-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Physical Chemistry
2012; 2(4): 43-47
doi: 10.5923/j.pc.20120204.01
C = C’ – r, A Silver Bullet for Evaluation of Components in Simple Equilibrium Reactions: The Relation Revisited
R. Sanjeev 1, V. Jagannadham 2, R. Veda Vrath 3
1Department of Chemistry, Mizan-Teppi University, Teppi Campus, Teppi, Ethiopia
2Department of Chemistry, Osmania University, Hyderabad-500007, India
3Department of Chemistry, L N Gupta Evening College, Hyderabad-500002, India
Correspondence to: R. Sanjeev , Department of Chemistry, Mizan-Teppi University, Teppi Campus, Teppi, Ethiopia.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
Phase rule is an integral part of the undergraduate curriculum in many universities across the globe. In an appreciable number of universities, it is dealt with in the freshman year. The students find it extremely confusing to find the number of components even in simple equilibrium reactions. Normally they end up giving wrong answers, or if at all they give correct answers, they are just conjectures. To overcome this overwhelming confusion we have first considered different simple and relatively uncomplicated equilibriums from standard physical chemistry books and research journals. In these different equilibriums first we evaluated the number of components as illustrated in the books. In each of the book, dissimilar methods were employed to evaluate the number of components which is not a straightforward task for the freshmen students. Then we presented an easy generic method which involved the use of the less involving relation C = C’ – r. Our endeavour in this article is to present a terse and lucid method, by means of which the number of components can be evaluated. Finally, one feels, after going through this article, that this convenient and handy relation C = C’ – r is undeniably a silver bullet for the evaluation of components in simple and relatively less complicated equilibriums.
Keywords: Phase, Components, Restrictions, Constituents, Phase Rule
1. Introduction
- It has been found that one of the most difficult areas in physical chemistry at freshmen level is the phase rule, which requires large amount of imagination on the part of the students when it comes to evaluation of the number of components, one of the vital terms in phase rule. Even though there are easy formulae to overcome this requirement, the students are not made aware of. One such formula is C = C’ – r for the evaluation of number of components C. Here C’ is the total number of number of chemical constituents in the system, and r is the number of restrictions imposed on the independent variation of these constituents. In this article, we have shown how the use of the relation C = C’ – r is far more superior to the other methods when it comes to the ease of evaluation of number of components.
2. Objective
- As described in the title the very purpose of writing this article is to revisit the relation C = C’ – r. This generic andhandy relation enables easy evaluation of components in various equilibriums, especially for freshmen students. Even in the case where large amount of imagination is required to evaluate the components, the relation C = C’ – r comes to aid.
3. Method
- Since this article involves the use of the handy relation C = C’ - r, the method is less involving. The evaluation of components of various equilibriums from standard physical chemistry text books and research journals are examined. Finally the easy evaluation of the components using the handy relation is presented and verified.
4. Discussion
- Most of the textbooks give the example of decomposition of calcium carbonate reaction to illustrate the evaluation of number of components in an equilibrium reaction (and so does Glasstone1).
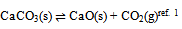 | (1) |
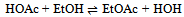 | (2) |
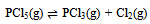 | (3) |
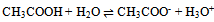 | (4) |
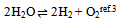 | (5) |
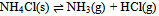 | (6) |
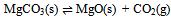 | (7) |
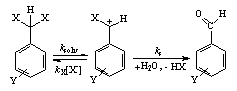 | Scheme 1. Depicting the solvolysis of benzyl-gem-dichloride |
5. Conclusions and Results
- The system is discussed and concluded as correct when the result obtained from C = C’ – r is the same as we imagined. In the forgoing examples for the illustration of evaluation of components, uniformity in the explanation is not maintained. Therefore a general expression for evaluation of components would be of much benefit, both to the teacher and taught. The number of components in a system can be determined in the following general manner1.Let C’ be the number of chemical constituents in the system, and let r be the number of restrictions imposed on the independent variation of these constituents. The number of components C is given by the equation:
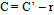 | (8) |
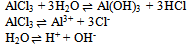 And one condition of electrical neutrality
And one condition of electrical neutrality 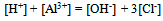 Hence for this system, C’ = 7 and r = 4 i.e. the three equilibriums and the condition of electrical neutrality restrict the independent variation of the constituents. Hence C = 7 – (3+1) = 3.Let us take a similar but a little more complex example9 where in one finds the number the components of (a) Na2HPO4 in water at equilibrium with water vapour but disregarding the fact the salt is ionized. (b) The same but taking into account the ionization of the salt (disregarding the water vapour). In the former case (a) total numbers of constituents (C’) are three, namely, salt, water and water vapour, there is an equilibrium condition (restriction) between liquid water and its vapour. Hence the total number of components are C = 3 – 1 = 2; In the latter case (b) there are seven (C’) species: Na+, H+, H2PO4-, HPO42-, PO43-, H2O and OH-. There also exist three equilibriums (disregarding the water vapour), namely
Hence for this system, C’ = 7 and r = 4 i.e. the three equilibriums and the condition of electrical neutrality restrict the independent variation of the constituents. Hence C = 7 – (3+1) = 3.Let us take a similar but a little more complex example9 where in one finds the number the components of (a) Na2HPO4 in water at equilibrium with water vapour but disregarding the fact the salt is ionized. (b) The same but taking into account the ionization of the salt (disregarding the water vapour). In the former case (a) total numbers of constituents (C’) are three, namely, salt, water and water vapour, there is an equilibrium condition (restriction) between liquid water and its vapour. Hence the total number of components are C = 3 – 1 = 2; In the latter case (b) there are seven (C’) species: Na+, H+, H2PO4-, HPO42-, PO43-, H2O and OH-. There also exist three equilibriums (disregarding the water vapour), namely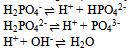 Further there are also two conditions of neutrality, namely[Na+] = [phosphates] and [H+] = [OH-] + [phosphates] where [phosphates] = [H2PO4-] + 2[HPO42-] + 3[PO43-]Hence the total numbers of restrictions are the three equilibriums, and the two conditions of neutrality i.e., they restrict the independent variation. Thus the total number of components (C) are C = 7 – (3 + 2) = 2. The comprehension of the important term restriction is better understood if one studies the derivations of phase rule. The derivation of phase rule10 was first put forth by J. Willard Gibbs in 1878. Unfortunately it was published in a rather obscure journal Transactions of the Connecticut Academy of Arts and Sciences11 and was overlooked for 20 years. The derivation of phase rule like any other derivations has essential concepts and assumptions embedded in it. It is here the teaching acumen of the instructor is wanting, and it is for the teacher to discern and accentuate the number of intensive variables fixed by free energy equilibrium relations. Let us recapitulate the phase rule obtained by J. Willard Gibbs.Consider the C components to be distributed throughout each of the P phases of a system as schematically indicated in Figure 1. To get the total number degrees of freedom one has to first add the total number of intensive variables required to describe separately each phase and then subtracting the number of intensive variables, whose values are fixed by free-energy equilibrium relations between different phases. To begin, each component is assumed to be present in every phase.
Further there are also two conditions of neutrality, namely[Na+] = [phosphates] and [H+] = [OH-] + [phosphates] where [phosphates] = [H2PO4-] + 2[HPO42-] + 3[PO43-]Hence the total numbers of restrictions are the three equilibriums, and the two conditions of neutrality i.e., they restrict the independent variation. Thus the total number of components (C) are C = 7 – (3 + 2) = 2. The comprehension of the important term restriction is better understood if one studies the derivations of phase rule. The derivation of phase rule10 was first put forth by J. Willard Gibbs in 1878. Unfortunately it was published in a rather obscure journal Transactions of the Connecticut Academy of Arts and Sciences11 and was overlooked for 20 years. The derivation of phase rule like any other derivations has essential concepts and assumptions embedded in it. It is here the teaching acumen of the instructor is wanting, and it is for the teacher to discern and accentuate the number of intensive variables fixed by free energy equilibrium relations. Let us recapitulate the phase rule obtained by J. Willard Gibbs.Consider the C components to be distributed throughout each of the P phases of a system as schematically indicated in Figure 1. To get the total number degrees of freedom one has to first add the total number of intensive variables required to describe separately each phase and then subtracting the number of intensive variables, whose values are fixed by free-energy equilibrium relations between different phases. To begin, each component is assumed to be present in every phase. 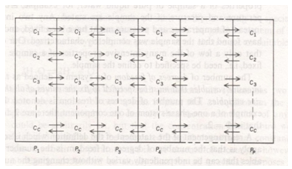 | Figure 1. Depicting ‘C’ components distributed throughout each of the ‘P’ phases |
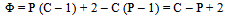 In this derivation of phase rule C (P – 1) as already described, is the number of intensive variables, whose values are fixed by free energy equilibrium relations. Teachers should try to emphasize this aspect and try to be as lucid as possible. This would enable the students to comprehend the extremely important term ‘r’ that is the restriction13 imposed on the independent variation of the constituents. Once the students comprehend this, the evaluation of components would be a blithesome experience.One million dollar perplexing query, the students raise, is that in the decomposition of CaCO3, why isn’t [CaO] = [CO2] although it is analogous to [CH3COO-] = [H3O+] for reaction 4 or [EtOAc] = [HOH] for reaction 2 or [PCl3] = [Cl2] for reaction 3. The thermodynamic property, intensive variable, answers this query. The “concentration” of a solid, like its density, is an intensive property and therefore does not depend on how much of the substance is present. For example, the “molar concentration” of copper14 (density: 8.96g/cm3) at 20oC is the same, whether we have 1 gram or 1 ton of the metal:[Cu] = 8.96g/1 cm3 x 1 mol/63.55g = 0.141 mol/cm3 = 141 mol/LFor this reason, the concentration of solid [CaO] is constant, and thus has nothing to do with concentration of [CO2]. And since [CaO] is not equal to [CO2], the number of restrictions is reduced to one in this case. The same reasoning can be extrapolated for decomposition of MgCO3, where [MgO] is not equal to [CO2].Atkins15 has a self-test problem, which is, “How many components does autoprotolysis of water has?” and the answer given is one. Let us solve this problem as follows:
In this derivation of phase rule C (P – 1) as already described, is the number of intensive variables, whose values are fixed by free energy equilibrium relations. Teachers should try to emphasize this aspect and try to be as lucid as possible. This would enable the students to comprehend the extremely important term ‘r’ that is the restriction13 imposed on the independent variation of the constituents. Once the students comprehend this, the evaluation of components would be a blithesome experience.One million dollar perplexing query, the students raise, is that in the decomposition of CaCO3, why isn’t [CaO] = [CO2] although it is analogous to [CH3COO-] = [H3O+] for reaction 4 or [EtOAc] = [HOH] for reaction 2 or [PCl3] = [Cl2] for reaction 3. The thermodynamic property, intensive variable, answers this query. The “concentration” of a solid, like its density, is an intensive property and therefore does not depend on how much of the substance is present. For example, the “molar concentration” of copper14 (density: 8.96g/cm3) at 20oC is the same, whether we have 1 gram or 1 ton of the metal:[Cu] = 8.96g/1 cm3 x 1 mol/63.55g = 0.141 mol/cm3 = 141 mol/LFor this reason, the concentration of solid [CaO] is constant, and thus has nothing to do with concentration of [CO2]. And since [CaO] is not equal to [CO2], the number of restrictions is reduced to one in this case. The same reasoning can be extrapolated for decomposition of MgCO3, where [MgO] is not equal to [CO2].Atkins15 has a self-test problem, which is, “How many components does autoprotolysis of water has?” and the answer given is one. Let us solve this problem as follows: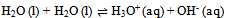 Pure liquid water has concentration of 55.5M (density = 1g/cc and calculations analogous to concentration of solids, as already explained in the foregoing paragraph), and since negligible concentrations of products are formed, the concentration of water can be taken as constant. The equilibrium expression for this process is Kw = [H3O+][OH-]. Therefore if concentration of H3O+ is known, the concentration of OH- is set. Analogously, if the concentration of OH- is known, the concentration of H3O+ is set. Hence auto-protolysis is one component system. Thus this example is underpinning the reasoning; put forth in the foregoing paragraph regarding concentration of solids (it is equally applicable for pure liquids).If we apply our generic formula, then C’ is 4 and r is 3 i.e. the equilibrium condition, the relation [H3O+] = [OH-] and the relation [H2O] = [H2O]. Therefore C = 4 - 3 i.e. 1. Thus this simple relation of C = C’ - r is a silver-bullet for freshman students to evaluate the number of components (for simple equilibrium reactions) when learning the phase rule.
Pure liquid water has concentration of 55.5M (density = 1g/cc and calculations analogous to concentration of solids, as already explained in the foregoing paragraph), and since negligible concentrations of products are formed, the concentration of water can be taken as constant. The equilibrium expression for this process is Kw = [H3O+][OH-]. Therefore if concentration of H3O+ is known, the concentration of OH- is set. Analogously, if the concentration of OH- is known, the concentration of H3O+ is set. Hence auto-protolysis is one component system. Thus this example is underpinning the reasoning; put forth in the foregoing paragraph regarding concentration of solids (it is equally applicable for pure liquids).If we apply our generic formula, then C’ is 4 and r is 3 i.e. the equilibrium condition, the relation [H3O+] = [OH-] and the relation [H2O] = [H2O]. Therefore C = 4 - 3 i.e. 1. Thus this simple relation of C = C’ - r is a silver-bullet for freshman students to evaluate the number of components (for simple equilibrium reactions) when learning the phase rule. Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-Text HTML
Full-Text HTML