proximate Analysis
- Moisture content was determined at 105oC. Ash content was determined at 550o C. Crude protein, lipid and fibre were also determined according to the procedures of[7]
2.2. Crude Nitrogen
- Crude Nitrogen was determined based on the Kjeldhal procedure and crude protein value was obtained by multiplying the nitrogenvalue by a factor of 6.25 while estimation of available carbohydrate was done by difference as:Where CHO, CP and CL stands for carbohydrate, crude protein and crude lipid respectively[8].
2.3. Mineral Analysis
- The sample was digested into solution by wet digestion using a mixture of conc. Nitric, perchloric and sulphuric acids in the ratio 9:2:1 respectively. Fe, Zn, Co, Mg, Ca and Mn were determined by AAS, (Alpha 4 model, Buck Scientific Ltd USA). While Na and K were determined using atomic emission spectrometer (200-A model, Buck Scientific Ltd UK), and colorimetric method was used to determined Phosphorus.Other analysis and adopted methods include the determination of oxalate, phytate and hydrocyanic acid[7] and nitrate[9].
3. Results and Discussion
|
|
3.1. Proximate Composition
- The result showed that the seed has moisture content of 5.00 % which is lower than those of vegetable spaghetti seed 45.67±2.03[10] and gardenia aqualla seed 49.00[10]. The lower moisture content of the seed will give it a storage advantage The ash content of the seed is 5.50 %. Which is close to that of gardenia aqualla seeds 5.20%[14]. The percentage ash of the sample gave an idea on the inorganic content of the samples from where the mineral content could be obtained. The ash content obtained is similar to the range of 1.63- 8.53% reported for commonly consumed fruits by[11].Sample with high ash contents is expected to have high concentration of various mineral elements, which are expected to speed up metabolic processes, improve growth and development.The crude lipid content of the seed is within the range of 38.00% this could be classified as an oil seed like groundnut, melon e.t.c. Lipids are essential because they provide the body with maximum energy[11].The fibre content of the seeds is 1.0 % which compared favourably with that of Gardenia aqualla seeds[9]. Although, fibre containing food are known to expand the inside walls of the colon, easing the passage of waste, thus making it an effective anti-constipation, it lowers cholesterol level in the blood and reduce the risk of various cancers. But emphasis has been placed on the importance of keeping fibre intake low in the nutrition of infants and weaning children because high fibre levels in weaning diet can lead to irritation of the gut mucosa[2]. It also enhances gut perturbation in young animals (e.g. piglets and chickens)[12]. Comparatively, the fibre content of the seed is close to 1.23± 0.03 % as reported for Arthocarpusheterophyllus seed[2] The protein content of the seed was found to be 27.48% which shows that the seed can serve as a source of protein considering the level of protein deficiency in the society. The available carbohydrate content of the seed is found to be 28.03%. The sample could not be considered as potential source of carbohydrate when compared to the content of some conventional sources like cereals with 72-90 g/100g Carbohydrate[13].
3.2. Elemental Analysis
- Potassium is the most abundant element found in the seed (273mg/100g), High amount of potassium in the body was reported to increase iron utilization[14] and beneficial to people taking diuretics to control hypertension and suffer from excessive excretion of potassium through the body fluid[15]. The concentration of sodium in sample is 170 mg / 100g; this element is required by the body to regulate blood pressure and blood volume. It helps regulate the fluid balance in the body; it also helps in the proper functioning of the muscles and nerves[16]. The calcium content of the sample was found to be 9.78mg/100g Calcium helps to ease insomnia and helps regulate the passage of nutrients through cell walls, without calcium the muscles in the body cannot contract correctly, the blood in the body will not clot and the nerves will not carry message. If the body don’t get enough calcium from the food we eat. The body automatically takes the calcium needed from the bones. If the body continue to tear down more Calcium than it replaces over a period of years the bones will become weak and break easily[16] (Payne, 1990). Calcium ions are also necessary for the normal functioning of nerves and muscles[17].The concentration of Magnesium in the sample is 67.41±0.05mg/100g Magnesium is a constituent of bone and teeth and is closely associated with calcium and phosphorus. Magnesium is necessary for the release of parathyroid hormone and for its action in the backbone, kidney and intestine and for the reactions involve in converting vitamin D to its active form. Magnesium is important in tissue respiration, especially in oxidative phosphorylation leading to formation of Adenosine triphosphate (ATP). It is also involved in normal muscular contraction; calcium stimulates muscles while magnesium relaxes the muscles[18]. Magnesium deficiency results in uncontrollable twisting of muscles leading to convulsion and tetanus, which may both lead to death[17].The concentration of Phosphorus in the sample was estimated as 47.68±0.04mg/100g, phosphorus is found bound in the blood and cells, while most of the non-skeletal phosphorus is inorganic in the form of nucleic acids, phosphor-lipids, ATP and sugar phosphate[16] (Payne, 1990). Phosphates play important roles as buffers that prevent change in the acidity of body fluids because of their ability to combine with additional hydrogen ion. The combination with phosphorus makes it possible for nutrients to cross the cell membrane[18].This work gave Iron content of 3.75±0.02mg/100g, Iron performs several functions in the body; it helps in the formation of blood, it also helps in the transfer of oxygen and carbon dioxide from one tissue to another[18].Iron deficiency results in anaemia which impairs muscles metabolism, iron deficiency in children result in impaired learning ability and behavioural problems[19].The concentration of Cobalt in sample was found to be 2.17±0.02mg/100g, Cobalt plays a role in the metabolism of vitamin B-12 hence, increase body ability in its absorption. Cobalt is believed to function as an activating ion in some enzymes[19].Manganese was found to be the least among all the minerals analysed in this sample (0.06±0.01mg/100g). Manganese plays an important role in all mental functions and aids in the transfer of oxygen from lungs to cells, it is important as an activator for enzyme reactions concerned with carbohydrate, fat and protein metabolism[16].Manganese deficiency is rarely observed owing to the large amount of it in human diet, but in a situation where the deficiency occur it leads to retardation of growth and skeletal abnormalities. Too much accumulation of manganese is toxic in the blood and is mostly associated with weakness of the body and psychological and nervous system difficulties. Toxicity of manganese however occurs more often with iron deficiency[19].Pumpkinseed presented fairly high value for Zinc (14.14±0.02mg/100g). Zinc is known for boosting the health of our hair, it is believed to play a role in the proper functioning of some sense organs such as ability to tastes, sense and smell[16]. Zinc plays a very important role in protein and carbohydrate metabolism and also help in mobilizing vitamin A from its storage site in the liver and facilitates the synthesis of DNA and RNA necessary for cell production[18].
3.3. Antinutritional Composition
- The concentration of oxalate found in the seed is 0.023mg/100g which is not high. High oxalate diet can increase the risk of renal calcium absorption and has been implicated as a source of kidney stones[20]. The level of oxalate in the sample is not high to pose any health treat.The phytate content (35.06±1.10mg/100) of the seed is lower than that of Dannielleaoliveri kernel seed 51.05 mg/100g[8],but higher than that of Jatrophacurcas kernel seed 34.37 mg/100g[21]. The problem with phytate in food is that it can bind some essential mineral nutrients in the digestive tract and can result in mineral deficiencies[2]. The phytate composition of the sample might not pose any health hazard when compared to a phytate diet of 10-60mg/g which if consumed over a long period of time that has been reported to decrease bioavailability of minerals in monogastric animals[22].The concentration of Hydrocyanic acid in the seed is 0.22mg/100g this shows that the level of the acid in the sample is within the acceptable range for human consumption. Only plants with more than 200 mg of hydrocyanic acid equivalent per 100 mg fresh weight are considered dangerous[23]. Consumption of high levels of Cyanide is associated with a serious health problem, a neurological disease known as Tropical Ataxis Neurophathy (TAN) was linked to consumption of high level of cyanide in cassava based diet[24].The concentration of nitrate in the seed is 2.27 mg/100g. These values are within the acceptable daily intake of 3.7 mg/kg body weight (WHO). Higher concentration of nitrate in the food can lead to a disease called methemoglobianaemia which is known to reduce the ability of red blood cells to carry oxygen[25].It should be noteworthy that in addition to the importance of Pumpkin (Cucurbita pepo) arrayed in this work, the seed has also received considerable attention in recent years an excellent source of protein and also has pharmacological activities such as anti-diabetic, antifungal , antibacterial and anti-inflammation, activities and antioxidant effects[26].Presented elsewhere and below was the fatty acid composition of the crude lipid fraction of seeds of Cucurbita spp. of Nigerian origin with four fatty acids accounting for >97% of the fatty acid total: palmitic acid (C 16:0; 13.0%), stearic acid (C 18:0; 7.9%), oleic acid (C 18: 1 n-9; 45.4%) and the essential fatty acid linoleic acid (C 18:2n-6; 31%) Glew et al. 2006 in[28].
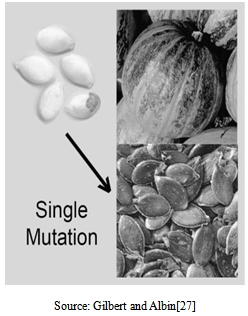 | Figure 1. Morphological differences between wild-type andhull-less pumpkin (Cucurbita pepo) seeds |
|
4. Conclusions
- It can be concluded that the seed is a good source of some protein and oil. From the highlights of the present study, we can also deduce that, these seeds may serve as constituents of minerals to animal and human through dieting. Generally, results and findings from this research agreed favourably with those of other wild plants recommended as food supplement in literature.
References
| [1] | Elemo, B. O., Elemo, G. N. Oladimeji, O. O. and Komolafe, Y. O. (2002). Studies on the Composition of Some Nutrients and Anti nutrients of Sheanut (Butyrospernumparkii) Nigerian Food Journal20: 69-73. |
| [2] | Bello, M. O., Farade, O. S., Adewusi, S. R. A and Olawore, N. O. (2008).Studies of Some Lesser Known Nigerian Fruits.African Journal of Biotechnology.7 (1): 3972-3979. |
| [3] | Omorayi, F. O. and Dilworth, L. (2007). Anti-nutritional Factors, Zinc,Iron and Calcium in some Cariggean tuber Crops and Effect of Boiling or Roasting. Nutrition and Food Science37 (1): 8-15. |
| [4] | Dietmar, F. (2005). Extract of Pumpkin Seeds Suppresses Stimulated Peripheral Blood Mononuclear Cell Invitro .American Journal of Immunology (1): 6-11 |
| [5] | Asaolu, M. F. and Asaolu, S. S. (2002). Proximate and Mineral Composition of Cooked and Uncooked Solanummelongena. International Journal of Food Science and Nutrition 3: 103-107. |
| [6] | Ahmed, U. and Birnin-Yauri, U. A. (2008). Comparative Analysis of the Nutrient Composition of Cashew (Anacardiumoccidentale) Apple and Nut.NigerianJournal of Basic and Applied Sciences16(1): 87-89. |
| [7] | AOAC (1990). Official methods of analysis (15th ed.). Washington D.C USA.Association of Official Analytical Chemists inch. 400- 2200Wilson Boalevard,Arlinton Virginia USA 2:910-928. |
| [8] | Hassan, L. G., Dangoggo, S. M. Umar, K. J. Saidu, I. and Folorunsho, F. A. (2008). Proximate, Minerals and Anti-nutritional Factors of Danielliaoliveri seed kernel.Chemclass Journal5: 31-36 |
| [9] | IITA (1988). Selected Methods for Soil and Plant Analysis (Manual series No.1) International Institute for Tropical Agriculture,Ibadan, Nigeria, Pp55-56. |
| [10] | DAngoggo, S.M., Mahammad, A., Aliero, A.I.,Tsafe, A.I. and Itodo, A.U.(2011). Proximate, Mineral and Antinutrient Composition of Gardenia aqualla Seeds. Achieves of Applied Sciences Research 3 (4): 485-492. |
| [11] | Oluyemi, E. A., Akilua, A. A., Adenuya, A. A. and Adebayo, M. B. (2006).Mineral Contents of Some Commonly Consumed Nigerian Foods.Science Focus.11(1):153-157. |
| [12] | Eromosele, I. C., Eromosele, C. O. and Kuzhkuzha, D. M. (1991).Evaluation of Mineral Elements and Ascorbic Acid Contents in Fruits of Some Wild Plants. Plant Food for Human Nutrition41:53- 57. |
| [13] | Adewusi, S. R. A., Udio, A. J., Osuntogun, B. A. (1995). Studies on the Carbohydrate Content of Bread Fruit (ArtocarpuscommunisForst) from south-western Nigeria.Starch Nutrition.85: 285-294. |
| [14] | Adeyeye, E. I. (2002). Determination of the Chemical Composition of the Nutritionally Valuable Parts of Male and Female Common West African Fresh Water Crab (Sudananoutesafricanus). International Journal of Food Sciences and Nutrition 53:189-196. |
| [15] | Arinanthan, V., Mohan, V. R. and Britto, A. J. (2003). Chemical Composition of Certain Tribal Pulses in South India. InternationalJournal of Food Sciences and Nutrition 3: 103-107. |
| [16] | Payne, W. J. A. (1990). An Introduction to Animal Husbandry in the Trophics. Longman Publishers Singapore Pp 92-110. |
| [17] | Hegarty, V. (1988). Decisions in Nutrition 5th edition. Time mirrowmosby LondonPp 80-132. |
| [18] | Guthrie, H. A. (1989). Introductory Nutrition (7th ed.). Time mirror Mosby college publishers, Boston. |
| [19] | McDonald, A., Edwards, R. A., Greenhulgh, F. D. and Morgan, C. A.(1995). Animal Nutriton.Prentices Hall, London Pp101-122. |
| [20] | Chai, W. Liebman, M. (2004). Assessment of Oxalate Absorption From Almonds and Black Beans With and Without the Use of an Extrinsic Label 172: 953-957. |
| [21] | Azza, A. A. and Ferial, M. A. (2010). Nutritional Quality of JatrophaCurcas Seeds and Effect of Some Physical and Chemical Treatments on Their Anti-nutritional Factors.African Journal of Food Science4 (3): 93-103. |
| [22] | Thompson, L. U. (1993). Potential Health Benefits and Problems Associated With Anti Nutrients in Foods. International Journal of Food Resources26:131-149. |
| [23] | Betancur-Ancona, D., Gallegos-Tintore, S., Ddelgado-Herrera, A., Perez- Flores, V., Ruelas, A. C. and Luis Chel-Guerrero, L (2008). Some Physicochemical and Anti-nutritional Properties of Raw Flours and Protein Isolates FromMucunapruriens(velvet bean) and Canavaliaensiformis (jack bean). International Journal of Food Science and Technology. 43: 816-823. |
| [24] | Hassan, L. G. and Umar, K. J. (2004). Proximate and Mineral Composition of Seeds and Pulp of African Locust Bean (Parkiabiglobosa). Nigerian Journal of Basic and Applied Sciences 13: 15-27. |
| [25] | Kim-Shapiro, D. B., Gladwin, M. T., Patel, R. P., and Hogg, N. (2005). Role of Nitrite in Haemoglobin –Mediated Hypoxic Vasodilation. Journal of Inorganic Biochemistry 99 (1):237-246 |
| [26] | Atuonwu, A.C. and Akobundu, E.N.T.(2010).Nutritional and Sensory Quality of Cookies Supplemented with Defatted Pumpkin (Cucurbita pepo) Seed Flour. Pakistan Journal of Nutrition 9 (7): 672-677. |
| [27] | Gilbert O. F. and Albin H.(2007). Seeds and oil of the Styrian oil pumpkin:Components and biological activities. Eur. J. Lipid Sci. Technol. 109 (2007) 1128–1140 |
| [28] | EMA- European Medicines Agencies (2011). Assessment report on Cucurbita pepo L. A report by the Committee on Herbal Medicinal Products (HMPC).Ref No.EMA/HMPC/136022/2010. Pp.4 |
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-Text HTML
Full-Text HTML