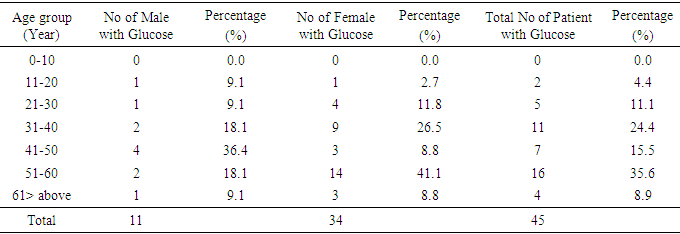-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Journal of Microbiology Research
p-ISSN: 2166-5885 e-ISSN: 2166-5931
2019; 9(1): 1-5
doi:10.5923/j.microbiology.20190901.01

Bacterial Infection of the Urinary Tract and Glucose Level in Patients Presented with Urinary Tract Infection in General Hospital Minna
Sani A. R.1, Hussaini M.1, Baba J.1, Katako G. U.2
1Department of Microbiology, Ibrahim Badamasi Babangida University, Lapai, Nigeria
2Department of Medical Laboratory Science General Hospital, Minna, Nigeria
Correspondence to: Sani A. R., Department of Microbiology, Ibrahim Badamasi Babangida University, Lapai, Nigeria.
| Email: |  |
Copyright © 2019 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

s This study evaluated the incidence of Urinary tract infection (UTI) and Glycosuria in patients attending General Hospital Minna, Niger State Nigeria. Two hundred (200) midstream urine samples and 2ml of blood were obtained from all the patients. Fasting blood sugar level between 150mg/dl to 450mg/dl (8.3mmol/l to 25mmol/l). 73(36.5%) males and 127(63.5%) females were examined out of which 45(22.5%) had diabetes and 155(77.5%) had no diabetics. All the samples were cultured unto freshly prepared Eosine methylene blue (EMB), MacConkey (Difco) and blood agar plates and incubated for 24hours at 37°C. Gram staining and some biochemical tests, antimicrobial sensitivity tests were carried out on isolates using agar diffusion methods. Escherichia coli 21(27.3%) were the most predominant isolate causing UTI, as well as in females 9(27.9%), this is followed by Staphylococcus aureus 19(24.6%)and Pseudomonas specie was the least with a prevalence of 1(1.3%). Klebsiella species, Pseudomonas species and Streptococcus species were recovered from both male and female patients.
Keywords: Bacteria, Glucose, Urinary Tract Infection
Cite this paper: Sani A. R., Hussaini M., Baba J., Katako G. U., Bacterial Infection of the Urinary Tract and Glucose Level in Patients Presented with Urinary Tract Infection in General Hospital Minna, Journal of Microbiology Research, Vol. 9 No. 1, 2019, pp. 1-5. doi: 10.5923/j.microbiology.20190901.01.
Article Outline
1. Introductions
- The kidneys remove liquids waste from the blood in the form of urine, keep a stable balance of salts and other substances in the blood and produce a hormone that aids the formation of red blood cells (RBC). Narrow tubes called ureters carry urine from the kidneys to the bladder a triangle shaped chamber in the lower abdomen. Urine is stored in the bladder and emptied through the urethra (Ahmed & Avasara, 2008). The average adult passes about a quart and half of urine each day the amount of urine varies depending on the fluids and foods a person consumes. The volume formed at night is about half that formed in the daytime (NIDDK, 1997). Malnutrition, poor hygiene, low socio-economic status are associated with urinary tract infections and these factors are rife in rural settings (Ahmed & Avasara, 2008). Sugar in the urine promotes bacterial growth which leads to the development of “lazy bladder” that enhanced bacteria proliferations. The most common pathogens isolated from urine of diabetic patients with UTI are Escherichia coli, other Enterobacteriaceae such as Klebsiella species, Proteus species, Enterobacter species and Enterococci (Geerlings, Wu, et al., 2002). Patients with diabetes are more prone to have resistant pathogens as the cause of their UTI, including extended-spectrum β-lactamase-positive Enterobacteriaceae (Inns et al., 2014), (Colodner, et al., 2004) and fluoroquinolone-resistant uropathogens (Wu, et al., 2014) carbapenem-resistant Enterobacteriaceae, (Schechner, et al., 2013) and vancomycin-resistant Enterococci. (Papadimitriou-Olivgeris, et al., 2014) This might be due to several factors, including multiple courses of antibiotic therapy that are administered to these patients, frequently for asymptomatic or only mildly symptomatic UTI, and increased incidence of hospital-acquired and catheter-associated UTI, which are both associated with resistant pathogens. Type 2 diabetes is also a risk factor for fungal UTI (Sobel, et al., 2011). Escherichia coli has been reported as the predominant isolate causing urinary tract infection, few authors have reported changing patterns in the prevalence of uropathogens (Omoregie & Avasana, 2008). Urinary pathogens especially from community patients have been known to include strains that are resistant to many of the commonly used antibiotics (Omoregie et al., 2008).One woman in five develops a UTI during her life time. UTIs in men are not so common but they can be very serious when they do occur (Grabe et al., 2009). UTI is a common problem in the elderly diagnosis, prevention and treatment can often be complex because clinical manifestations can be typical and host defense diminishes with age (Grabe et al., 2009).The urinary tract is divided into two major divisions. The upper UTI (the kidneys, renal pelvis and urethras) and the lower UT (urinary bladder and urethra). UTIs are serious health problems, if it affects the lower urinary tract, it is known as a bladder infection (cystitis) and when it affects the upper urinary tract, it is known as kidney infection (pyelonephritis) (Lane & Takhar, 2011). UTI occurrence is commonly related to social class, age of patients, birth rank or size of family. UTI affects people in varying incidences depending on age group and gender. Most times boys are at greater risk before the age of 3 months but girls become at greater risk thereafter (CDC, 2015). Studies have shown that 3% of pre pubertal girls and 1% of pre pubertal boys are diagnosed with a UTI (Kolawole et al., 2009 and Prakasam et al., 2012).Patients with urogenital anomalies, over distention of the urinary bladder from out flow obstruction or neurogenic malfunctions or women with over distention of the uterus during pregnancy are particularly susceptible to ascending UTI, of the renal pelvis (pyelitis) and chronic pyelonephritis are the most common complication.Upper UTI may result from hematogenous spread of bacteria in to the glomeruli and renal cortex in patients with septicemia (Grabe et al., 2009).Glycosuria is defined as a concentration of urinary glucose detectable using relatively intensive but specific screening test (Gidabayda et al., 2017).Glycosuria occurs only when the plasma and therefore glomerular filtrate, concentration exceed the tubular restorative capacity. This may be because: -Ø The plasma and glomerular filtrate concentration are more than about I1 mmol/c and therefore tubular restorative capacity is significantly exceeded.Ø The tubular restorative capacity is reduced as for example during pregnancy so that glycosuria occurs at lower filtrate concentration (renal glycosuria) usually harmless and glycosuria may be due to Glucose infusion.Ø Renal glycosuria which may be divided as an autosomal dominant trait and pregnancy. (National Kidney and Urologic Disease Centre, USA, 2003).Autosomal DNA is a term used in genetic genealogy to describe DNA which is inherited from the autosomal chromosomes. This is a chromosome that is different from the sex chromosomes (Griffiths et al., 2007).
2. Materials and Method
2.1. Materials
- Materials used includes, Urine containers, Register, Pen, Incubator, Hot Air Oven, Bunsen burner, Conical flask, measuring cylinder, Gram staining reagents, media, petri-dishes, Autoclave, Microscope, Slides, Combi-9, Glucometer with the strips, Lancets, swabs, cotton wool and Kidney plate.
2.2. Ethical Considerations
- Permission to conduct this study was sought from Ethics and Publication Committees of the Hospital. All the information obtained from the participants was treated confidentially. The results were used in the management of patients since the research is hospital based.
2.3. Sample Collections
- Two hundred early morning, mid-stream urine samples was collected from one hundred patients presenting with urinary tract infection, age between 1-70 years and above were studied for the presence of bacterial species and 2mls of blood was obtained for glucose screening (glycosuria).
2.4. Samples Processing
- Sterilized universal (glass) containers were used in collecting early morning midstream urine from patients attending General Hospital Minna (GHMX), subjects were instructed to void 5mls of Mid-Stream urine. The containers were labeled with the following data.(i) Age(ii) Sex
2.5. Sample Processing
2.5.1. Testing for Glucose (Blood Samples)
- A drop of blood was placed on an electronic device for measuring the blood glucose level called Blood glucose meter and the results was documented immediately.
2.6. Processing Urine Samples
2.6.1. Macroscopic Examinations
- The colour, and appearance of the urine specimens were noted, whether the specimen appears clear or turbid.
2.6.2. Microscopic Examination of Centrifuged Urine
- A drop of well mixed centrifuged, unstained urine was placed on a microscopic slide, and covered gently with cover slit and examined under x 10 and x 40 objectives of the microscope for bacterial cells, yeast, erythrocytes etc.
2.7. Culturing of Urine
- Samples were aseptically inoculated on MacConkey Cled and blood agar and incubated aerobically at 37°C for 24 hours after which the plates were read for the presence of bacteria.
2.7.1. Gram Stain Procedures
- A loop full of the pure colonies growth from nutrient agar was placed on a glass slide and spread to make a thin smear and allowed to air dry, fixed with gentle heat and placed on a staining rock. The smear was flooded with 0.5 percent crystal violet solution for 60 seconds. The crystal violet was replaced with Lugol's iodine and allowed to act for 60 seconds and rinsed in water. The smear was then colonized rapidly with acetone until no purple colour was produced and then counter stained with neutral red for 1 minute, rinsed with water and allowed to dry. The slide was then examined microscopically with x 100 objective. Both gram positive and negative bacteria were subjected to biochemical test which gave different type of isolates.
3. Statistical Analysis
- The objective of the statistic is to find the correlation coefficient between bacterial population and the glycosuria level of the subjects. That is using the person 'r' coefficient correlation coefficient is a popular index of the degree of association continuous variable. This help to test whether two observed estimates differ significantly or are similar to each other (Grabe et al., 2009).
4. Results
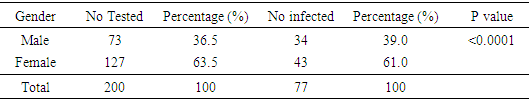
4.1. Prevalence of Patients Screened for Urinary Tract Infection Based on Gender
- A total of 77 (38.5%) out of 200 patients had urinary tract infection. Female gender was a significant risk factor for acquiring UTI with 43(55.8%) compared to the male with 34(44.2%) and the value of P < 0.0001. Table 1, revealed the prevalence of patients screened for UTI with the female been the highest 127(63.5%) while the male had 73(36.5%).
|
|
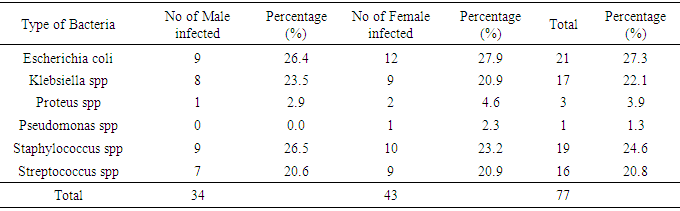
4.2. Prevalence of Urinary Bacteria
- Escherichia coli 21(27.3%) were the most predominant isolated bacteria causing UTI, as well as in females 9(27.9%) (Tables 3 and 4). This is followed by Staphylococcus aureus 19(24.6%) and Pseudomonas specie was the least with a prevalence of 1(1.3%). Klebsiella species, Pseudomonas species and Streptococcus species were recovered from both male and female patients.
|
|
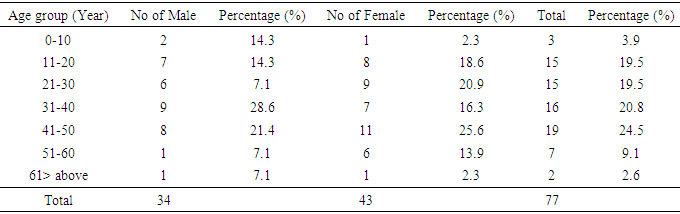
4.3. Prevalence of Patients with Glycosuria Based on Age Group
- The prevalence of patients with glucose was 45(22.5%). This revealed that the highest prevalence is within the ages of 51-60 years which is illustrated in Table 5. Female of the age group 51 to 60 had the highest prevalence with 14(41.1%) while the male counterpart had 2(18.1%).
|
|
5. Discussion
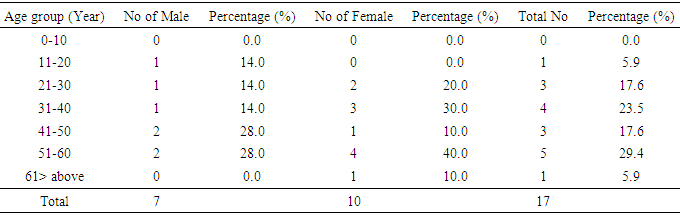
- This study has provided the basic data concerning the occurrence of urinary tract infection due to bacteria and its relationship with glucose. All in all only 77(%) patients were positive for bacteria and 45 (%) out of the 200 samples were glucose positive. Among the 77(%) positive bacteria only 17(%) were positive for glucose. In the statistic carried out on the correlation of bacterial population and glucose level, it shoes that there is no relationship between them, presence of glucose does not really indicates the presence of bacteria. This work is similar to that a Omoregie and Eghafona 2009, who stated that not all may result to bacteria and "r" is less than stated table value. The study is also similar to the work of Bankole et al., 2011 who revealed that E.coli is the highest isolated bacteria and female have the highest bacterial isolate than the male counterpart.
6. Conclusions and Recommendations
6.1. Conclusions
- High prevalence of Urinary tract infection found in this study revealed that female are at risk of contracting the disease than the male counterpart and E. coli is the predominantly isolated bacterial infection despite several studies that have been done As a result, urine stays in the bladder too long and becomes a breeding ground for bacteria.
6.2. Recommendations
- Undetected, untreated, asymptomatic bacteria can lead to pyelonephritis. Right treatment should be administered with suitable antimicrobial chemotherapy to those infected. Health education on how to prevent UTI should form part of government programs. From this and other studies it is been established that, there are intestinal bacteria that causes contamination of the urinary opening, also washing of hands with soap and rinse with water before and after using the toilet can reduce the risk of UTI.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML