-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Nanoscience and Nanotechnology
p-ISSN: 2163-257X e-ISSN: 2163-2588
2012; 2(3): 22-48
doi: 10.5923/j.nn.20120203.01
Mechanical Milling: a Top Down Approach for the Synthesis of Nanomaterials and Nanocomposites
Thakur Prasad Yadav1, 2, Ram Manohar Yadav3, Dinesh Pratap Singh4
1Department of Physics, The University of Liverpool, Liverpool, L69 3BX, UK
2Department of Physics, Banaras Hindu University, Varanasi-221005, India
3Department of Physics, VSSD College, Kanpur, India-208002
4Avenida Ecuador 3493, Estación Central, Departamento de Física, Universidad de Santiago deChile Santiago-9170124, Chile
Correspondence to: Thakur Prasad Yadav, Department of Physics, The University of Liverpool, Liverpool, L69 3BX, UK.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
Synthesis of nanomaterials by a simple, low cost and in high yield has been a great challenge since the very early development of nanoscience. Various bottom and top down approaches have been developed so far, for the commercial production of nanomaterials. Among all top down approaches, high energy ball milling, has been widely exploited for the synthesis of various nanomaterials, nanograins, nanoalloy, nanocomposites and nano -quasicrystalline materials. Mechanical alloying techniques have been utilized to produce amorphous and nanocrystalline alloys as well as metal/non-metal nanocomposite materials by milling and post annealing, of elemental or compound powders in an inert atmosphere. Mechanical alloying is a non-equilibrium processing technique in which different elemental powders are milled in an inert atmosphere to create one mixed powder with the same composition as the constituents. In high-energy ball milling, plastic deformation, cold-welding and fracture are predominant factors, in which the deformation leads to a change in particle shape, cold-welding leads to an increase in particle size and fracture leads to decrease in particle size resulting in the formation of fine dispersed alloying particles in the grain-refined soft matrix. By utilizing mechanical milling various kind of aluminium/ nickel/ magnesium/ copper based nanoalloys, wear resistant spray coatings, oxide and carbide strengthened aluminium alloys, and many other nanocomposites have been synthesized in very high yield. The mechanical milling has been utilized for the synthesis of nanomaterials either by milling and post annealing or by mechanical activation and then applying some other process on these activated materials. This review is a systematic view of the basic concept of mechanical milling, historical view and applications of mechanical milling in the synthesis of various nanomaterials, nanosomposites, nnaocarbons and nano quasicrystalline materials.
Keywords: Mechanical Milling, Nanomaterials, Nanocomposites, Nanostructure, Synthesis, Mechanical Properties, Processing-Structure-Property
Article Outline
1. Introduction
1.1. Mechanical Milling
- In mechanical milling (MM), a suitable powder charge (typically, a blend of elemental) is placed in a high energy mill, along with a suitable milling medium. The objective of milling is to reduce the particle size and blending of particles in new phases. The different type of ball milling can be used for synthesis of nanomaterials in which balls impact upon the powder charge[1]. The balls may roll down the surface of the chamber in a series of parallel layers or they may fall freely and impact the powder and balls beneath them. For large scale production with nano size grain, mechanical millings are more economical processes[2]. The kinetics of mechanical milling or alloying depends on the energy transferred to the powder from the balls during milling[3]. The energy transfer is governed by many parameters such as the type of mill, the powder supplied to drive the milling chamber, milling speed, size and size distribution of the balls, dry or wet milling, temperature of milling and the duration of milling[4]. Since the kinetic energy of the balls is a function of their mass and velocity, dense materials (steel or tungsten carbide) are preferable to ceramic balls, and the size and size distribution should be optimized for the given mill[5]. Too dense packing of balls reduces the mean free path of the ball motion, while a dilute distribution minimizes the collision frequency[6]. The temperature during milling can depend on the kinetic energy of the ball and the material characteristics of the powder and milling media. The temperature of the powder influences the diffusivity and defect concentration in the powder influencing the phase transformations induced by milling[7]. Higher temperatures are expected to result in phases which need higher atomic mobility (intermetallics) while at lower temperatures the formation of amorphous phases is expected if the energy is sufficient[8]. Low temperature can also enhance the formation of nanocrystalline phases[9]. In addition, the high strain-rate deformation and cumulative strain accompanying during collisions of balls lead to particle fracture[10]. These competing fracture and coalescence events continue throughout processing. Indeed, a suitable balance between them is required for success with the alloying process. In most application, the balance is such that an approximate steady-state powder size distribution is obtained[11]. In this stage, the particles are often shaped like flakes, and even through a steady-state powder size are found, continued microstructural refinement occurs as a result of the repetitive fragmentation and coalescence events[12].
1.2. Synthesis of Materials by Mechanical Milling: A Historical Development
- The synthesis of materials by high energy ball milling of powders was first developed by John Benjamin (1970)[13] and his co-workers at the International Nickel Company in the late 1960's. The goal of this work was the production of complex Oxide Dispersion-Strengthened (ODS) alloys for high temperature structural applications. It was found that this method, termed mechanical alloying, could successfully produce fine, uniform dispersions of oxide particles (Al2O3, Y2O3, ThO2) in nickel-base superalloys which could not be made by more conventional powder metallurgy methods. In addition, reactive alloying components such as Ti, could be incorporated from master alloy powders such that it was clear that "alloying" could occur on the atomic level during the mechanical milling process. Benjamin and his co- workers at Inca Paul D. Merica Research Laboratory also explored the synthesis of other kinds of materials, e.g. solid solution alloys and immiscible systems, and pointed out (Benjamin, 1977) that mechanical alloying (MA), in addition to synthesis of dispersion-strengthened alloys, could make metal composites, compounds, and/or new materials with unique properties[14]. However, one of the major applications of MA so far has been the production of oxide- dispersion-strengthened (ODS) materials[15].
2. Mechanical Milling: Equipment and Process Variables
- The milling of materials has been a major component of the mineral, ceramic processing, and powder metallurgy industries. The objectives of mechanical milling include particle size reduction, mixing or blending, particle shape changes and synthesis of nanocomposite. The typical mill used for these purposes has been the high energy ball mill such as tumbler ball mills, vibratory mills, planetary mills, and attritor mills (Szegvari attritor) etc. A detailed description of the different mills available for MM can be found in Suryanarayana handbook [25]; however we have discussed a brief description regarding high energy ball mills.
 | Figure 1. A rock tumbler Ball mills |
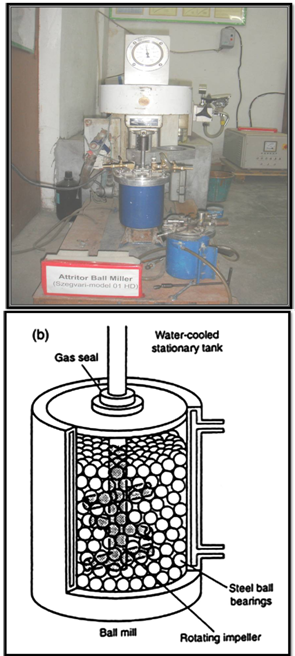 | Figure 2. (a) High energy attritor ball mill (b) Arrangement of rotating arms on a shaft in the attrition ball mill (department of Physics, BHU) |
 | Figure 3. SPEX 8000 mixer/mill in the assembled condition |
 | Figure 4. Planetary ball mill (RETSCH PM 400: department of Physics, BHU) |
2.1. Mechanical Milling: Mechanics and Physics
- The central occurrence in mechanical milling is the ball-powder-ball collision. Powder particles are trapped between the colliding balls during milling and undergo deformation and/or fracture processes which define the ultimate structure of the powder. The nature of these processes depends upon the mechanical behaviour of the powder components, their phase equilibria, and the stress state during milling. It is convenient to classify the powder components into (i) ductile - ductile, (ii) ductile - brittle, and (iii) brittle - brittle systems.
2.1.1. Ductile - Ductile Components
- The phenomenological description of the mechanical milling of ductile components (and ductile -brittle components) was first presented by Benjamin and Volin (1974) [41]. They discuss the mechanical alloying of ductile components in terms of a competition between cold-welding and fracture. They divided the mechanical alloying process into five sequences as delineated by observations with optical microscopy of the powders at various stages of milling. Initially, micro-forging flattens ductile powders into plates and fragments more brittle components into finer particles. A schematic drawing of a ball-powder-ball collision of a powder mixture is given in Fig. 5. Extensive cold-welding follows the first stage with a composite lamellar structure of the ductile components in the form of plates. At longer milling times, the composite powder particles are further refined, the lamellar spacings decrease, and lamellae become convoluted. At this stage, alloying begins, aided by any heating that may be introduced by milling, the enhanced diffusion paths of the lattice defects created by the deformation, and the shortened diffusion paths as the lamellae become finer and more convoluted. Eventually, the lamellar spacing becomes too fine to be resolved by optical microscopy. With continued milling, components which exhibit complete solid solubility mix too the atomic level, i.e. a true alloy is formed.
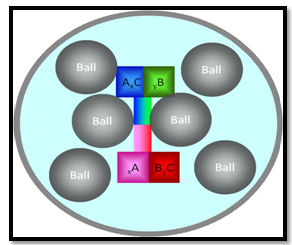 | Figure 5. Ball-powder- ball collision of powder mixture during mechanical milling |
2.1.2. Ductile -Brittle Components
- The evolution of microstructure during mechanical milling of ductile - brittle systems has also been described phenomenological[42-44]. The hard brittle powders are fragmented during milling, and the fragments are trapped at the boundaries between the ductile powders. As milling proceeds and the welds between ductile particles come closer together - and finally blend - a fine dispersion of the brittle phase results if it is insoluble in the ductile matrix, as inert oxides in ODS alloys. However, brittle intermetallics are also incorporated into, for instance, Ni-base ODS superalloys by mechanical alloying. The brittle intermetallics are fragmented but apparently alloy with the Ni-base matrix so that they are not resolvable by optical microscopy when mechanical alloying is complete. Another example of the alloying of ductile / brittle components by milling wherein a homogeneous alloy is formed is the mixture of Zr (ductile) with NiZr2 (brittle) powder to form an amorphous Ni-24 at % Zr alloy[45]. Schultz et al (1988)[46] did not observe the alloying of brittle amorphous boron particles into Fe, Fe-Si or Fe-Zr powders by ball milling. In all cases the boron particles were dispersed in the metal matrix. In Fe-Zr-B the boron did alloy after annealing the dispersion below the crystallization temperature of the amorphous Fe-Zr alloy. Brittle Si did form an alloy with Fe during mechanical alloying. These observations presumably reflect the differences in the mutual solid solubility’s of the component. Boron has negligible solubility in Fe, while Si exhibits significant solid solubility in Fe. Similarly, the stable oxide disperse solids, such as Al2O3, Y2O3, ThO2, are essentially insoluble in the metal matrices of ODS alloys in which they are incorporated by mechanical alloying. Thus, alloying of ductile - brittle components during milling requires not only that the brittle particles become fragmented so that short-range diffusion can occur, but the brittle component must also have some solubility as indicated by either the stable or metastable phase equilibriums in the ductile matrix module.
2.1.3. Brittle- Brittle Components
- It might be expected that mechanical alloying of brittle - brittle powder systems would not occur and that milling would simply reduce the brittle powder components size down to what is called the limit of combination. The grinding of brittle mineral powders to achieve fine particles for subsequent processing reaches a limit on particle size below which further fracturing stops the limit of combinations[47]. It has been suggested that this limit may be due to extremely small particles deforming plastically rather than fracturing, the increased cohesion between fine particles leading to aggregation, or phase changes in surface layers.It has been shown; however, that milling of certain nominally brittle components can lead to homogeneous alloys. The brittle - brittle systems which have been studied in this regard include Si/Ge (which form solid solutions) (Davis and Koch, 1987)[48], Mn/Bi (which forms the intermetallic compound MnBi), (Davis et al., 1988)[49] and various mixtures of intermetallic compound such as NiZr2/Ni11Zr9 (Lee and Koch 1987)[50]. Davis and Koch (1987)[48] observed the formation of a Si-Ge solid solution on mechanical alloying of the pure components which are both nominally brittle at ambient temperature. The solid solution formation was detected by measurement of precise lattice parameters for Si and Ge as a function of milling time. The lattice parameters of Si and Ge move towards each other with increasing milling time until they merge to a single value, which is identical to that for the solid solution of this composition (Si-28 at % Ge) prepared by conventional metallurgical methods. Thus, it is possible to attain atomic level alloying by milling brittle components together. The microstructural evolution during milling differs markedly from the lamellar morphology of ductile components. A granular morphology is observed during mechanical milling of Ge and Si powder milled for two hours in a SPEX mill. It would appear that the harder Si particles are embedded in the softer Ge. It was also noted in the Si-Ge system that mechanical alloying was suppressed by milling in a vial cooled by liquid nitrogen. In this brittle- brittle system, thermal activation- diffusion is apparently a critical requirement for mechanical alloying. In contrast to this observation, mechanical alloying has been achieved at sub-ambient temperatures in ductile - ductile systems. This difference may reflect the longer diffusion distances required in the brittle - brittle granular vs. ductile - ductile lamellar geometry, and the enhanced diffusion paths provided by severe plastic deformation in ductile - ductile systems. The material transfer in the brittle powder agglomerates during milling may be related to the conditions that control the "limit of communication", as described above. When the limit of combination is reached in multi- component brittle powders, the conditions may be favourable for the cold welding or other material transfer mechanisms that result in mechanical alloying[51].
2.2. Mechanical Milling: Temperature Effects
- The temperature during milling can depend on the 'kinetic energy' of the ball and the material characteristics of the powder and milling media. The temperature of the powder influences the diffusivity and defect concentration in the powder influencing the phase transformations induced by milling. Higher temperatures are expected to result in phases which need higher atomic mobility (intermetallics) while at lower temperatures the formation of amorphous phases is expected if the energy is sufficient. Low temperature can also enhance the formation of nanocrystalline phases.The measured maximum temperature of the SPEX mill was 50℃ with 13 balls (40℃ without leads) and concluded the most of the heat comes from the motor and bearings [52]. Kimura and Kimura reported a maximum macroscopic temperature of 172℃ in the attritor ball mill[53]. Borzov and Kaputkin have measured a temperature in the range 100-215℃ for an attritor using SiC and diamond sensors [54]. Thus the macroscopic temperature during milling appears to be low and sensitive to the mill design. The microscopic temperature of the powder just after the impact can be realized very high, often exceeding the melting points of some of the component metals. As it is not possible to measure the microscopic temperature during milling, investigators look two approaches to estimate it. One of the approaches is to calculate it by appropriate models and the other is to infer the temperature from the structural, microstructural changes during milling[55].
3. Nanomaterials and Nanocomposites
3.1. Al-based Nanomaterials and Nano Composites
- J. Sun et al[56] investigated the microstructural evolution of the ordered L12+DO22 multiphase Al67Mn8Ti24Nb1alloy by mechanical milling and subsequent annealing. The ordered L12+DO22 multiphase Al67Mn8Ti24Nb1alloy first transformed into disordered fcc supersaturated solid solution after 15 h milling and again into the full amorphous structure as the milling proceeded to 60 h. The full amorphization of the Al67Mn8Ti24Nb1alloy was resulted due to introduction of high dense nano-crystalline grain boundaries and the large internal lattice strain caused by the super-saturated dissolving of the Al3(TiNb) second phase in fcc solid solution. P. Nandi et al[57] synthesized amorphous and/or nanocrystalline Al-based alloys from elemental powder blends with the stoichiometry Al65Cu35-xNbx(x=/5-25 at.% Nb) by high energy planetary ball milling. The mechanical alloying of Al65Cu30Nb5, Al65Cu20Nb15and Al65Cu10Nb25powder blends for appropriate time (30-/50 h) in a planetary or shaker mill resulted into the formation of a single phase nanocrystalline disordered metallic phase, an amorphous alloy and a nano-aluminide mixture of NbAl3, and fcc-Nb solid solution or Nb(CuAl), respectively. Presence of Nb in appropriate amount played a crucial role for introducing adequate disorder and solid state amorphization of these ternary system. They showed that, mechanical alloying was a potential route of synthesizing Al-based nanocrystalline or amorphous matrix Al-Cu-Nb alloy powders/composites for applications below 350℃. By starting from the elemental powder M. Zawrah et al[58] investigated the microstructural and compositional evolution of Al93Fe3Ti2Cr2alloys via mechanical alloying. They studied the potential of the MA-processed Al93Fe3Ti2Cr2alloy for structural applications at both ambient and elevated temperatures. The nanocrystallization process of the Al alloy proceeded via an initial increase followed by a subsequent decrease in dislocation density to form nano-grains with a dislocation-free interior. I. Manna et al[59] reported the formation of amorphous and/or nanocrystalline products by mechanical alloying of Al65Cu35−xZrx(x = 5, 15 and 25at.% Zr) elemental powder by planetary ball milling up to 50h. Mechanical alloying of Al65Cu35−xZrx(x = 15, 25, 5at.% Zr) by planetary ball milling up to 50h led to the formation of a single-phase amorphous microstructure in Al65Cu20Zr15, nanocrystalline and amorphous mixture in Al65Cu10Zr25, and nanocrystalline intermetallic phases in Al65Cu30Zr5, respectively. Isothermal annealing of the final milled (50h) and amorphous Al65Cu20Zr15alloy at 400–800 K in vacuum for 2 h resulted the precipitation of nanocrystalline intermetallic phases. Al–Zn/Al2O3and Al–Zn–Cu/Al2O3 were prepared by T.G. Duraiet al[60] via reaction sintering of partially reacted oxide mixtures derived from a high-energy ball milling process. It was observed that the wear resistance of the milled composites was better than that of the unmilled composites, since Al2O3 particle size played important role in bearing the external load, and there was a good interfacial cohesion between Al2O3 particles and the matrix. They observed that the wear rate increased linearly with the applied load irrespective of the material and decreased with the increasing sliding distance. The coefficients of friction of the composites against EN32 steel decreased with the decreasing alumina particle size as well as the crystallite size of the matrix. Nanocrystalline Nb3(Al1-xGex) mixtures with x = 0, 0.3 and 1 were fabricated by E. Pusceddu et al[61] by wet mechanical milling using copper milling media and distilled water as a process control agent (PCA) with the intention of improving the yield, properties and the performance of these materials. Very high yields of nanocrystalline material were achieved but significant copper contamination. A typical grain size of a few nm was obtained for the Nb-Al-Ge mixtures after several hours milling. M. Khakbiz et al[62] synthesized the Al–B4C nano-composite powders by mechanical alloying. Firstly they milled the commercially available (0.7 μm) boron carbide (B4C) particles, in a attritor ball mill and prepared B4C nano-particles. Secondly the mechanical alloying was employed to synthesize metal matrix composite powders with a nanocrystalline Al 6061 alloy as the matrix and B4C as the reinforcement. Different amounts of B4C particles (5 wt.% and 10 wt.%) having various sizes of 90 nm (produced in the first part of study), 0.7 μm and 1.2 μm were mixed with different sized (21 μm and 71 μm) Al 6061 powder particles and were milled for different times. The results showed that the nano-sized B4C particles were fabricated when milled for 110 h. The size of powder particles in the milled powder mixture was affected by the initial size and content of B4C particles and Al powders. The crystal size of aluminum reached to 57 nm after 16 h milling of powder mixture and addition of B4C resulted in a finer grain size of Al in the Al + B4C mixture during the early stages of milling. High energy mechanical milling of a mixture of alumina and nanostructured Fe48Co52powders was used in producing an Al2O3-20vol%Fe48Co52composite powder by M.Yusop et al [63].
3.2. Mg-based Nanomaterials and Nanocomposites
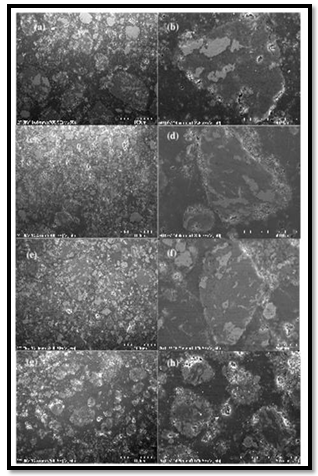
- Nanocrystalline and nano-/amorphous Mg1.9M0.1Ni (M5Ti, Zr,V) alloys were synthesized by T. Spassov et al[69] by mechanical alloying followed by annealingdepending on the milling parameters as rotation speed, time of milling and annealing (temperature and time of annealing) conditions different microstructures were obtained.The milled materials had hexagonal Mg2Ni crystal structure and the crystallites size of the milled as well as of the milled and annealed alloys was in the range of 10–20 nm. The heating of the as obtained materials above 700 K increased the grain size. High energy ball milling has been proved a suitable way for processing Mg based nanostructured material, especially when milling is carried out under a hydrogen atmosphere, which provides a nanostructured and hydrogenated final product. J.F.R. de Castro et al[70] investigated the structural evolution of Mg, Nb and a mixture of Mg–5 at.%Nb by reactive milling. They showed that Nb rapidly absorbed hydrogen, and formed the NbH0.9 phase whereas Mg was only partially transformed into ß-MgH2 and γ-MgH2 phases. When Mg–5 at.%Nb mixture was milled for 48 h, almost all Mg and Nb were transformed into hydridesindicating the catalytic effect of Nb on the hydrogen sorption of Mg. By utilizing the elemental powders of Mg, Al and Ti with polyethylene–glycol L. Lu et al[71] synthesized the Nanocomposite Mg5wt%Al– 10.3% Ti via mechanochemical milling. Nanoparticles were embedded in the Mg matrix and the grain size of the milled specimen was about 77 nm.TiH2 was also observed after milling and the concentration of TiH2increased after sintering. These nanocomposite showed an improvement in yield strength and ductility compared to their counterpart fabricated by using a conventional powder metallurgy process.The structure and magnetic properties of spinel-related Mg-doped Li0.5Fe2.5O4 nanocrystalline particles prepared by milling a pristine sample for different times were investigated by Widatallah et al[72]. Mg-doped Li0.5 Fe2.5 O4 of the composition Li0.41Fe2.41 Mg0.17 O4 was prepared by sintering in air at 600℃ (12 h) a 5: 1 molar mixture of hydrothermally prepared Mg-doped α-Fe2O3 and Li2CO3. The material was then milled in air using a Retsch PM400 planetary ball mill with stainless steel balls for periods of 6.5, 15, 38 and 50 h keeping fixed the ball-to-powder mass ratio at 20: 1. TEM micrographs of samples obtained after milling for different times (Figure 7). In Fig. 7(a) the non-milled ferrite was mostly formed of cubic particles with an average size of ∼0.2 mm. Milling the material for 6.5 h resulted in irregular particle shape distributed over a broad size range extending from ∼40 nm to ∼0.3 mm (figure 7(b)). Further fragmentation and shape irregularity were observed for the sample milled for 38 h which was found to have a particle size distribution ranging between ∼20 nm and∼0.2 mm (figure 7(c)). After 50 h f milling (figure 7(d)) a mixture of spherical and irregularly shaped nanoparticles with an average size of ∼15nm was obtained. These nanoparticles tended to agglomerate forming grains that vary in size between ∼50 and ∼150nm. Nano-sized BaxMg1−xFe2 O4 ferrite particles less than about 150 nm were prepared by Y. Choi et al[73] by using self-propagating high temperature synthesis and mechanical ball milling. It was observed that as the ratio of BaO2to MgO in the initial powder composition varied from 0.25 to 4.0, coercive force decreased about 102%.
3.3. Co-based Nanomaterials and Nanocomposites
- Zhou et al.[77] reported that the initially ordered intermetallic compound Co2Si with orthorhombic structure was resulted in to anti-site disorder and nanocrystalline material through mechanically milling in a high-energy ball mill. The generation of anti-site disorder in Co2Si was confirmed by the continuous increase of magnetization by a factor often. The average crystallite size is about 8-10 nm after long milling time. Both anti-site disorder and grain boundaries were major sources of energy storage during ball milling. Ultrafine Co and Ni particles were prepared by mechanochemical processing by[78]. These uniform sized (10-20 nm) particles were obtained by the chemical reduction of cobalt and nickel chlorides during mechanical milling with Na. The excess of NaCl was used as a diluents to prevent the occurrence of combustion during milling. The magnetic properties of the ultrafine powders were reducted in magnetization relative to their bulk values and the coercivities were significantly increased. Elina Manova et al.[79] prepared cobalt ferrite, CoFe2O4, nanoparticles from pre-prepared layered double hydroxide carbonate, LDH-CO3, by mechanical milling and the particle size were controlled by the treatment time. The 5h milling of the LDH-CO3 resulted cobalt ferrite nanoparticles. Longer- term milling induced particle growth, while prolonged milling resulted in the formation of some cobalt metal. They showed that the magnetic properties exhibited a strong dependence on the particle size as a result of an unusual action distribution and of surface effect. By this method large scale CoFe2O4nanoparticles can be synthesized by the combination of co-precipitation under ultrasonic treatment and subsequent mechanical milling. The nano-crystalline rare earth doped WC/Co powder with grain size of 8.45 nm was prepared by L. Sha et al[80] by high energy ball milling. The results showed that adding trace rare earth elements into carbides is effective to minimize the grain size of WC/Co powder. They observed that the grain size of rare earth doped powder became two times smaller as compared with the undoped powder within ball milling times of 25–45 h. The rare earth doped powder with grain size of 8.45 nm was obtained after 45 h ball milling. The Fe48Co48V4alloy was synthesized by B. Chitsazanet al[81] in a planetary high-energy ball-mill under an argon atmosphere. Inter-metallic Co3V was also observed during the mechanical alloying. The lattice parameter decreased up to 55 h of milling time with an oscillation and then increased from 55 to 125 h of milling time.
3.4. Ni- based Nanomaterials and Nano Composites
- Doppiu et al.[82] demonstrated that Ni can be formed controllably by partial reduction of NiO upon reactive ball milling, leading to Ni-NiO nanocomposites. Mechano- chemical reduction of NiO was carried out at room temperature by ball milling under H2 atmosphere. During the milling, NiO gradually converted into Ni, without formation of any intermediate phases. The amount of Ni and its crystallite size were tuned by the milling parameters, which led to the formation of Ni-NiO (metal- oxide) nanocomposites. Besides the initial microstructure of the NiO powder also played an important role in the reduction rate and on the final microstructure of the nanocomposites. Their results indicated that the mechanically induced defects had a strong influence on the kinetics of the reduction process. Nanocrystalline Ni0.5Zn0.5Fe2O4 spinel ferrite with a grain size of 50 nm was prepared by N Sivakumar et al[83] by using the ceramic method. The grain size was further reduced to 14 nm by milling the as-prepared ferrite particles in a high-energy ball mill. The increase in conductivity of the milled sample was, therefore, attributed to conduction by the oxygen vacancies created by mechanical milling. The higher values obtained for the activation energy for conduction were also evidence for the oxygen vacancy conduction. The increase in Neel temperature from 573 to 611 K on reducing the grain size from 50 to 14 nm were explained based on the changes in the cation distribution. The observed increase in the coercivity of the milled sample has been attributed to surface anisotropy of increasing number of ions on the surface. M. N. Mancheva et al[84] synthesized the nanocrystalline NiWO4 powders by mechano-chemical activation. A stoichiometric mixture of NiO and WO3 in a 1:1 molar ratio was subjected to intense mechanical treatment in air using a planetary ball mill for different periods of time. It was found that 7.5 h of milling of the reagents led to complete crystallization of single nanostructured phase NiWO4at room temperature. NiFe-CNT and Ni3Fe-CNT nanocomposites were fabricated by S. Azadehranjbar et al[85] by high energy mechanical alloying method. As obtained ball milled powders were heat treated at 500℃ for 1 h to release the milling induced stresses. Bulk samples were prepared by sintering of cold pressed (300 MPa) samples at 1040℃ for 1 h. The hardness value of as-milled NiFe and Ni3Fe powders reached to 660 and 720 HV, respectively. It was proposed that the introduction of CNTs at the matrix led to decrease of particles size of composites and therefore the size of porosities in the bulk samples hence the bulk density of the samples increased.
3.5. Cu-based Nanomaterials and Nano Composites
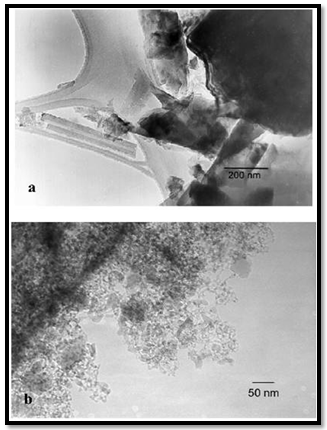
- Nano-crystalline copper with a mean crystallite size of 27 nm was synthesized by S. Sheibani et al[86] through solid state reduction of Cu2O by graphite using high energy planetary ball mill. Mechanochemical reduction of Cu2O with graphite resulted in a continuous decrease in the Cu2O mean crystallite size and an increase in internal strain during initial stage of milling operation. They observed that as the milling time was increased, fine Cu powders are produced and the crystallite size and internal strain of the copper particles changed irregularly with further milling time due to the progressive formation of copper during milling. Prolonged milling resulted in the agglomeration of the copper powder. The mean crystallite size of the copper produced after 30 h of ball milling was about 27 nm. High energy mechanical milling (HEMM) of a mixture of Cu powder and Al2O3 nanopowder was used to produce Cu-(2.5-10)vol.%Al2O3 nanocomposite powders with a ultrafine grained or nanocrystalline Cu matrix by D. L. Zhang et al[87]. High energy mechanical milling was effectively used to disperse (2.5-10%) vol.% Al2O3 nanoparticles into a ultrafine grained or nanocrystalline Cu matrix. Using larger diameter balls or increasing the volume fraction of Al2O3 nanoparticles to 7.5% or higher allowed, synthesis of Cu-Al2O3 nanocomposite powders with nanocrystalline Cu matrix.Cu and Al2O3 were used as starting powders and two milling routes were used: in Route 1, the powder mixture was milled for 12 hours using 60 balls with a diameter of 12.5mm; and Route 2 was Route 1 plus further milling for 12 hours using 12 balls with a diameter of 12.5mm and 6 balls with a diameter of 25mm. In both routes, the ball to powder weight ratio was 5:1.The samples of the milled powders were annealed for 1 hour at different temperatures in a tube furnace which has a vacuum of 10-6 bar. It was observed that 5vol.%Al2O3, almost uniform dispersion of Al2O3nanoparticles in the Cu matrix was achieved with a relatively short time of 6 hours of milling, as shown in Figures 8(a) and (b). Increasing the milling time to 12 hours causes little change in terms of the dispersion of the Al2O3 nanoparticles (Figure 8(c) and (d)). In the Figure 8, the bright particles are Al2O3 nanoparticles. It appeared that in with a short milling time of 6 hours, the Al2O3 nanoparticles are predominately distributed along the grain boundaries, whereas with a short milling time of 12 hours, a large fraction of the Al2O3 nanoparticles are distributed inside the grains. For Cu- 5vol.%Al2O3, with Route 1 milling of up to 12 hours, ultrafine grains with sizes in the range of 100- 300nm formed. The second 12 hours milling using larger balls in Route 2 caused significant refinement of the grains into the size range of 50-150nm, as shown in Figure 8(e). Nanostructured Cu-7.5vol.%Al2O3 and Cu-10vol.%Al2O3 composite powders were produced by Mukhtar et al [88] from a mixture of Cu powder and Al2O3 nanopowder using two routes of high energy mechanical milling. The as-milled and annealed nanocomposite powder particles showed little increase of the Cu matrix grain sizes and decrease of micro-hardness after annealing at temperatures up to 400℃. However significant coarsening of Cu grains and Al2O3 nanoparticles and sharp decrease of micro-hardness was observed with increasing annealing temperature to 500℃.This confirmed that Cu-7.5vol % Al2O3 nanocomposite was thermally stable at temperatures up to 400℃. The degree of microstructure coarsening and micro-hardness decrease with annealing of the 24 hours milled (Route 2) powder was clearly smaller than that of the 12 hours milled (Route 1) powder annealed under the same condition.
3.6. Ti- based Nanomaterials and Nano Composites
- S. Indris et al [91] prepared nanocrystalline powders of the anatase TiO2 as well as the rutile TiO2 modification by high-energy ball milling of the coarse grained source materials for 4 h. The average grain size was varied via the milling time in the range from some micrometers down to about 20 nm. They showed that the catalytic activity of TiO2 powders was strongly influenced by the powder morphology and the electronic structure of the samples. It was observed that for all milling times the decrease of the grain sizes led to an increase in the BET surface area. Wang et al[92] reported the synthesis of stable Ti50Zr50 binary nano-composite of grain size about 7.6 nm by the roller vibration milling with a batch volume about 170 g at room temperature. The composite remained the basic hexagonal feature, but the axial ratio was much smaller than that of the ideal hexagonal close-packed structure and the standard value of Ti and Zr. For the alloying and insertion into the Ti matrix, the size reduction of Zr was very important. The grain size of Ti varied periodically with time, but the period was dependent on the grain size of Zr. L.X. Qiu et al[93] synthesized of TiN–TiB2 nano-composite by mechanical milling of mixture of Ti and hexagonal boron nitride (h-BN) and subsequent sintering under high pressure and high temperature. A TiN–TiB2 composite was fabricated by ball milling of the mixture for 70 h and then sintering at 5 GPa, 1300℃. X. Zhao et al[94] used mechanical milling to treat the crystalline Ti2Ni alloy prepared by solid-state sintering. The particle size of the alloy decreased first and then increased after the milling. Moreover, MM contributed to a decrease in grain size and the formation and increase of the amorphous phase by increasing the milling time, resulting in improvements of the antipulverization ability and cycle life after the milling. M. Ali et al[95] synthesized nano-structured TiC from mixtures of TiO2 and graphite through high- energy mechanical milling and subsequent heat treatment at a temperature that was considerably lower than that used in conventional carbothermal reduction synthesis of TiC. TiO2 powders mixed with graphite were milled in a planetary ball mill for 30–90 hand then milled mixtures were heat treated at 1200℃ and 1300℃ for 60 min. The crystallite size of TiC increased with increasing milling time after heat treatment. A nano-composite structure containing of an intermetallic matrix with dispersed Al2O3 particles was obtained by S. Alamolhoda et al[96] via mechanical activation of TiO2 and Al powder mixture for different time and subsequent sintering. The results revealed that the reaction began during milling by formation of Al2O3 and L12Al3Ti and further milling caused partial amorphization of powder mixture. Phase evolutions of the milled powders after being sintered revealed that by increasing the milling time and formation of L12Al3Ti in the milled powder, intermediate phase formed at 500℃ changes from DO22Al3Ti to Al24Ti8 phase. Increasing the milling time from 8 to 30 h caused formation of finer microstructures in composite and a reduction in crystallite size of TiAl phase from about 70 to 18 nm. Mashhadi et al [97] reported the synthesis of titanium nitride solid solution by the reduction of aluminum nitride with titanium by MM using a planetary ball mill. A nanostructure composite of in-situ titanium nitride solid solution was produced through exchange reaction between Ti and AlN. It was observed that, with increased milling time, Al resulted from decomposition of aluminum nitride dissolved in the Ti lattice, led to the formation of Ti(Al) solid solution and a reduction of the Ti lattice interplanar distance. Consequently, the Ti(Al) solid solution reacted with nitrogen to form in-situ Ti(Al)N nano particles. The milled powder had a spheroid shape and a very narrow size distribution of about 1 μm at the end of milling. In-situ Ti(Al)N particles with an average size of 10 nm were attained at a ball-to-powder weight ratio of 10:1 and a Ti:AlN molar ratio of 1:1.
3.7. C-based Nanomaterials and Nano Composites
- Awasthi et al[98] reported the formation of carbon in different nanoparticle forms by ball-milling of graphitic carbon. Ball-milling of graphite was carried out in attritor ball mill at room temperature in hexane medium for different times i.e. 24, 48 and. The self-coagulated carbon agglomerates were obtained in the case of 24 and BMC samples. The formation of coiled nanotubes and nanofibres were observed in the BMC sample. The BMC samples with and without nickel (Ni) catalyst were subjected to hydrogenation cycling also. The vibratory ball mill was used to decrease the single-wall carbon nanotubes length and to increase their specific surface area by N. Pierard et al[99]. Different measurements indicated that the optimum time of treatment seems to be 2 h. After that, the SWNTs quality decreased, with a complete disruption of the tubular structure leading to multi-layered polyaromatic carbon materials observed after 50 h.H. G. Connan et al[100] reported the formation of new nanocarbons by rod milling and annealing of graphite in the presence of yttrium. They investigated the mechanisms of ordering and disordering graphite in the presence of 1% and 10% yttrium through mechanical activation and subsequent thermal annealing at 1350℃ for 6 h. Graphite nano-sheets with high aspect ratio, having a thickness of the order of 10 nm, high shape asymmetry, and a well crystalline order, were produced by Antisari et al[101] by a mechanical method based on the grinding of graphite powder under low energy pure shear milling using water as a lubricant. The gentle plastic deformation activated the plastic slip mainly on the easy glide basal planes without any appreciable increase in the density of crystal defects. Consequently, the resulted nano-sheets had relatively well preserved crystal order. A. Touzik et al[102] studied the structural transformations and amorphization of graphite upon high-energy ball-milling under argon and hydrogen atmosphere.Up to approximately 40% of the adsorbed nitrogen was stored in ultra micropores with diameter less than 0.8 nm and it was independent from milling atmosphere. Micro and mesopores with a radius under 4nm were pre- dominantly formed at the early stage of milling. Breakage of graphite stacks increased fraction of larger mesopores. The samples milled under hydrogen atmosphere contained a large fraction of pores with radius about 4 nm even after long milling time. They observed that hydrogen inhibited breakage of graphite sheets, and preserved the crystal structure of graphite and suppressed the specific surface area growth during the milling process. Single-walled carbon nanotube (SWCNT)/silicon composites were produced by J. Y. Eom et al [103] from the purified SWCNTs and Si powder by high-energy ball-milling and then electrochemically inserted with Li using Li/(SWCNT/Si) cells. The SWCNT/Si composites produced by high-energy ball- milling exhibited a structure in which Si particles were densely packed by the flattened and fractured SWCNTs.
3.8. Some Other Nanomaterials and Nano Composites
- Lee et al[104] studied the Phase evolution of Fe2O3 nanoparticle during high energy ball milling. High-energy ball milling of α-Fe2O3 powder was performed in a stainless steel attritor at a speed of 300 rpm for 10–100 h. The powder-to-ball mass ratio was 1:50 with a powder mass of 100 g. Figure 10 shows micrographs of (a) as received powder observed by SEM and (b) 100 h ball-milled Fe2O3 by TEM. Their results revealed that the prolonged milling remarkably reduced particle size from 1 mm in as received powder to about 15 nm in 100 h ball milled powder. T. P. Bobin et al [105] investigated the chemistry and structure of nanosized silicon as-obtained by a high energy ball milling process. They investigated Si/SiOx suspensions on glass surfaces as a function of pH in a variety of oxidizing agents and atmospheres. For pH below 5 and in the absence of H2O, the Si-OEt groups present on the surface of these nanoparticles got protonated and attracted to a less positively charged glass substrate.
4. Synthesis of Nano-quasicrystals by Mechanical Milling
- Several methods have been reported to synthesize quasicrystalline alloys such as solidification of molten alloys, rapid quenching techniques (melt spinning and gas atomisation), mechanical alloying, electro deposition, physical vapour deposition, gas evaporation, electron-beam superficial fusion, electron irradiation, low-temperature annealing of amorphous phases, the high-temperature heat treatment of crystalline intermetallic phases and stacks of pure-element layers[135-145]. Among these, MM has become one of the important techniques for production of metastable/ stable quasicrystalline phase with nano grain size. Much work has been done on producing quasicrystal (QC) by MA in the Al based, Ti based, Zr based and Mg base system[146-151]. It was found that milling of the elemental powder does not always result in quasicrystal phase formation[152,153]. Thus it has been suggested that a sufficient mechanical energy condition during milling experiments should be provided in order to synthesize quasicrystalline phases. Therefore, a combination of post-annealing treatment and milling has been proposed to improve the volume fraction of quasi crystalline phase[154-156]. However, in some report they have shown the direct formation of QC phase by MA [157,158].
4.1. Icosahedral phase
- The formation of icosahedral quasicrystalline phase in a Al, Cu, Fe mixed elemental powders with A165Cu20Fe15 composition has been reported under certain milling conditions and undergoes a structural change during annealing [159]. Several reports have been published on the formation of icosahedral QC phase by MM/MA subsequently annealing in Al-Cu-Fe system[160-169]. These alloys in the MA state contain a β(CsCl_type) solid solution along with icosahedral phase[170]. In a low-energy mill, 40-h milling produces a structure consisting of the icosahedral phase along with the cubic AlFe(Cu) phase and a small amount of Al2Cu intermetallic phase. When milled for 50 h, the intensity ratio of the icosahedral phase to cubic phase decreases considerably as compared to the situation after 40-h milling. In the case of high-energy milling, the evolution and disappearance of the icosahedral phase is faster than in the low-energy milling. Srinivas et al. have proposed that the evolution of the icosahedral phase in mechanical milling takes place by a reaction between the AlFe(Cu) and Al2Cu phases[171]. The ratio of Al to (Cu+Fe) is suggested to play the most important role in the formation of the icosahedral phase during MA in Al–Cu–Fe alloys. However, the ordering of the icosahedral structure into the face-centered type structure is observed only after heat treatment of Al70Cu20Fe10 powders milled for 30 and 40 h[168]. Kim et al. have noticed icosahedral phase development after only 10 h of milling[172]. At temperatures above ~ 475℃, the icosahedral quasicrystalline phase is transformed to the ordered face-centered icosahedral phase. High- or low-energy milling for a sufficient time can raise the temperature to this value. For powders milled for 5, 10 or 20 h, formation of the icosahedral phase and its ordering are observed after further heating. The upper limit for the annealing temperature is 858℃, which is the melting point of the ordered icosahedral structure[170]. Once the Al62.5Cu24.4Fe13 powder mixture, compositionally typical for the icosahedral quasicrystalline phase, has been milled, most of the produced structure consists of Al(Cu, Fe) intermetallics, the quasicrystalline phase being a secondary phase. The initial powder charge has, accordingly, been depleted in aluminium. This variation in chemical composition can be compensated for by selecting a powder mixture rich in aluminium. For Al67Cu22Fe11, the formation of the quasicrystalline phase proceeds directly by mechanical alloying under intense milling conditions. However, the ordered and stable icosahedral phase can then be obtained by annealing treatment at 750–780◦C. The formation of the stable and ordered quasicrystalline structure is suggested to be a result of the ordering of metastable intermetallics[173]. Mechanical activation has been utilised to successfully synthesise the icosahedral phase in Al64.5Cu24.5Fe11alloy. However, although attempts to fabricate the icosahedral phase in an Al63Cu25Fe12 alloy by mechanical alloying have been made, no signs of icosahedral quasicrystal formation have been noticed[170]. Thus, the critical aluminium content for quasicrystalline phase formation in Al–Cu–Fe alloys lies somewhere between 63 and 64.5 wt.%. In some cases additional alloying by a fourth element has been shown to be beneficial for icosahedral phase formation. Recently, synthesis MA Al70Cu20Fe10 icosahedral phase have been investigated by differential thermal analysis and they shown that the short-time milled powders consisting of a series of exothermic effects around 300–500℃, correspond to the formation of crystalline phases[168]. However, the long- time milled powders have not shown any exothermic peak.
4.2. Decagonal Phase
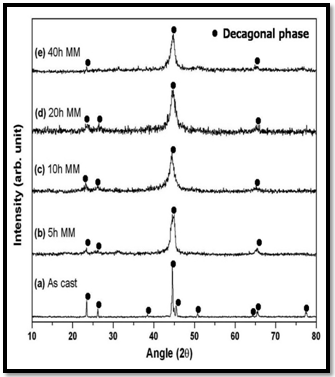
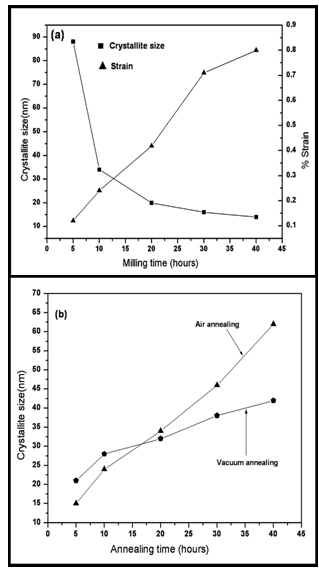
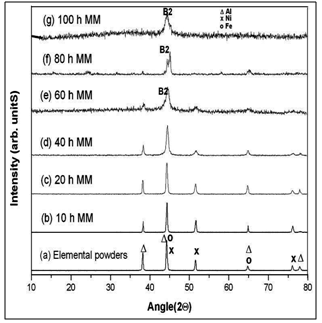
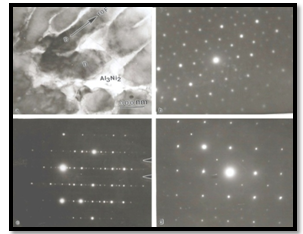
- The decagonal quasicrystal (DQC) phase in the Al-Co-Ni system was prepared by rapid solidification in a compositional range of 9 to 21 at % Co, and 9 to 16 at % Ni by Tsai et al. (1989) [174]. This system is of pronounced scientific and industrial interest because of the mechanical properties of its alloys [Edagawa et al. (1992, 94), Liu et al. (2000),) and Salimon et al. (2001)] [175-178]. Most of the Al-Co-Ni has been grown from the melt and considered as stable phases at least in the high temperature region. Recently, a single-phase decagonal quasicrystal was synthesized by MM and sub- sequent annealing[179]. In this case, MM was plying to enhance the reactivity of elements in subsequent steps. Mechanical activation facilitates the ignition of self- propagation reactions leading to the synthesis of material in relatively short time. Example of this synthesis technique usually referred as mechanically activated self-propagation (MASP) has been reported recently by Singh et al. (2005) [110]. The application of mechanical alloying and self- propagating synthesis for preparation of stable Al-Co-Ni decagonal quasicrystals has been reported by Bokhonov and Korchagin (2004)[179]. In this case, the X-ray and electron microscopic investigations demonstrated the principal possibility to use SHS in combination with preliminary mechanical activation for the synthesis of stable decagonal quasicrystals. The decagonal quasicrystals formed by the SHS process have faceted decaprismatic morphology with a typical length of 300–400 μm and the thickness of 30 μm. In this case the short-term ball milling did not destroy the decagonal structure completely. However, the structure was disturbed significantly, preferentially its periodic component. Order was restored within the first ∼100 h of room temperature ageing. There are indications that recovery of the quasiperiodic order within the planes of tenfold symmetry, which occurred in a second stage, is due to the relaxation of phason strains. Fourier analysis of the diffraction patterns indicated the preferred restoration of bond-lengths corresponding to TM–TM pairs (Kupsch et al. (2002))[180]. The mechanical milling of Al-Co-Ni single phase decagonal QC has been reported by Yadav et al.[181,182]. They were shown the gradual grain refinement with milling time. Fig. 15(a) shows XRD patterns of the decagonal phase obtained from the as cast Al70Co15Ni15 alloys. Fig. 15(b-e) shows the XRD patterns from various milled specimens. In Fig. 15(a), the diffraction pattern shows particularly sharp diffraction peaks and all peaks can be indexed as the decagonal quasicrystal without other phases, which indicated that a high quality of single-phase decagonal quasicrystal is produced from the Al70Co15Ni15 alloy. The XRD patterns exhibit broadening of peaks (Fig. 15(b-e)) belonging to nano decagonal phase. The broadening increases with milling time i.e. 10 to 40h. In the case of 40h milling XRD peaks can still be recognized as due to Al70Co15Ni15 decagonal phase though there is a significant broadening. From the XRD pattern the initially sharp diffraction lines are considerably broadened after ball milling due to a decrease in the particle size and an increase in internal lattice strains. The peak broadening of XRD peaks particularly (102202) peak and creation of particles of sizes ~15nm are features suggestive of creation of nano-decagonal phase. This phase can be considered as a stable phase at this composition. They were calculated particle size (D) as a function of milling time. Fig.16 (a) exhibits variation of Al70Co15Ni15 powder crystallite size at different milling intervals. The powder attained nanocrystallite size at early milling stages, i.e. around ~ 90 nm at 5 h of milling. The crystallite size decreases with milling time up to 40 h of milling. The variation of lattice strain induced by milling based on Voigt function analysis has shown in Fig.16 (a). The strain increases as the milling time increases. Through ball milling, mechanical energy is pumped into the Al70Co15Ni15 matrix. A part of the energy is used in breaking the alloy particles, and another part is used in creating the defects such as dislocations and the distortion of the lattice.In other words, the mechanical energy provided by ball milling gives rise to the decrease in crystallite size and the increase in lattice strain. The effect of the annealing (vacuum and air) on crystallite size of 40 h milled Al70Co15Ni15 DQC alloy have been shown in Fig.16 (b). The variation of crystallite size in both annealing condition are similar up to 20 h, whereas after 20 h, the crystallite size in case of air annealing increases very fast. In the rigorous transmission electron microscopic investigation by obtaining selected area micro diffraction patterns and micro-structural features at different stages have been carried out.
5. Summary and Outlook
- In conclusions, it was suggested that a non-equilibrium processing technique such as mechanical milling with controlled parameters could be used for the synthesis of nano materials, nano-composite and nano-quasicrystalline materials. Mechanical milling can induce defects and activate the frozen state of nano materials configuration and lead to the formation of a stable or metastable composition. We have to understand how to control the parameter so that we can pick up the nano materials, nano-composite and nano- quasicrystalline materials formation and establish the most stable state. The experimental parameters often play some roles to change the composition and consequently may lead to a phase transformation which does not form the equilibrium or stable phase. Therefore, we have to do very controlled experiments without any contamination so that the nano materials, nano-composite and nano-quasicrystalline materials can be achieved. Even after having the mechanically activated state, controlled thermal treatment may be useful to achieve the perfect materials. These investigations are worthy of being pursued. The fomation of nano materials at room temperature by machanical milling is still an open issue which needs to be addressed.
ACKNOWLEDGEMENTS
- The authors would like to record his deep sense of gratitude to Prof. O.N. Srivastava, Prof. N.K.Mukhopadhyay and Prof. R.S.Tiwari, who introduced and inspired me in this fascinating area of mechanical milling and nanomaterials. Authors would like to thank Prof. B.S. Murty and Dr. K. Awasthi for motivating discussions. We are thankful to Dr. V.S. Subrahmanyam for going through the manuscript and offering valuable suggestions.TPY thanks the Department of Science and Technology, India for BOYSCAST Fellowship during which period a part of the work could be completed.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-Text HTML
Full-Text HTML