-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Public Health Research
p-ISSN: 2167-7263 e-ISSN: 2167-7247
2019; 9(3): 56-63
doi:10.5923/j.phr.20190903.03

A Systematic Review of Caregiver Cost of Alzheimer's Disease
Zahra Shahin , Ursula Spence , Samuel Opoku
Jiann-Ping Hsu College of Public Health Department of Health Policy and Community Health, Georgia Southern University, Statesboro, Georgia, USA
Correspondence to: Zahra Shahin , Jiann-Ping Hsu College of Public Health Department of Health Policy and Community Health, Georgia Southern University, Statesboro, Georgia, USA.
| Email: |  |
Copyright © 2019 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Introduction: Alzheimer's disease (AD) is a form of neurodegenerative disease that is progressive and irreversible and leads to loss of intellectual and cognitive functions. These changes are so severe that they can affect the everyday life of the individual to do simple things. Therefore, AD patients increasingly reliant on others, mainly family members. Informal care is provided by family caregivers, and that increases with disease progression over time. The cost of care for people with AD is a significant burden on both family caregiver and society. This systematic review of the literature explores the use and cost of informal care of dementia patients. Methods: A systematic review was conducted through electronic database to detect relevant studies on the cost of care for AD and other dementia population. A determination of the eligibility was made through assessment of the full-text articles. For an article to meet inclusion criteria, it had to include information about the cost of informal care for the population with dementia or Alzheimer’s disease in America. Results: Eight published articles were selected to assess the cost of informal care for AD and other dementia population. Most studies used the secondary source of data and different measures to determine the cost of informal care. Informal caregiving cost was estimated to be between $255,120 and $378,690. Conclusion: Informal care constitutes a substantial portion of total care costs. Therefore, it is essential to provide policies that support caregiver programs in ways that would be accessible and applicable to a culturally diverse population and mitigate the burden of caregiving for dementia population.
Keywords: Alzheimer's disease
Cite this paper: Zahra Shahin , Ursula Spence , Samuel Opoku , A Systematic Review of Caregiver Cost of Alzheimer's Disease, Public Health Research, Vol. 9 No. 3, 2019, pp. 56-63. doi: 10.5923/j.phr.20190903.03.
Article Outline
1. Background
- Alzheimer’s disease (AD) is an age-related disease and the most common cause of dementia among elderly population (Mayeux & Stern, 2012). In 2015, AD-affected 47 million people across the globe (WHO Dementia, 2017). Currently, there are more than 5.5 million Americans aged 65 and over living with AD and other types of dementia, and it is predicted that this number will be tripled by 2050 (Alzheimer's Facts, 2017). In the U.S. almost 5 percent of men and women ages 65-74, and about 37.4 percent of those who are 85 years and older developed dementia (Plassman, 2007). Dementia has three stages including mild, moderate, and severe. A person with dementia relies on others for daily activities, and the progression of disease symptoms causes the increased demand for care (Feldman& Woodward, 2005).The social and economic impact of dementia is immense and it continues to grow as the “baby boomers” age. Informal care forms a significant component of the care received by those with dementia. The cost of informal care is substantial because informal care plays a critical role in reducing the cost of formal care that could lead to significant savings in health expenditures (Sari et al., 2013). Consequently, it is crucial to estimate the cost and hours spent by family caregivers for patients with AD and other types of dementia. This systematic review of the literature examines the informal care cost for AD and other dementia patients. In the end, it provides recommendations to minimize the burden of such care on family caregivers.
2. Method
- We followed the revised PRIMA guidelines on systematic review. (Moher, Liberati, Tetzlaff & Atlman, 2009). Systematically, we searched electronic databases such as PubMed, Science Direct, and other sources such as NIH, and Alzheimer’s Association that were published between 2000 and 2017, and selected articles that had information regarding informal care cost for AD population. The key terms were used to find relevant studies included “cost, burden, economic, Alzheimer’s disease, caregivers, informal care.”
3. Study Selection
- The initial search resulted in 16,575 studies. Duplicate articles were removed, and studies screened for the second time resulted in 16,036 studies. We excluded articles that focused solely on the pathophysiology of AD; Alzheimer’s progression; medical care; studies that investigated the effectiveness of intervention program on behavior and psychological aspect of AD; the cost-effectiveness of drug intervention program; quality of life; studies that concentrated on the cost of aging and other chronic diseases among the aging population; economic burden of AD in other countries. A determination of the content was made through a review of the published abstract. We included 56 out of 321 published articles that were identified under approved headings. Full text articles were assessed for eligibility and at the end 8 out of 56 articles were selected for this review. A PRISMA 2009 flow diagram was used to summarize information related to the process of an exclusion and inclusion criteria through different phase of this study.
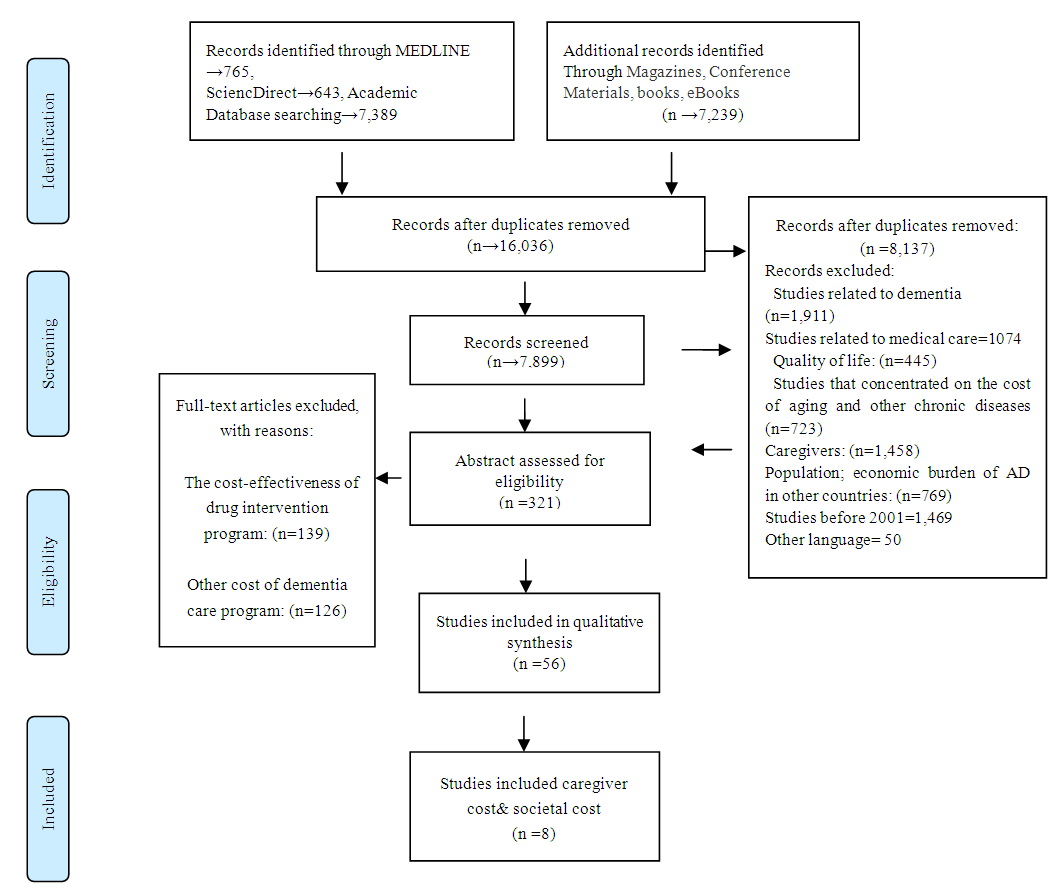 | Figure 1. Process of study selection (PRISMA 2009) |
4. Result
- Eight out of fifty-six articles were selected that contained information about informal care cost. The cost of informal care for dementia patients was apparently higher when compared to their counterparts without dementia and when also compared to other diseases. One study also found that the estimated cost of informal care was even higher for those who were Black, unmarried, and less educated (Kelly et al. 2015).
5. Study Setting
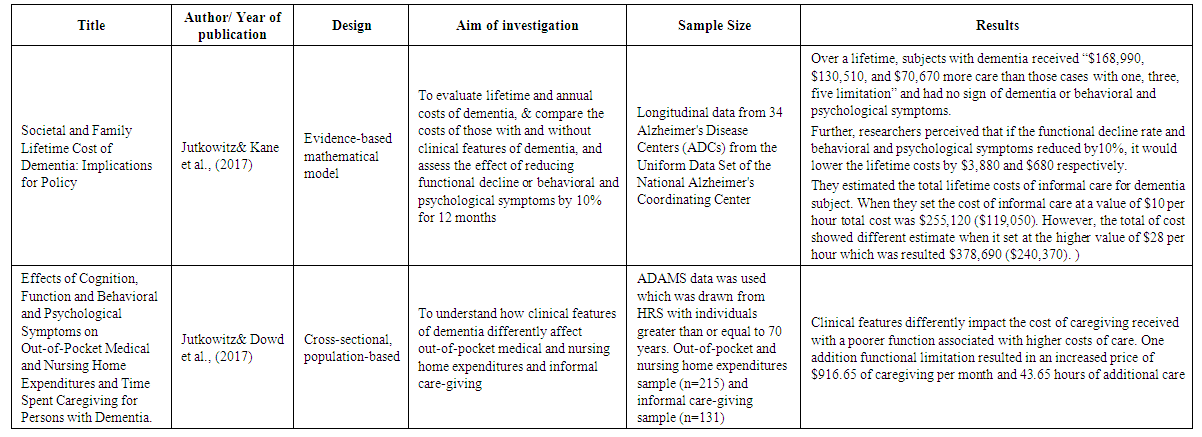
- A few studies estimated the cost of informal caregiving for dementia patients. The focus of these studies was different. Two studies used data from Health and Retirement Study (HSR). The HSR is a longitudinal and a nationally representative survey of individuals aged 5o and older who are also evaluated for cognitive decline (Hurd et al., 2013). Researchers estimated the cost of caregiving using the value of care cost in 2010 dollars. However, both studies calculated the cost of formal and informal care and the measurement of the probability of dementia differently. For instance, Hurd et al. (2013) estimated dementia with and without coexisting conditions, but Kelly et al. (2015) assessed the estimation for both dementia and non-dementia groups after adjusted with coexisting conditions. Lacey et al. (2017) collected data from two clinical trials to examine the pattern of caregiver time among dementia patients who were prescribed bapineuzumab, an antibody acting on the nervous system, and then calculated the cost. One study also estimated the yearly monetary costs and the cost of caregiving for people without dementia and compared that with patients in different stages of dementia. Overall, this document found three publications that were focused on the cost of informal care; most studies had estimated other costs of care in addition to the cost of informal care, or just indicated the number of caregivers and estimated the cost of their time. A summary of 8 studies regarding informal care included in this systematic review provided in table 1.
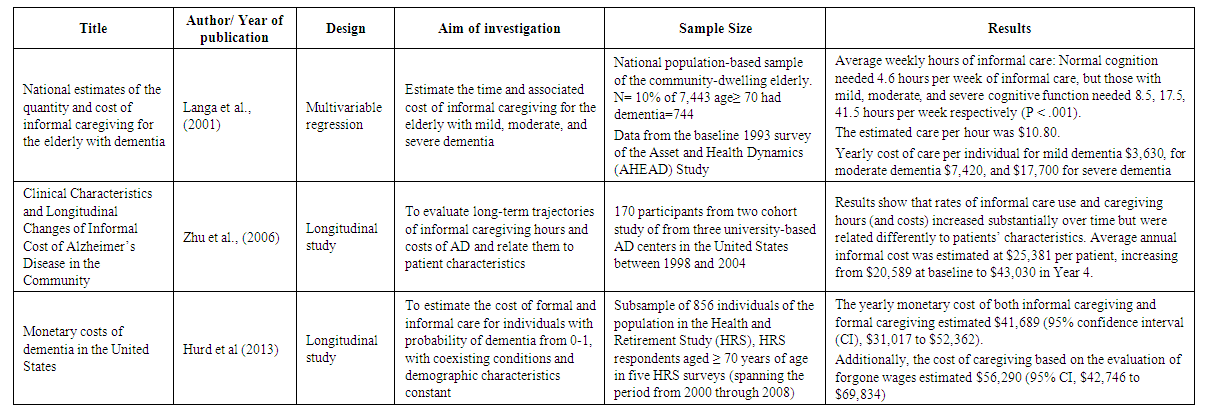 | Table 1. Summarizes of 8 included studies |
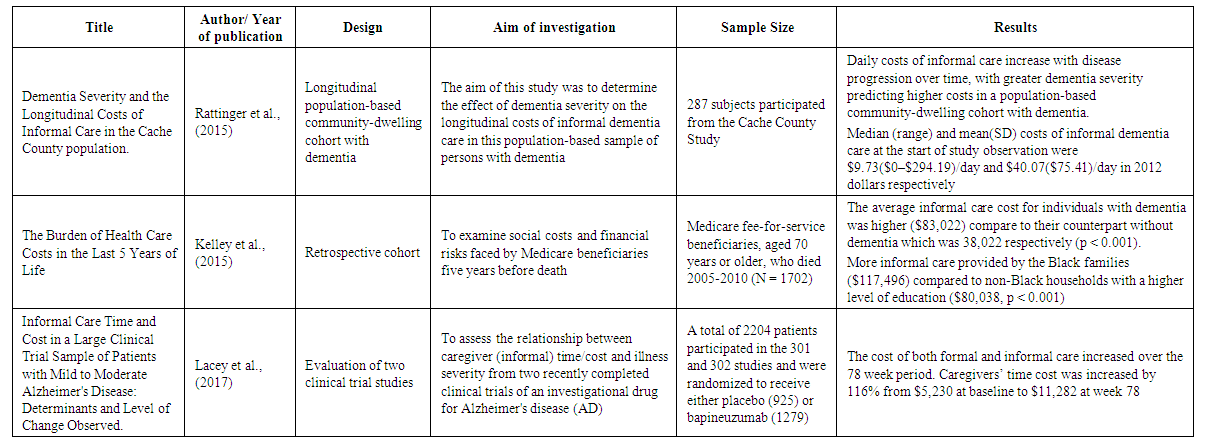 | Table 1. Summarizes of 8 included studies |
 | Table 1. Summarizes of 8 included studies |
6. Cost of Caregiving
- In general, researchers used different surveys, tools, and various themes to study informal care cost for dementia patients. They investigated informal care cost and time spent on the elderly population from different perspectives. For instance, they compared the cost of informal care on individuals with and without dementia (Friedman et al., 2015; Kelley et al., 2015; & Lacey et al., 2017); elderly with different stages of dementia (Langa et al., 2001); dementia patients with neuropsychiatric symptoms (NPS) (Jutkowitz & Dowd et al., 2017); comorbidities, and functional capacity (Zhu et al., 2006); dementia patients “with and without coexisting conditions and demographic characteristics” (Hurd et al., 2013). To demonstrate, their finding concluded that informal care increased by more than double in dementia patients compared to their counterparts with normal cognitive function, 171 hours/month to 66 hours/month respectively (Friedman et al., 2015). Additional hours of informal care were required as the cognitive impairment worsened. Researchers estimated a national annual cost of $18 billion for dementia population (Langa et al. 2001). Zhu et al. (2006) also found that the use and cost of informal care depended on the worsening of patients’ clinical characteristics, and are significantly associated with patients’ functions. For example, the use of informal care was increased for each additional point in the severity of comorbidity scale, and functional capacity scores by 62.2%, and 4.5%, respectively. Each additional point in functional limitation score also increased the use of informal care with an estimated cost of $1,112 a year. The average hours of informal care per week increased over time from 27.9 at baseline to 53.0 in year 4, which led to an increase of the average annual informal care cost from $20,589 at baseline to $43,030 in Year 4 (Zhu et al., 2006). Researchers also estimated the annual cost of care per person “with and without adjustment for coexisting conditions and demographic characteristics” (Hurd et al., 2013). The results demonstrated that the annual cost of informal care in terms of forgone wages was 31% of total cost which was $41,689 (95% CI, $31,017 to $52,362). However, the annual cost was estimated to be 49% of total cost when using the replacement cost which was $56,290 (95% CI, $42,746 to $69,834). The cost estimate showed a reduction of 14% after adjusting for coexisting conditions and demographic characteristics. The cost of informal care was estimated based on the average hourly wage for care in 2010 (Hurd et al., 2013). Rattinger et al. (2015) also noticed that daily informal care cost increased over time by 18% annually. They estimated that the mean cost of daily informal care for very mild dementia was $13.63($37.63), mild dementia $37.84($72.57), moderate dementia $51.78($76.13), and severe dementia $101.23($104.13). The cost of informal care was computed based on Utah's average wage in 2012 dollars. Researchers found that mild dementia patients received more than double the informal care, while moderately affected dementia patients received five times the amount of care. Six times the amount of informal care was reported for severe dementia cases. They also observed that women had a mild impairment with longer dementia duration, yet showed more NPS compared to men (Rattinger et al., 2015). Likewise, Kelley et al. (2015) discovered that the cost of informal care and out-of-pocket was significantly higher for dementia group than non-dementia. After adjusted for coexisting conditions and demographic characteristics, the five years cost of informal care for dementia patients estimated that out-of-pocket and informal care costs were higher for the dementia group ($61,522, and $83,022, respectively) than for the non-dementia group ($34,068, and $38,272, respectively). Further, the average out of pocket spending was higher among non-Blacks ($64,819), than Blacks ($23,425), but Black caregivers provided more informal care ($117,496) than non-black caregivers ($80,038) (Kelley et al., 2015). Another study illustrated that the cost of both formal and informal care increased over the 78 week period. Researchers found that direct medical cost increased by 54%, and that was changed from $2591 at baseline to $3746 at week 78. Caregivers’ time cost was also increased by 116%, and that was changed from $5230 at baseline to $11,282 at week 78 (Lacey et al., 2017).Further, Jutkowitz & kane et al. (2017) observed that life expectancy for dementia patients was 60 months. They estimated the total (net) cost of lifetime informal care to be $255,120 ($119,050) when the value of care set to $10 per hour, and $378,690 ($240,370) when the value of care set to $28 per hour. The total lifetime costs of care for a person with dementia in the best-case analyses was $214,700 ($111,500), and worst-case analysis was $420,850 ($242,500). In addition, the researchers found that the cost of informal care increased as memory function declined and psychological and behavioral symptoms (PBS) progressed. They computed $132,850 excess caregiving received by dementia cases over the lifetime (Jutkowitz & kane et al., 2017). Researchers found that one additional functional limitation and PBS increased the monthly cost by $916.65 and the use of informal care by 43.65 hours. The cost of informal care was estimated at the value of $21 per hour which was based on the value of similar care in the market (Jutkowitz & Dowd et al., 2017). To conclude, based on a recent study was conducted by Jutkowitz & Kane et al. (2017), the estimated annual average informal care cost for dementia population (5.5 million) in the United States would result in an estimated $280 billion and $417 billion. To clarify, when researchers set the informal care cost at a value of $1o per hour, the estimated care cost was $280 billion which was close to the cost estimation of Alzheimer’s Association in 2017, and $417 billion when they set the cost at the value of $28 per hour.
7. Discussion
- Main Findings. This review demonstrated that the cost of care for dementia population continues to rise as the symptoms progress. However, inconsistencies observed among studies regarding estimating the cost of care for this population because there was no standard measure to calculate informal care cost. Therefore, estimations could be influenced by other circumstances, such as local and state programs, recorded data, personal appraisal for estimating the care cost, and the year that study was conducted.Moreover, studies that assessed the cost of informal care for dementia population did not specify the type of dementia (Hurd et al 2013, Kelley et al 2015, Jutkowitz & kane et al., 2017). Additionally, most studies used a secondary source of data to estimate the cost of both informal and formal care; thus, the accuracy of cost estimation is questionable. Some studies also hypothetically assigned subjects to dementia; this was based on the information provided in the survey (Kelly et al., 2015). Accordingly, the estimated time and cost of informal care can be biased because each study reported different hours of caregiving which might impacted by the respondents reporting time. Some of these studies had lack of information regarding informal care, and that was whether such care provided exclusively for dementia or with other coexisting conditions (Langa et al.2001). In addition, one study that collected data from two randomized trials, double-blinded, and placebo-controlled, did not provide information regarding pattern differences of caregivers’ time or costs between patients who took the medication compared to those who received the placebo. Lastly, one study indicated the increasing costs of formal and informal care for the additional functional limitations (Jutkowitz & Dowd et al., 2017), but it did not define whether the rising cost of care was because of progression of the dementia or related to other types of complication among such population (Quentin et al., 2010). Eventually, there was no indication of economic impacts of family caregivers that quit their jobs to provide care for patients with dementia (Schulz & Eden, 2016).Limitation. Several limitations in our study need to be addressed. First, our data were limited, and we excluded other costs of care for the dementia population such as long-term care, healthcare systems, and more which reflected significant aspects of the economic impact of dementia. Also, it was difficult to compare the estimated cost of care for these studies with one another because each study conducted in a different year, different state, and the type of dementia or severity of the disease was not defined (Quentin et al., 2010). Again, most studies used a secondary source of data to estimate the cost. Therefore, the impact of these choices could not be addressed.Recommended Policy. Caring for dementia patients accompanies with financial, physical, and emotional stress for family caregivers. The societal and financial costs associated with caregivers' time for dementia patients should be considered, amongst reducing efficiency at work which sometimes leads to early retirement. Therefore, it is critical that local and state government policies and programs are designed to support informal care and mitigate the burden on family caregivers (Jutkowitz et al., 2017). Support programs should be accessible and applicable to diverse population’s needs. Also, policies should allow employ caregivers to have a flexible workplace to encourage family caregivers to keep dementia patients at home for a longer period of time (Riggs, 2001). If informal care well managed it may reduce the impact of formal care on both society and economy. This is possible by designing and implementing skilled training programs that educate caregivers on how to balance work and family responsibilities. (Schulz & Eden, 2016). Moreover, arranged services such as counseling to help families to cope with the challenges of caregiving (Zhu & Sano, 2006), or to provide public services such as time banks to help dementia patients and their families. Time banks are similar to a bank, but the currency is time, not money. It can be collected for social engagements, and people who are doing volunteer work, so they can earn time credits that can later be exchanged for services and their caring needs in the future (Hayashi, 2012).On the other hand, public health systems should develop strategies to enhance the public knowledge and beliefs about dementia and AD. For instance, implementing educational programs and increasing levels of knowledge about such disease among different groups of the population including healthcare professionals, caregivers, and lay people. To clarify, having higher levels of knowledge about the symptoms and risk factors of dementia and AD among healthcare professionals strengthen their skills to detect the disease at the early stage, and may help them to identify proper treatment and reduce the care costs (Smyth et al., 2013; Anderson et al., 2009). A study by Zhu et al. showed that each additional point in the MMSE score reduced the likelihood of receiving informal care by 9% (2006). Even more, promoting people's knowledge about dementia and AD can encourage them to seek professionals help at an early stage of the disease and possibly delay or prevent the disease (Anderson et al., 2009). Indeed, higher levels of knowledge about AD and dementia enhance caregivers’ skills to manage stress and anxiety that they experience during caregiving for dementia patients and results in an improved quality of life for both patients as well as caregivers (Marim et al., 2013; Werner, 2001). A type of survey research that entails interviewing family caregivers and patients is needed to estimate hours spent by family caregivers, verify the type of dementia, assess the cost of treatment and out of pocket spending, and evaluate the economic impact for the family caregivers who are working, or sometimes quit their jobs to provide care for dementia patients. Additionally, research should focus on evaluating and analyzing the past and present data, and electronic medical records to gain a better understanding of the effectiveness and cost-effectiveness of long-term care programs, and determine high priorities for future planning and related policies (Neumann, 2000).
8. Conclusions
- Overall, the cost of care for people with AD and other dementias is a significant burden on the U.S. family caregivers and society. Though, most studies indicated that the majority of the total cost of care for dementia patients attributed to informal care, the estimated time and cost of informal care provided by this review might not be accurate due to the nature of each study and how different data was reported for the cost of care. Further research is needed to investigate how the cost of informal care can be standardized.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML