Potapov V. V. 1, Kashutina I. A. 2, Shunina E. V. 1
1Geotechnological Research Center, Far East Branch of Russian Academy of Science, Petropavlovsk-Kamchatsky, Russia, 683002, Severo-Vostochnoe shosse, 30
2Kamchatka State University, Petropavlovsk-Kamchatsky, Russia, 683032, Pogranichnaya street, 4
Correspondence to: Potapov V. V. , Geotechnological Research Center, Far East Branch of Russian Academy of Science, Petropavlovsk-Kamchatsky, Russia, 683002, Severo-Vostochnoe shosse, 30.
| Email: |  |
Copyright © 2014 Scientific & Academic Publishing. All Rights Reserved.
Abstract
Nucleation of orthosilicic acid with formation of colloidal silica particles in hydrothermal solutions was studied by the method of numerical simulation. Numerical simulation was done at different parameters: chemical composition, temperatures, pH, ionic strengths of hydrothermal solutions. The rates of nuclei formation and its growth, time dependence of orthosilicic acid concentration, final sizes of colloidal silica particles were calculated. Results of calculations were compared with experimental data. Experiments were carried out with solutions of Mutnovskoye and Pauzhetskoye geothermal fields. The approach based on numerical simulation can be used for developing of technology for silica extraction from hydrothermal solutions.
Keywords:
Hydrothermal solution, Orthosilicic acid, Nucleation, Sizes of colloidal particles
Cite this paper: Potapov V. V. , Kashutina I. A. , Shunina E. V. , Numerical Modeling of Orthosilicic Acid Nucleation in Hydrothermal Solutions, Physical Chemistry, Vol. 4 No. 1, 2014, pp. 1-10. doi: 10.5923/j.pc.20140401.01.
1. Introduction
There is a problem to study physical and chemical characteristics of colloidal silica in hydrothermal heat carrier in the context of silica extraction technology and increasing of effectiveness of heat carrier using (Potapov V.V., Karpov G.A., Podverbny V.M., 2002; Potapov V.V., 2003).Colloidal silica forms in the hydrothermal solution during several stages (Potapov V.V., 2003). Silica comes into the solution as molecules of silicic acid as a result of water chemical interaction with silica-alumina rocks of hydrothermal fields at the depth 1.0-3.5 km in thermal anomaly zones under a high temperature (up to 250-350℃) and pressure (4.0-20 МPa). Hydrothermal solution is multicomponent. Compounds with Na, K, Si, Ca, Mg, Al, Fe, Cl, S, C, B, Li, As, Cu, Zn, Ag, Au and others are present at the solution as ions and molecules.Under the temperature 250-350℃ when solution contacts with rocks minerals a total silica content Ct (mole/kg) in water can be estimated by α-quartz solubility (Crerar D.A., 1971):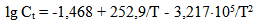 | (1) |
Equation (1) gives the following values of quartz solubility SiO2 (mg/kg): 25℃ – 3.46, 50℃ – 10.29, 100℃ – 47.6, 200℃ – 256.0, 250℃ – 415.6, 300℃ – 592.5. Over rising filtration in fractured-and-porous rocks or during solution movement through the producing wells of geothermal electric power plants (GeoPP) pressure and temperature of the solution reduce and solution is divided in steam and liquid phases. Total silica content Ct in a liquid phase can reach 700-1500 mg/kg. Consequently water solution becomes supersaturated relative to a solubility of amorphous silica Ce (Marshall W.L., 1980). In accordance with experimental data (Marshall W.L., 1980) Ce value (mole/kg) for pure water depends on an absolute temperature T in the following way: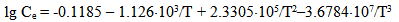 | (2) |
Under the temperature 200℃ solubility Ce is 940.8 mg/kg, under the temperature 150℃ – 651.8 mg/kg, 100℃ – 405.3 mg/kg, 25℃– 130.8 mg/kg.Such condition of monomeric silicic acid in water solution is unstable. Solution supersaturation Sm which is equal to the difference (Cs-Ce) of orthosilicic acid concentration Cs and solubility Ce causes nucleation and polymerization of silicic acid molecules with condensation of silanol groups and formation of siloxane links and partial dehydration in accordance with the following reactions (Iler R., 1982):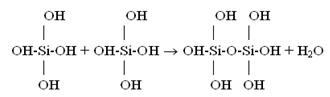 | (3) |
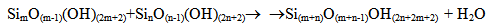 | (4) |
2. Chemical Composition of Hydrothermal Solution
It is necessary to study the process of orthosilicic acid nucleation to work out the technology of extraction of silica-containing materials with given characteristics from the hydrothermal heat carrier. Final concentration and the size of colloidal particles before their extraction from aqueous solution depend on the parameters of the process nucleation of orthosilicic acid. Numerical simulation in different conditions corresponding to the heat carrier characteristics of Verkhne-Mutnovsky GeoPP was made to study the process of nucleation. Table 1 presents physical and chemical characteristics of liquid phase of hydrothermal heat carrier (separate) of GeoPP. This phase is separated from two-phase water-steam flow in separators of GeoPP. Aqueous solution of separate refers to natrium-potassium-chloride-sulphate type of thermal waters. Total silica content in solution varies in the range of 500-1000 mg/kg, pH=8.0-9.4, ionic strength Is = 0.01-0.02 mole/kg.| Table 1. Ions and cations concentrations in hydrothermal separate from the pipe-line of reverse pumping of Verkhne-Mutnovskaya GeoPP, ionic strength Is = 14.218 mmole/kg, conductivity σ = 1250 microS/cm, n/d means that concentration was not determined, (-) means that values were not estimated |
| | Component | mg/l | mg•equ/l | | mg•equ/l | %mg•equ/l | | Na+ | 239.4 | 10.413 | 88.044 | | K+ | 42.0 | 1.074 | 9.080 | | Ca2+ | 1.6 | 0.0798 | 0.6747 | | Mg2+ | 0.72 | 0.0592 | 0.5005 | | Fe2, 3+ | <0.1 | <0.0053 | 0.0448 | | Al3+ | 0.27 | 0.033 | 0.2790 | | NH4+ | 1.1 | 0.0609 | 0.5149 | | Li+ | 0.71 | 0.102 | 0.8624 | | Cations sum | 285.9 | 11.827 | 100.0 | | Cl- | 198.5 | 5.591 | 47.664 | | HCO3- | 81.0 | 1.327 | 11.312 | | CO32- | 19.9 | 0.663 | 5.652 | | SO42- | 192.1 | 3.9995 | 34.096 | | HS- | 4.95 | 0.15 | | | H2S0 | 5.92 | - | - | | F- | n/d | n/d | - | | Anions sum | 496.5 | 11.73 | 100.0 | | H3BO3 | 106.9 | - | | (H4SiO4)t | 1190 | - | | (H4SiO4)s | 222 | - | | Mineralization Mh, mg/kg | 1638.9 | | pH | 9.35 |
|
|
3. Mathematical Model
Mathematical model (Weres 0., Yee A., Tsao L., 1981) worked out by Weres, Yee and Tsao in Lawrence Berkeley Laboratory (USA) was used for numerical simulation. Supersaturation SN(T) which is equal to Cs/Ce and pH are main factors defining nucleation rate IN of silicic acid in aqueous solution: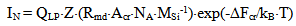 | (5) |
where ΔFcr is a free energy change connected with nuclei formation of critical radius Rcr, Acr is the surface area of critical nuclei, Acr=4•π•Rcr2, σsw is the surface tension on silica-water boundary, Rmd is the rate of molecular precipitation of SiO2 onto a solid surface, g•(cm2•min)-1, kB is Boltzmann constant, MSi is the molar mass SiO2, NA is Avogadro constant, QLP is Lothe-Paund factor, QLP =3,34•1025 kg-1, Z is Zeldovich factor, ncr is the quantity of molecules of SiO2 in nuclei of critical size.Critical radius, quantity of molecules in nuclei of critical radius, free energy and Zeldovich factor were estimated in accordance with the following formulas (Weres 0., Yee A., Tsao L., 1981): 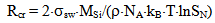 | (6) |
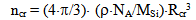 | (7) |
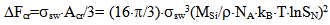 | (8) |
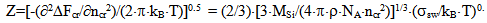 | (9) |
ρ is the density of amorphous silica.The dependence of the function Rmd of the rate of SiO2 molecules deposition on the temperature and pH solution in a given model is shown in the following equations (Weres 0., Yee A., Tsao L., 1981):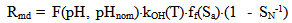 | (10) |
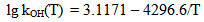 | (11) |
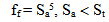 | (12.1) |
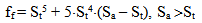 | (12.2) |
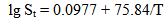 | (12.3) |
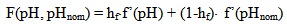 | (13.1) |
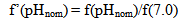 | (13.2) |
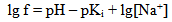 | (13.3) |
The dependence of surface tension on the temperature and pH coefficient σSW is given by the function I(pH, pHnom):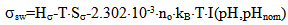 | (14.1) |
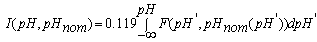 | (14.2) |
where Sa = (1-αi)•SN, αi is a fraction of silicic acid in ionic form, pHnom = pH + lg([Na+]/0.069), [Na+] is an ion activity [Na+], mole/kg, pKi=6.4, f(7.0) = 0.119, hf=0.45, Hσ, Sσ are specific enthalpy and entropy of silica surface in water, Hσ = 63.68•10-3 Joule/m2, Sσ = 0.049•10-3 Joule/m2•К, no = 6.84 nm-2.Equation (5) shows maximum rate IN of nucleation for the particles with the radius that is higher critical. Its molecules quantity of SiO2 is equal to n = ncr+0.5/Z. IN(t) value reaches maximum in induction time τin that is necessary to grow and form stable population of the particles which sizes are close to critical. Time dependence IN(t) has got the form (Weres 0., Yee A., Tsao L., 1981):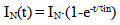 | (15) |
To calculate time 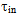 the following formula is used:
the following formula is used: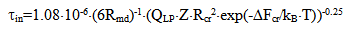 | (16) |
4. Program for Numerical Simulation
The system of equations (1), (2), (5)-(16) was used to develop MSANUC.FOR program. The solution algorithm in accordance with MSANUC.FOR was the following:1) Initial values entry: temperature T, pH, concentration of ions and ionic strength of solution, the assignment of total silica Ct content of initial time “TIME” and time step DT, the assignment of initial radii R(I) and particles quantity MP(I) of every class “I” which are present in the solution before nucleation.2) The calculation of colloidal silica content in solution by summation of particles of all classes: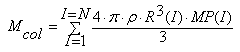 | (17) |
3) The calculation of concentration of monomeric orthosilicic acid as difference between total content and colloidal silica content: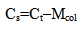 | (18) |
4) The calculation of current value of supersaturation solution SN: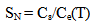 | (19) |
5) The calculation of values of σsw, Rmd, Rcr, Z, 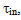 IN(t) corresponding to current values SN, pH and T by equations (5)-(16).6) The calculation of the quantity of new particles appearing during the time DT in accordance with current value of nucleation rate IN on a given program step and educing a new class of particles N+1:
IN(t) corresponding to current values SN, pH and T by equations (5)-(16).6) The calculation of the quantity of new particles appearing during the time DT in accordance with current value of nucleation rate IN on a given program step and educing a new class of particles N+1: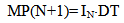 | (20) |
7) The calculation of summary concentration of particles: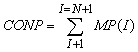 | (21) |
8) The calculation of particle mass difference DPM of every class during the time DT in accordance with the equation: DPM(I)=4•π•R2(I) •Rmd•DT9) The calculation of particle radius R(I) corresponding to a new value of particle mass in every class: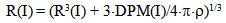 | (22) |
And the calculation of mean values of Rср, R2ср, R3ср in all classes of particles: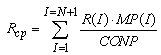 | (23) |
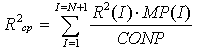 | (24) |
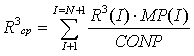 | (25) |
10) The calculation of new temperature values, pH, ionic strength of solution if these values change with time. After the tenth step we must return to the second step of the program to make the next cycle. Registration of calculations results in a separate file is provided for: mean radius, summary concentration of particles and concentration of monosilicic acid for given time.
5. Results
Table 2 a,b presents the results of numerical simulation of nucleation with MSANUC.FOR program under different temperatures and pH solution. During the initial time colloidal silica content supposed to be zero and all silica was in a monomeric orthosilicic acid form:Cs(t=0) = Ct| Table 2. Final mean sizes of silica particles under a constant temperature and different рН; CS=700 mg/kg; (-) means that it was not estimated |
| | рН | t, ℃ | | 20 | 40 | 50 | 60 | 80 | 100 | | 4 | 5.289 | 19.247 | 48.067 | 156.551 | 6342.132 | - | | 5 | 5.290 | 19.054 | 47.305 | 153.158 | 5858.727 | - | | 6 | 2.983 | 16.823 | 40.691 | 128.018 | 4684.754 | - | | 7 | 2.978 | 7.667 | 15.282 | 38.175 | 701.463 | - | | 8 | 0.988 | 1.219 | 1.508 | 1.775 | 3.488 | 18.77 | | 8,5 | 0.645 | 0.731 | 0.784 | 0.841 | 1.013 | 1.268 |
|
|
Simultaneous results show that critical radius Rcr increases due to supersaturation reduce when temperature becomes higher. Consequently the tendency for decrease of nucleation rate IN and increase of induction time τin takes place; it leads to reduce of particles concentration Np and increase of final mean radius of the particles Rf. Under the temperature 120-150℃ and higher nucleation hasn’t time to develop during a real residence time of hydrothermal solution in wells and thermal equipment of GeoPP.After nucleation and polymerization completion a part of silica continues to be as molecules of orthosilicic acid H4SiO4 which concentration is close to solubility of Ce. It is in equilibrium with colloidal silica. Small amount of silicic acids ions (H3SiO4-, H2SiO42-, HSiO3-, etc.) and macromolecules of polysilicic acids are in the solution parallel with colloidal particles and molecules of silicic acids. Data processing obtained by Rothbaum and Rohde (Rothbaum H.P., Rohde A.G., 1979) has showed the concentrations of dimmers Cdimer and trimers Ctrimer of silicic acid are approximated by the equations (mole/kg):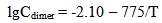 | (26.1) |
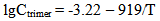 | (26.2) |
The dependence of constants of orthosilicic acid ionization of the first K1 = [H+]•[H3SiO4-]/[H4SiO4] and second K2 = [H+]•[H2SiO42-]/[H3SiO4-] stages on the temperature has the following form: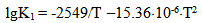 | (27.1) |
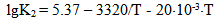 | (27.2) |
In accordance with equations (26-1, 26-2) under the temperature 20-180℃ and pH=7.0-9.2 a fraction (portion) of dimmers with respect to orthosilicic acid which concentration is close to a solubility of Ce(T) doesn’t exceed 1.0%, a fraction(portion) of trimers is 0.1%, a fraction(portion) of tetramers and low-molecular circular polymers (up to 6 units SiO2) (Rothbaum H.P., Rohde A.G., 1979) is < 0.1%. A fraction (portion) of ions H3SiO4- and H2SiO42- under such conditions is not more than 14.0%.The calculations by the method of photon correlation spectroscopy show that particles mean radius of the polymerized silica in hydrothermal solution has values from 7.0 up to 30.0 nm, values of particles radii vary from 1.0 up to 50.0 nm (Potapov V.V., 2003).With the help of MSANUC.FOR final mean size of silica particles under different constant temperatures 20℃, 40℃, 50℃, 60℃, 80℃, 100℃ and different рН=4-8.5 were estimated. Table 2 presents estimated final mean sizes of the particles (nm).When pH=5-6 and the temperature is 50℃ the calculations were made with step by рН 0.1. It made the possibility to study the influence of pH solution on the size of silica particles more detailed. The estimated results are shown in Table 3 and on Figure 1.| Table 3. Final mean sizes of silica particles under the temperature 50℃ and different рН; CS=700 mg/kg |
| | рН | RA, nm | рН | RA, nm | | 5 | 47.305 | 5.5 | 45.423 | | 5.1 | 46.995 | 5.6 | 44.854 | | 5.2 | 46.771 | 5.7 | 44.007 | | 5.3 | 46.42 | 5.8 | 43.077 | | 5.4 | 45.954 | 5.9 | 42.028 |
|
|
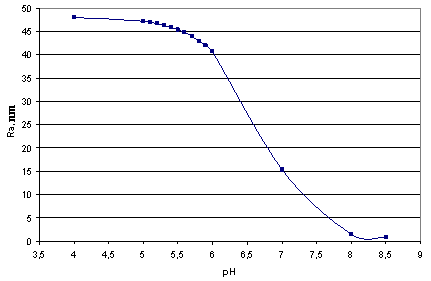 | Figure 1. The dependence of final mean size of silica particles on рН. t=50℃, CS = 700 mg/kg |
On Figure 2 the dependence of concentration of dissolved silicic acid CS (mg/kg) on time Tp (min) is shown for t=50℃, initial concentration CS=700 mg/kg and different values рН; Figure 3 presents the dependence of supersaturation SN on time Tp (min); Figure 4 (а-f) presents the dependence of distribution of final sizes of silica particles.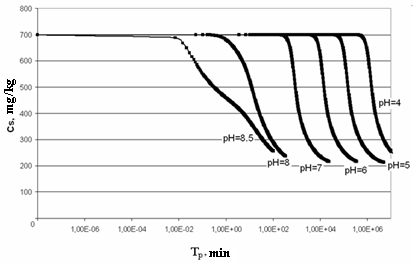 | Figure 2. The dependence of concentration of dissolved silicic acid on time. t=50℃, initial concentration CS=700 mg/kg |
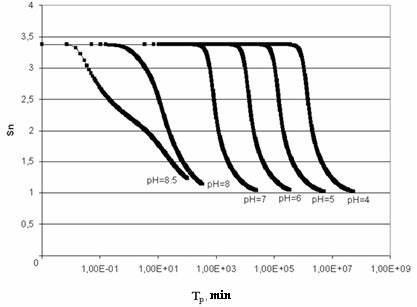 | Figure 3. The dependence of supersaturation SN on time. t=50℃, initial concentration CS=700 mg/kg |
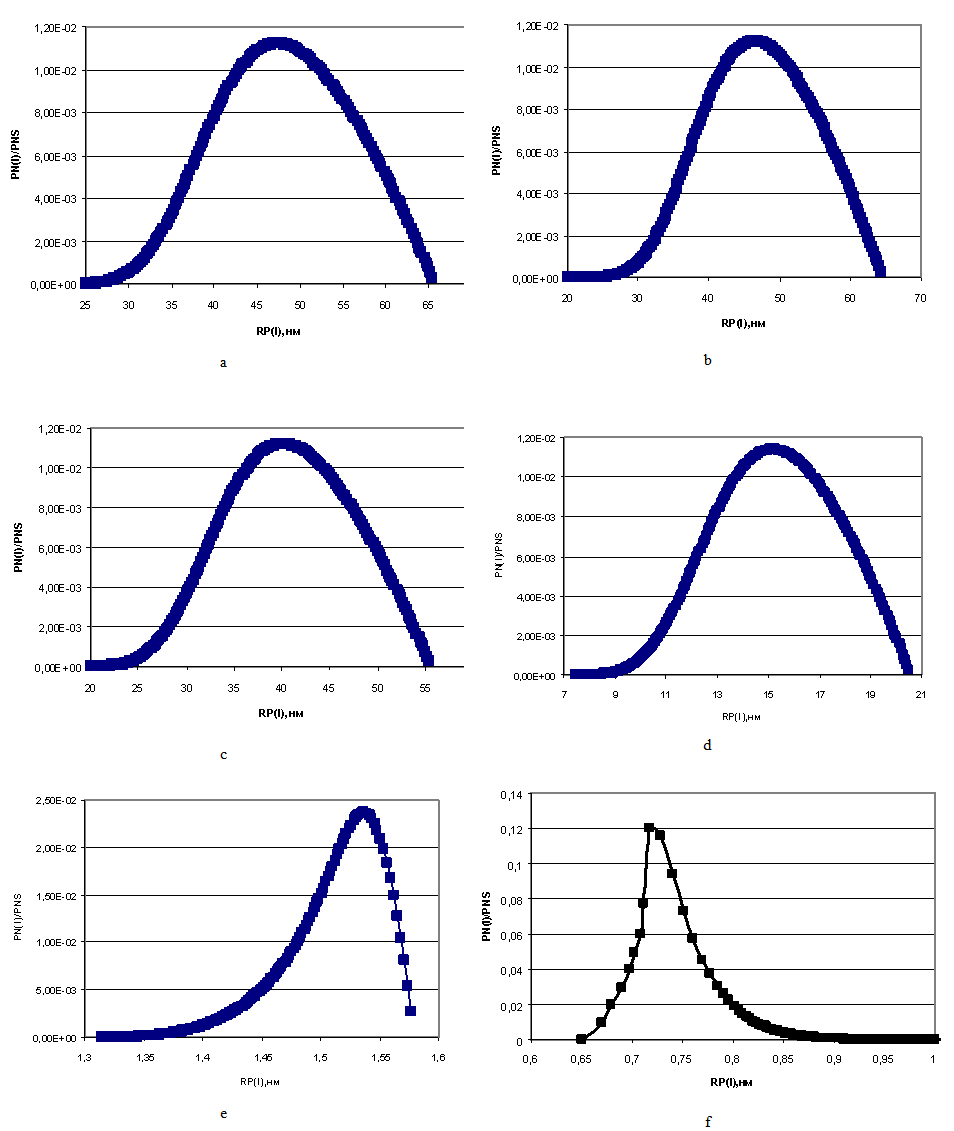 | Figure 4. The distribution of particles sizes under the temperature t=50℃, initial concentration CS=700 mg/kg. a – рН=4; b – рН=5; c – рН=6; d – рН=7; e – рН=8; f – рН=8.5 |
The influence of initial solution concentration CS (mg/kg) on a final mean size of silica particles (nm) is shown in Table 4 and Figure 5. Calculations were made under different solution temperatures: 20℃, 40℃, 60℃, 80℃, 90℃; different initial solution concentrations CS (mg/kg): 500, 600, 700, 800, 1000.| Table 4. Values of final mean size of silica particles (nm) under different temperatures t and initial concentrations of solution CS, рН=8; (-) - means that it was not estimated |
| | CS,mg/kg | t, ℃ | | 20 | 40 | 50 | 60 | | 300 | 93.57 | - | - | - | | 350 | 12.46 | 1019.9 | - | - | | 400 | 4.35 | 49.11 | 586.3 | - | | 500 | 1.79 | 4.088 | 8.968 | 32.71 | | 600 | 1.246 | 1.846 | - | 3.808 | | 700 | 0.988 | 1.219 | - | 1.775 | | 800 | 0.876 | 1.054 | - | 1.311 | | 1000 | 0.739 | 0.836 | - | 0.952 | | CS,mg/kg | t, ℃ | | 70 | 80 | 85 | 90 | | 300 | - | - | - | - | | 350 | - | - | - | - | | 400 | - | - | - | - | | 500 | - | - | - | - | | 600 | 7.56 | 24.14 | 56.14 | 169.8 | | 700 | - | 3.488 | - | 6.496 | | 800 | - | 1.815 | - | 2.308 | | 1000 | - | 1.108 | - | 1.212 |
|
|
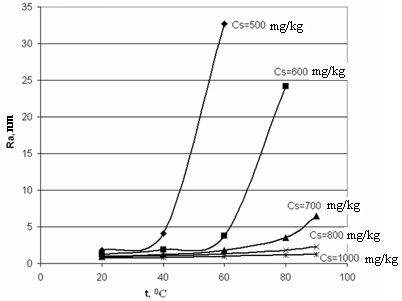 | Figure 5. The dependence of a final mean size of silica particles on different temperatures t and different initial concentrations of the solution CS; рН=8 |
Figure 2 shows that curves reflecting the dependence of concentration of dissolved silicic acid on time have two areas: convex curve reflects homogeneous nucleation, that is the stage of formation of new particles centres, concave part reflects heterogeneous nucleation, i.e. the growth of centres formed earlier.Figure 6 (a–c) presents the dependence of quantity of the formed particles PNS (nucl) on time TP (min) and dependence of nucleation rate Rnuc (nucl/kg*s) on time ТР (min) under the temperature t=50℃, initial concentration CS=700 mg/kg and different рН. 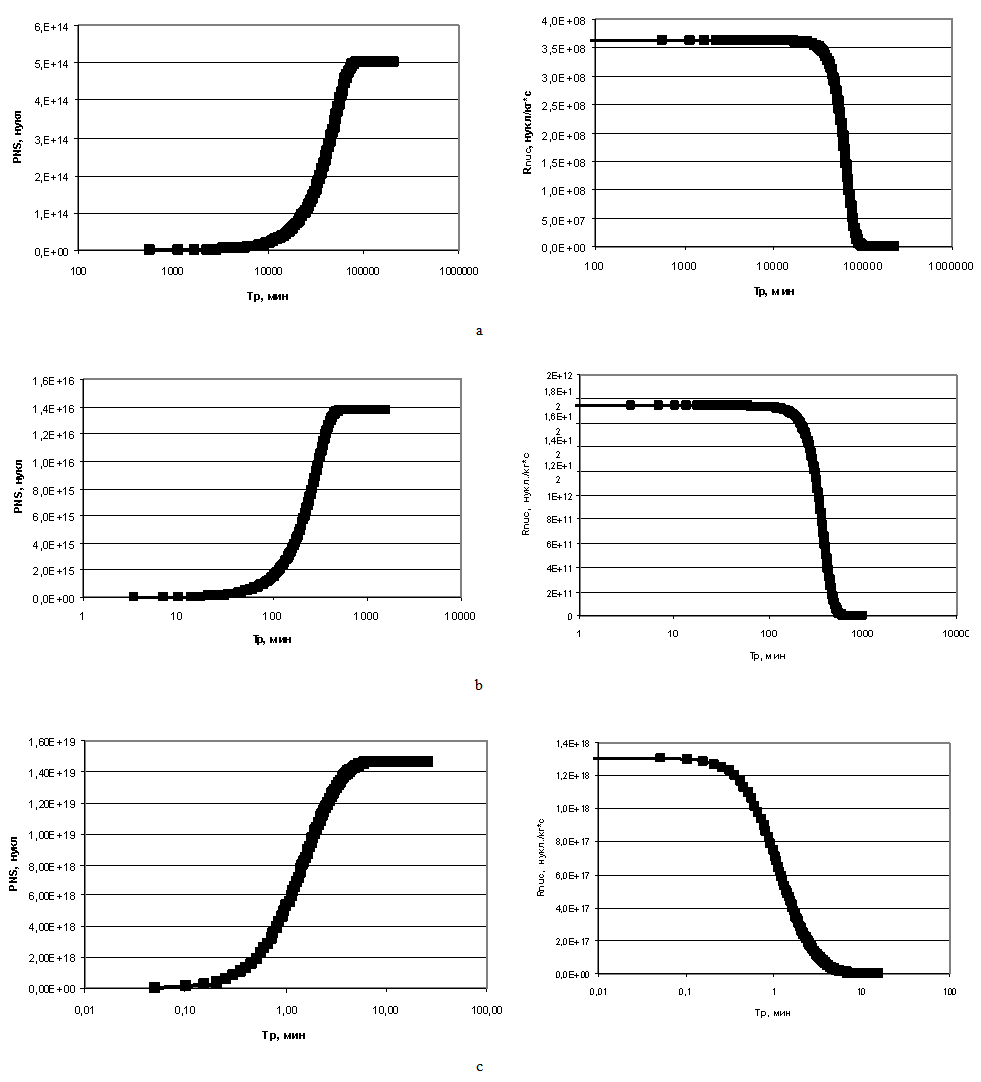 | Figurer 6. Dependences of formed particles quantity PNS on time ТР and dependences nucleation rate Rnuc on time ТР; t=50℃, initial concentration CS=700 mg/kg. а – рН=5; b – рН=7; c – рН=8 |
On Figure 4 left curve part of particles sizes distribution is concave and right curve part is convex.Figure 7 presents the dependence of concentration of dissolved silicic acid CS (mg/kg) on time TP (min) for t=60℃, рН=8 and different initial; Figure 8 shows the dependence of supersaturation SN on time TP (min); Figure 9 (a-e) presents the dependence of distribution of final sizes of silica particles.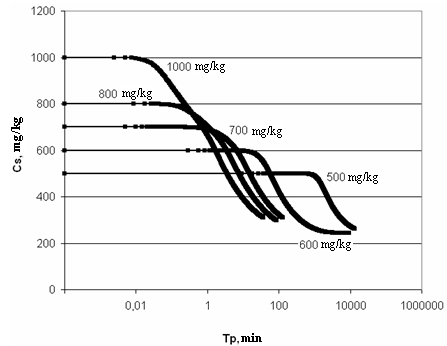 | Figure 7. The dependence of dissolved silicic acid concentration on time. t=60℃, рН=8 |
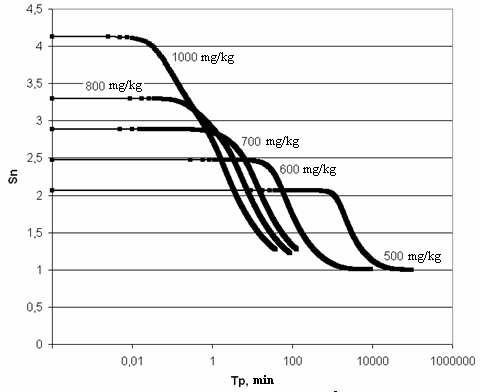 | Figure 8. The dependence of supersaturation SN on time. t=60℃, рН=8 |
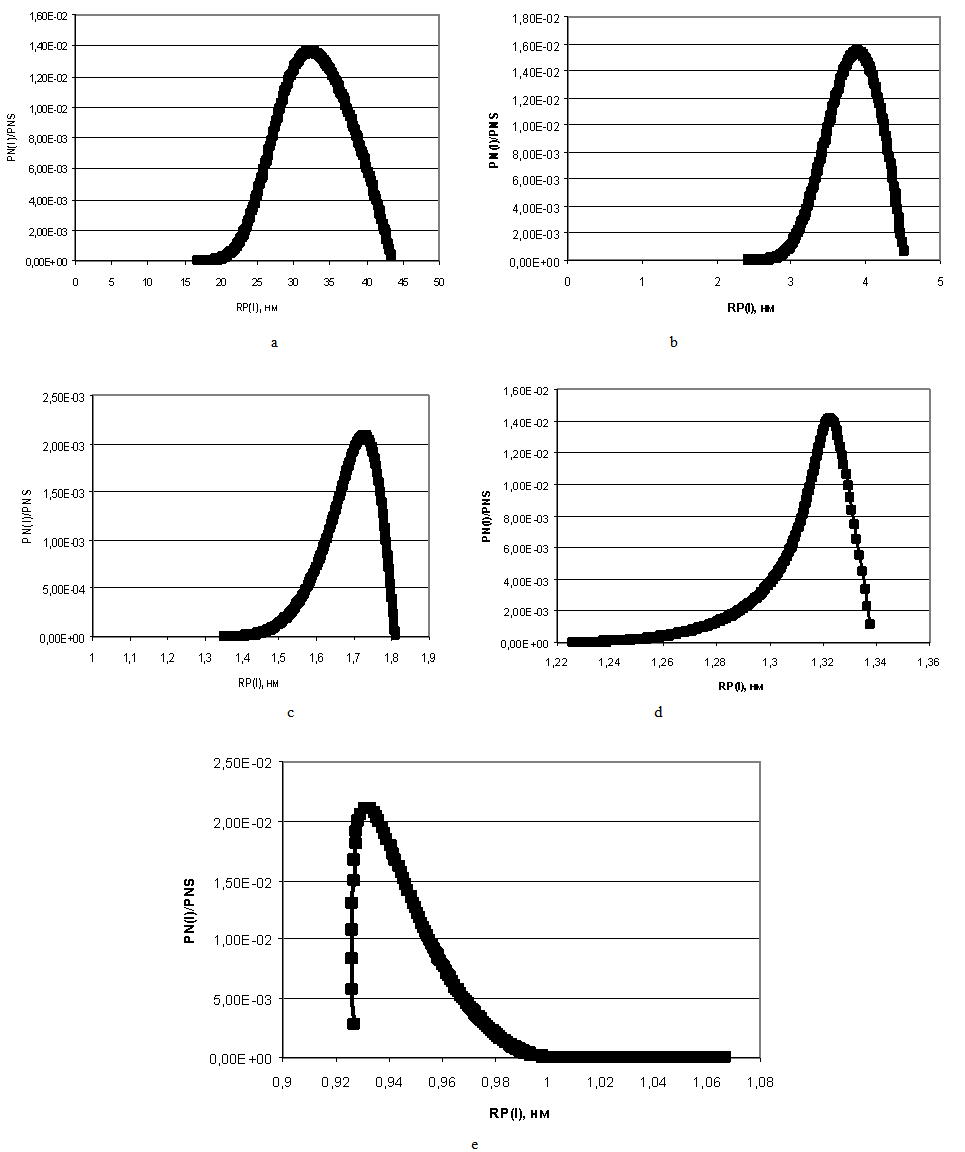 | Figure 9. The distribution of particles sizes at the temperature t=60℃, рН=8. а – CS=500 mg/kg; b – CS=600 mg/kg; c – CS=700 mg/kg; d – CS=800 mg/kg; e – CS=1000 mg/kg |
Final mean sizes of silica particles (nm) for initial concentration CS=300, 350 mg/kg, рН=8 and ionic strength corresponding to the solution characteristics of Pauzhetsky geothermal field are in Table 5.| Table 5. Values of final mean size of silica particles (nm) under different temperatures t and different initial solution concentration CS. (-) - means that it was not estimated |
| | CS, mg/kg | t, ℃ | | 20 | 30 | 40 | 50 | | 300 | 13.99 | 129.9 | 9095.69 | - | | 350 | 3.59 | 10.26 | 63.95 | 2103.6 |
|
|
The results of simulation show that critical radius (nm) Rcr and induction time τin (min) increase when рН is stated and temperature becomes higher due to supersaturation reduce SN. Consequently nucleation rate IN reduces; it causes to increasing of final mean particles size RA (nm).At the same temperature when рН reduces the surface tension σsw increases and rate of molecular deposition of silicic acid reduces. Owing to it critical radius of particles and induction time increase, nucleation rate reduces and final mean size of particles increases.
6. Conclusions
1. The results of numerical simulation show that in hydrothermal solutions the transformation of orthosilicic acid into colloidal silica happens as a result of two sequential processes: 1) homogeneous nucleation during which the transformation of orthosilicic acid into colloidal silica happens mainly as a result of increasing of new centers quantity; 2) heterogeneous nucleation which happens due to increasing of sizes of centers formed earlier if their quantity is practically constant. Durations of these stages depend on process parameters: temperature, pH, ionic strength, total silica content and supersaturation ratio.2. The results of numerical simulation show that the nucleation of orthosilicic acid molecules and formation of colloidal particles in hydrothermal solution actively develop under the temperature below 120-150℃. Under the temperature 120-150℃ and higher the induction time becomes so long that colloidal partiles are not formed time of geothermal water treatment in heat equipment of GeoPP.3. pH solution is an another factor considerably influencing the nucleation kinetics. When pH is low critical radius of particles increases considerably, the nucleation rate reduces and induction time increases. When pH is 7.0 and lower the induction time considerably exceeds the time of of geothermal water treatment in heat equipment of GeoPP and colloidal particles are not formed. 4. The method based on of numerical simulation carried out in this paper can be applied to project equipment for flow sheet of silica extraction from the hydrothermal solution during the stage of ageing. Such method gives a possibility to take into account how temperature profile, pH, ionic strength, concentration of solution components, duration of ageing during solution movement through the wells, pipe lines and equipment influence on final concentration and size of colloidal particles.
References
| [1] | Crerar D.A., Anderson G.M. Solubility and solvation reactions of quartz in dilute hydrothermal solutions // Chem. Geol. 1971. V. 8. P. 107-122. |
| [2] | Deryagin B.V., Churaev N.V., Muller V.M. Surface forces. М.: Nauka, 1985. P. 398. |
| [3] | Deryagin B.V. The theory of stability of colloids and thin films. М.: Nauka, 1986. P. 206. |
| [4] | Fleming B.A. Kinetics of reaction between silicic acid and amorphous silica surfaces in NaCl solutions. J. Coll. Interf. Sci. 1986. V. 110. No. 1. P. 40-64. |
| [5] | Iler R. Silica chemistry. М: Mir, 1982. parts1, 2. P. 1127. |
| [6] | Marshall W.L. Amorphous silica solubilities. I. Behavior in aqueous sodium nitrate solutions: 25 – 300℃, 0-6 molal // Geochimica et Cosmochimica Acta. 1980. V. 44. P. 907-913. |
| [7] | Potapov V.V. Colloidal silica in hydrothermal solution. Vladivostok: Dalnauka, 2003. P. 216. |
| [8] | Potapov V.V., Karpov G.A., Podverbny V.M. Silica extraction fom hydrothermal heat carrier // Theory of chemical technology. – 2002. – Т. 36, № 6. - Pp. 644-651. |
| [9] | Rothbaum H.P., Rohde A.G. Kinetics of silica polymerization and deposition from dilute solutions between 5 and 180℃ // J. Coll. Interf. Sci. 1979. V. 71. No. 3. P. 533-559. |
| [10] | Shabanova N.A., Sarkisov P.D. Sol-gel manufacturing science of nanodispersed silica. М., 2004, P. 208. |
| [11] | Weres 0., Yee A., Tsao L. Kinetics of silica polymerization // J. Coll. Interf. Sci. 1981. V. 84. No. 2. P. 379-402. |

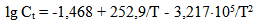
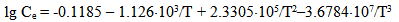
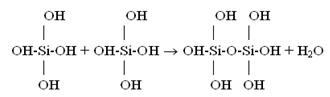
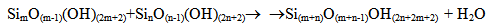
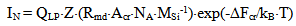
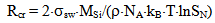
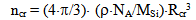
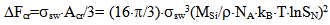
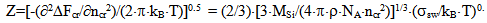
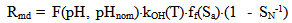
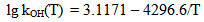
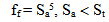
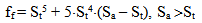
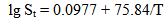
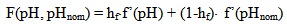
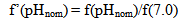
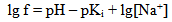
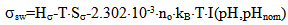
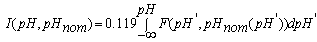
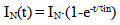
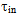 the following formula is used:
the following formula is used: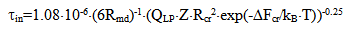
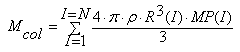
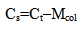
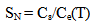
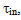 IN(t) corresponding to current values SN, pH and T by equations (5)-(16).6) The calculation of the quantity of new particles appearing during the time DT in accordance with current value of nucleation rate IN on a given program step and educing a new class of particles N+1:
IN(t) corresponding to current values SN, pH and T by equations (5)-(16).6) The calculation of the quantity of new particles appearing during the time DT in accordance with current value of nucleation rate IN on a given program step and educing a new class of particles N+1: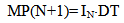
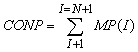
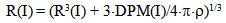
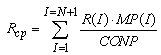
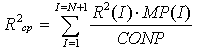
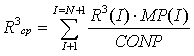
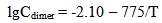
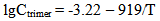
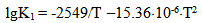
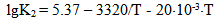









 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML