Rajani Singh
Department of Anatomy Uttar Pradesh University of Medical Sciences Safai, Etawah UP, India
Correspondence to: Rajani Singh, Department of Anatomy Uttar Pradesh University of Medical Sciences Safai, Etawah UP, India.
| Email: |  |
Copyright © 2020 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
Present study aims at uncovering the secrets of internal morphology of femoral nerves and its branches, cutaneous trunk, subcutaneous trunks, saphenous, medial cutaneous and intermedius cutaneous nerves innervating the skin of anteromedial thigh at fascicular level. Therefore, the aim of the study is to track, correlate, interpret and identify the pathways of fascicles and construct 3D schematic models of these nerves and trunks. These trunks and nerves of a cadaver were processed for histological slides staining with haematoxylin and eosin. The fascicles in the slides were identified/tracked/correlated and interpreted from cranial most slide to the last terminal slides of these nerves and trunks. The correlation data was mapped resulting in 3D models of nerves/trunks showing identified pathways of nerves/fascicles by computer aided design. These 3D models revealed the pattern of identified continuous, consistent and traceable pathways of fascicles of cutaneous trunk, subcutaneous trunks, saphenous, medial cutaneous and intermedius cutaneous nerves interrupted by split, fusion and multiplexing calibrated in distances from inguinal ligament. Femoral nerve/fascicles/nerve fibres are damaged impairing sensation of skin of anteromedial thigh creating helm of neurological complications. Hence these are to be repaired with least invasion. These findings will be of paramount importance for intraoperative stimulation to diagnose and identify the fascicle and for microneurosurgical manipulations/ neurotisation in cutaneous trunk, subcutaneous trunks, saphenous, medial cutaneous and intermedius cutaneous nerves, branches of thigh. This study is of utmost importance to diagnose and repair/graft/regenerate these injured elements at fascicular level.
Keywords:
Cutaneous nerves, Microanatomy, Transformational process, Pathways of fascicles
Cite this paper: Rajani Singh, Microanatomy and Clinical Significance of Cutaneous Nerves of Thigh, Basic Sciences of Medicine , Vol. 9 No. 2, 2020, pp. 15-23. doi: 10.5923/j.medicine.20200902.01.
1. Introduction
Variant configuration of branching patterns of femoral nerve (FN) and its classification have been described in literature. The cadaver which was selected for cropping femoral nerve surprisingly possessed the Type-II configuration of femoral branching pattern classified by Singh et al [1]. In this configuration, the FN bifurcates into muscular and cutaneous trunks one centimetre below the inguinal ligament. The cutaneous trunk further splits into sub-cutaneous trunk of thigh and the saphenous (S) nerve. The subcutaneous trunk, then, bifurcates into intermediate cutaneous (IC) and medial cutaneous (MC) nerves of the thigh. The group of afferent fascicles of S, IC and MC supplying skin of anteromedial thigh and specialised receptors collecting cutaneous sensitivity of skin may be defined as cutaneous sensory system of anteromedial thigh. These afferent fascicles collects the information from cutaneous area of anteromedial thigh which the central nervous system uses to monitor and control cutaneous sensitivity of skin by elegant feedback of afferent mechanisms to ensure that a planned action or movement is executed through efferent and feed forward [2]. Cutaneous innervation of skin consists mainly of unmyelinated fibres, accounting for around 90% of all dermal nerve fibres [3].Though the injuries to the IC and MC nerves have hardly been reported as there is no literature available on the clinical complications of these nerves yet few cases of pain and paraesthesia over the anterior and medial aspects of the thigh as a result of engagement of IC and MC nerves of the thigh are described. However, sensory loss on the medial side of the thigh, leg and foot up to the ball of the great toe because of engagement of the saphenous nerve through iatrogenic lapses or otherwise are also reported. The outcome of injuries may not be fatal or produce unbearably serious signs and symptoms so the patients may not be opting for costly neurosurgical diagnosis (MRI) for detection of location and degree of injury and procedures. The neuro-therapy of neuropathological morbidity requires accurate diagnosis and treatment. There is very limited scope of investigating location and identification of injured fascicle or nerve fibres under current knowledge of internal morphology of nerve. Though MR advanced neurography claims to detect the location and degree of injury [4] yet it has its own limitations regarding resolution and image defects. Therefore, a micro-anatomic study has been planned and designed to bring out the 3D picture of fascicular pathways by tracking fascicles in histological slides of S, IC and MC for improving identification of injured fascicle, its location and degree of injury for diagnosis and imagery interpretation together with neurosurgical repair/grafting/regeneration of injured nerve fibres.
2. Material and Methods
The right femoral nerve was cropped from 70 year old female cadaver in the department of Anatomy AIIMS Rishikesh India. The femoral nerve was exposed cranially from its emergence at the lateral border of psoas major muscle caudally to its branches S, MC and IC. The FN was cut into 24 pieces from the cranial most point to its point of bifurcation into cutaneous and muscular trunks. 3 cm long Cutaneous trunk then was cut into 6 (C1, C2, C3, C4, C5 and C6), S nerve into 6 (S1, S2, S3, S4, S5 and S6), subcutaneous trunk into 3 (SCT1, SCT2 and SCT3), MC into 1 (MC1) and IC into 2 (IC1 and IC2) (Fig. 1) pieces of 0.5 cm each amounting to 18 pieces. These pieces were converted into blocks by embedding the tissue in paraffin wax. 3 sets of slides at an interval of 1mm each were prepared from each block. Each set consisted of 3-8 slides of 5 micron thickness each which were cut by microtome. Each of the slide was stained with haematoxylin and eosin. These slides were photographed by 16 mega pixel Sony camera under a high resolution microscope of model PZRM-26 having software “Future Winjoe”. | Figure 1. Showing cutaneous nerves/trunks and their block plan considered for study |
All the distance given in numbers are in cms. A23B/A24B are the bottom of 23rd and 24th block. Red line is the position of inguinal ligament (IL). M0T/C1T=Common Tops of 0th block of muscular and 1st block of cutaneous trunk; C6B/S1T /SCT1T=Bottom of 6th block of cutaneous trunk/Top of 1st block of saphenous nerve/Top of 1st block of subcutaneous trunk; SCT2T/SCT2B=Top and bottom of 2nd block of subcutaneous trunk; SCT3T/SCT3B= Top and bottom of 3rd block of subcutaneous trunk; MC1 and IC1are first blocks of medial cutaneous and intermedius cutaneous nerves. Reference of Measurements:All the lengths of FN, cutaneous trunk, subcutaneous trunk and their branches, S, MC and IC nerves were measured with reference to inguinal ligament together with distances of point of bifurcation of FN into muscular and cutaneous trunks, point of bifurcation of cutaneous trunk into S nerve and subcutaneous trunk and point of bifurcation of subcutaneous trunk into IC and MC. The inguinal ligament has been taken as origin or reference point. The position of all the slides have also been computed. All the distances above inguinal ligament has been considered positive and below it negative as per convention.Slide plan: As the fascicular configuration in proximal FN is more complex because of presence of more transformational processes than distal FN so continuous sampling of blocks at 1-2 mm interval between sets of slides has been kept whereas in distal part, the blocks were cut at intervals of 10-80 mm. Consequently the inter-block slide interval has been kept 10-80 mm while intra-block slide interval has been consistently planned at 1-2 mm (Fig. 2). Detailed table reveals the complete planning of blocks and slides of cutaneous trunk (Table 1), saphenous nerve (Table 2), sub cutaneous trunk (Table 3), medial cutaneous and intermedius cutaneous nerves (Table 4).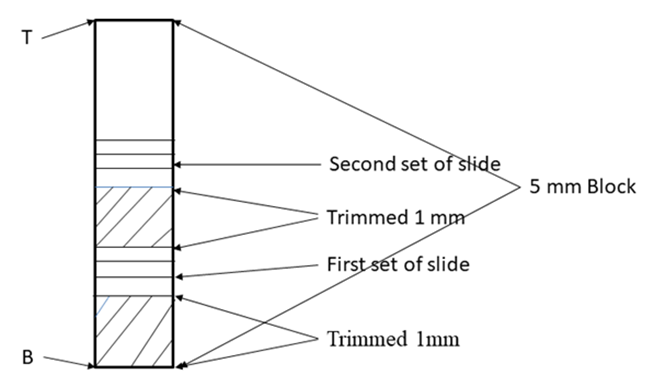 | Figure 2. Showing typical slide plan from typical block |
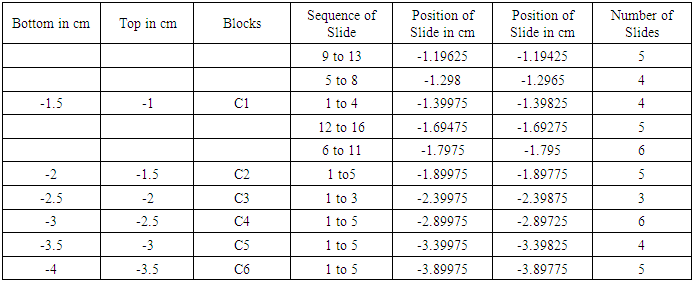
Table 1. Showing block and slide plan of Cutaneous Trunk
 |
| |
|
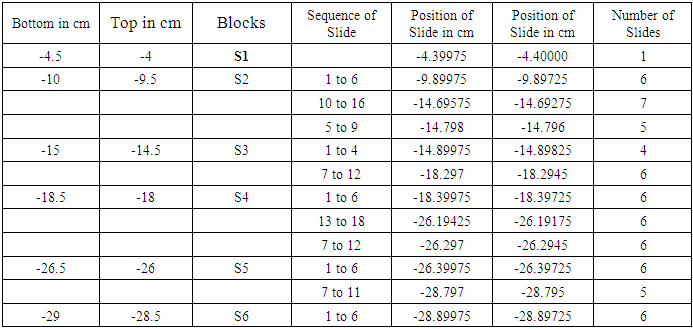
Table 2. Showing block and slide plan of saphenous nerve
 |
| |
|
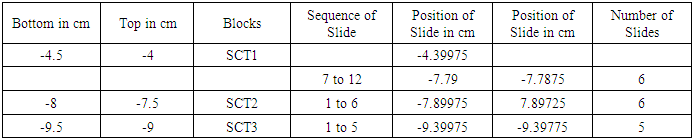
Table 3. Showing block and slide plan of subcutaneous trunk (SCT)
 |
| |
|
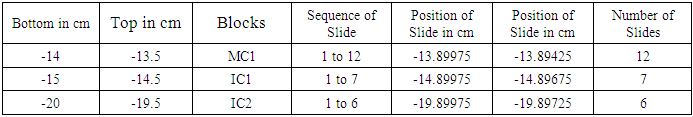
Table 4. Showing block and slide plan of Medial Cutaneous (MC) and Intermedius cutaneous (IC) nerves
 |
| |
|
It is a typical nerve block of 5 mm. T is top and B is bottom of block. The block was trimmed by 1 mm from the bottom shown by oblique lines. Then 1st set of 3-7 slides was cut. Again 1 mm of tissue was trimmed and 2nd set of slides was cut. And so on. Each block was processed for 15/18 slides.First column presents the bottom and second top of blocks e.g. -1.5 cm is bottom and -1 cm is top of C1 below inguinal ligament. Column third shows the blocks (C1-C6). Fourth column presents set wise sequential number of slides. Fifth and sixth column present the computed distances in the order of slides e.g. in fourth row, -1.19625 cm corresponds to 9th slide and -1.19425 cm to 13th slide shown in 4th column. 7th column shows tot nuber of slide in a set.First column presents the bottom and second top of blocks e.g. -10.0 cm is bottom and -9.5 cm is top of S2 below inguinal ligament. Column third shows the blocks (S1-S6). Fourth column presents set wise sequential number of slides. Fifth and sixth column present the computed distances in the order of slides e.g. in third row, -9.89975 cm corresponds to 1st slide and -9.89725 cm to 6th slide shown in 4th column. 7th column shows total number of slide in a set.First column presents the bottom and second top of blocks e.g. -8.0 cm is bottom and -7.5 cm is top of SCT2 block below inguinal ligament. Column third shows the blocks (SCT1-SCT3). Fourth column presents set wise sequential number of slides. Fifth and sixth column present the computed distances in the order of slides e.g. in fourth row, -7.89975 cm corresponds to 1st slide and -7.89725 cm to 6th slide shown in 4th column. 7th column shows total number of slide in a set.First column presents the bottom and second top of blocks e.g. -14.0 cm is bottom and -13.5 cm is top of MC1 below inguinal ligament. Column third shows the blocks (MC1, IC1-IC2). Fourth column presents set wise sequential number of slides. Fifth and sixth column present the computed distances in the order of slides e.g. in second row, -13.89975 cm corresponds to 1st slide and -13.89425 cm to 12th slide shown in 4th column. 7th column shows total number of slide in a set.142 slides were then processed and stained with haematoxylin and eosin from caudal part of femoral nerve below inguinal ligament. The correlation and tracking of fascicles in a total of (486+190) slides including 344 slides in 24 blocks of cranial FN and 142 slides in distal part of femoral nerve from cutaneous trunk, saphenous nerve, subcutaneous trunk, medial cutaneous and intermedius cutaneous nerves. It was a tedious and voluminous work therefore, the study was divided into four parts:1). The fascicular configuration pattern of cranial FN together with its branches, nerves to iliacus was brought out in this part of study. Here fascicular correlation was carried out from blocks A1 to A11 considering only those fascicles which partly or fully contributed to nerves to iliacus. Primarily, all the 21 fascicles were named in slide A1 8 to start the correlation with identification of fascicles in entire pathways [5].2). In the 2nd part of study, though all the fascicles were tracked further starting from the block A12 to A24 yet only the fascicles contributing partly or fully to nerves to sartorius and pectineus were correlated [6] in this study. 3). The femoral nerve bifurcated into cutaneous and muscular trunks after A24 block. But only group of fascicles pertaining to cutaneous nerves namely S, MC and IC emanating from cutaneous trunk were identified, correlated and tracked extending their pathways cranially up to the slide A1 8 and vice versa in this part of study. This part of study constituted present study. 4). In the fourth part of study, the fascicles belonging to muscular nerves will be correlated and tracked extending their correlation cranially to the slide A1 8 in future (in process). For identification of fascicles and getting rid of duplicity and confusion, a naming scheme has been designed as under-Naming scheme of fascicles:For deciphering the fascicles of individual nerves and to avoid confusing duplicate numbers, the name of the fascicles at the point of transformational processes was changed in sequential order [6] with prefix from CF of fascicles (FN) to CCF in cutaneous trunk and SCCFs in subcutaneous trunk extending it to SCF in S nerve, MCCFs and ICCFs in MC and IC respectively. As long as the CF was found consistent and continuous during correlation in different sections in cranial to caudal direction, the same number was retained but when any CF underwent transformational processes, a sequential new unique identity number/s was given to new CF so formed [6].Correlation of slides and construction of 3D schematic Model of nerve: The correlation and tracking of fascicles was carried out caudally by comparing the fascicles on the basis of correlation parameters [6] in consecutive slides. Further, the fascicles of branches/trunks were identified at the point of emanation/bifurcation. Now, these identified fascicles were grouped and tracked cranially to cranial most slide A1 8 for deciphering the pathways.Modelling of pathways and configuration of fascicles: 3D schematic models of subcutaneous trunks, S, MC and IC nerves consisting of pathways and configuration of fascicles were planned to be constructed by considering only those slides having transformational processes showing discontinuities in fascicular pathways and slide intervals between which the fascicles were traceable, consistent and continuous were squeezed to one slides whose two faces were representing both the terminal slides. This was done using Corel draw 16 software.
3. Results
The compound FN bifurcated into cutaneous and muscular trunk at -1.0 cm after A24 1. The 3 cm cutaneous trunk advanced further downwards and bifurcated into subcutaneous trunk and S nerve at -4 cm after C6 1. After this, 5.5 cm long subcutaneous trunk was divided into MC and IC nerves at -9.5 cm after SCT3 1 (Fig. 1).CorrelationAll the CFs were correlated and tracked in slides cranially from A1 8 (8.6035 cm) to caudally up to the terminal slides S6 1 (-28.89975 cm), MC1 1 (-13.89975 cm) and IC2 1 (-19.89975 cm) passing through branching/bifurcating points and undergoing numerous transformational processes defined as splits, fusions and multiplexings [6].These slides and points of bifurcation/branching are calibrated in distance from inguinal ligament. CFs, 1-21 having been correlated from A1 8 to A24 1, transformed into CFs {(303, 304); (280, 257, 270, 312, 313): (316, 317, 318)} in A24 1. The CFs {(303, 304); (280, 257, 270, 312, 313): (316, 317, 318)} were observed surrounded by internal epineurium bifurcated into muscular and cutaneous trunks consisting CFs 316, 317, 318 and {(303, 304); (280, 257, 270, 312, 313)} respectively (Fig. 3). In the present study, we are correlating only sensory fascicles emerging from cutaneous trunk so correlation of CFs in cutaneous trunk was extended further as described below.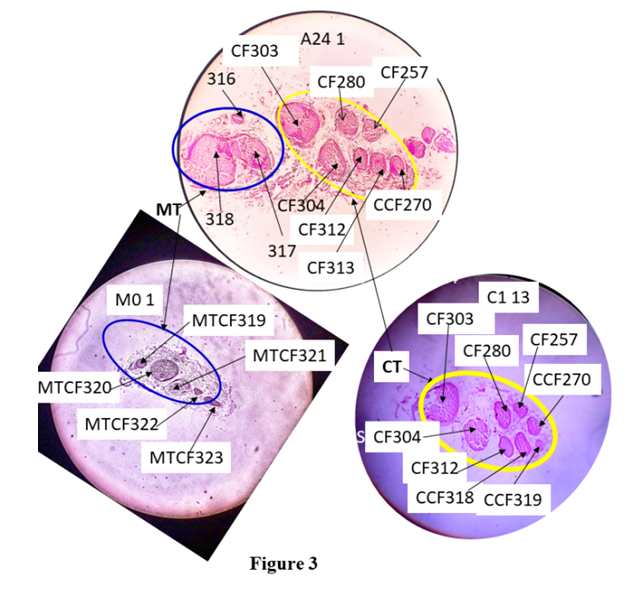 | Figure 3. Correlation between slides, A24 1 and C1 13 |
The femoral nerve bifurcates into muscular trunk (MT enveloped in blue curve) and cutaneous trunk (CT enveloped in yellow curve) after the slide, A24 1. CF313 in A24 1split into CCFs 318 and 319 in C1 13. MTCF= CFs in muscular trunk, M01= 1st set of 0tt block of muscular trunk.Correlation of CFs in cutaneous trunk:The CFs, having been correlated from A1 8 to the point of bifurcation of cutaneous and muscular trunks, transformed into CFs, (303,304); (280,257,270,312,313) in A24 1. Then the CFs, (303,304); (280,257,270,312,313) were identified and confirmed forming cutaneous trunk in its cranial most slide, C1 13 at the point of bifurcation. These CFs were grouped as fascicles of cutaneous trunk. Then these grouped fascicles of cutaneous trunk were tracked back to A1 8, the cranial most slide and found to correspond to CF7*, 17, 6, 9*. Here the CFs shown with stars (CFs 7*, 9*) were partially contributing to cutaneous trunk whereas CFs without star (6, 17) were fully contributing to it. Then the CFs, {(303,304); (280,257,270,312,313)} emerged out as cutaneous trunk as observed in C1 13 slide. The CF313 in A24 1 split into CCFs 318 and 319 in C1 13. Then these CFs or CCFs were traceable from C1 13 to C2 12. CF312 and CCF318 fused into CCF320 in C2 12 undergoing shape change and CF280 in C2 12 split into CCF321, 322 and 323 in C2 11. C2 11 is traceable to C2 1 except shape changes. Then, CCF321, 322 and 257 in C2 1 fused forming CCF324 in C3 3. CF270 and CCF324 in C3 1 fused forming CCF325 in C4 4. CCF325 in C4 1 split into CCF326 and 327 in C5 5. C5 5 is traceable up to C6 1. After C6 1, cutaneous trunk bifurcated (Fig. 4) into a) S nerve containing CCFs 319, 320, 323, 326 and 327 in S1 1 and b) sub-cutaneous trunk consisting of CFs 304, 305 and 306 in SCT1 1.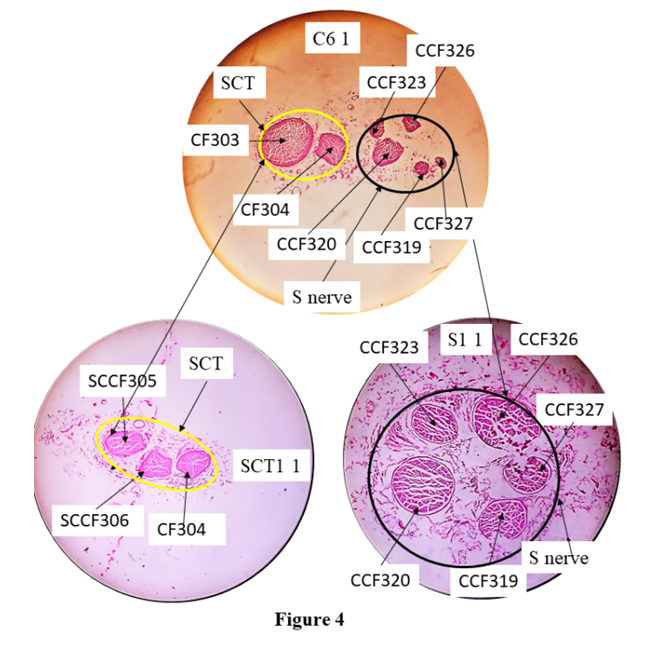 | Figure 4. Correlation of slide, C6 1 with S1 1 and SC1 1 |
C6 1, the caudal most slide of cutaneous trunk consisting of fascicles bifurcating into subcutaneous trunk (yellow) in SC1 1 and saphenous nerve (black) in S1 1. CF303 split into SCCFs, 305 and 306. SCT- subcutaneous trunk, S nerve- saphenous nerve.a). Correlation of fascicles in S nerve:The CCFs, 319, 320, 323, 326 and 327 were identified in S nerve at the point of emergence and were grouped together and correlated back cranially until A1 8. These fascicles were found corresponding to CFs 7*+9*+17* in A1 8. So the nerve fibres from CFs 7*+9*+17* were partially contributing to S nerve in A1 8 slide.The S nerve having CCFs 319, 320, 323, 326 and 327 in S1 1 emerged out after C6 1. These CCFs were traceable from the slides of block S1 through S2 1. The CCFs 319 and 320 in S2 1 fused together forming SCF 328 in S3 13. SCF328 in S3 1 split into SCFs 329 and 330 in S4 12. CCF330 emerged out as branch of S nerve after S4 1. CCFs323 split into SCFs331 and 332, CCFs326 into SCF333 and 334, CCF329 into SCF335 and 336 in S4 1 respectively. The CCF327, SCFs 331, 332, 333, 334, 335 and 336 are traceable up to S5 1 except change of position due to migration and minor shape changes. These SCFs reorganize their position through migration and rearrange in S6 11 from S5 1. These fascicles got separated laterally into infrapatellar branch having SCFs (331, 332, 335, 336) and main S nerve having SCFs (327, 333, 334) in S6 11 (Fig. 5). The fascicles are traceable between S6 11 and S6 7. Again reorganization of fascicles is taking place from S6 7 to S6 6. The infrapatellar physically emanated laterally from S nerve after S6 1. The main S nerve goes caudally. It is pertinent to mention that CFs which are undergoing transformational processes have been cited in above results.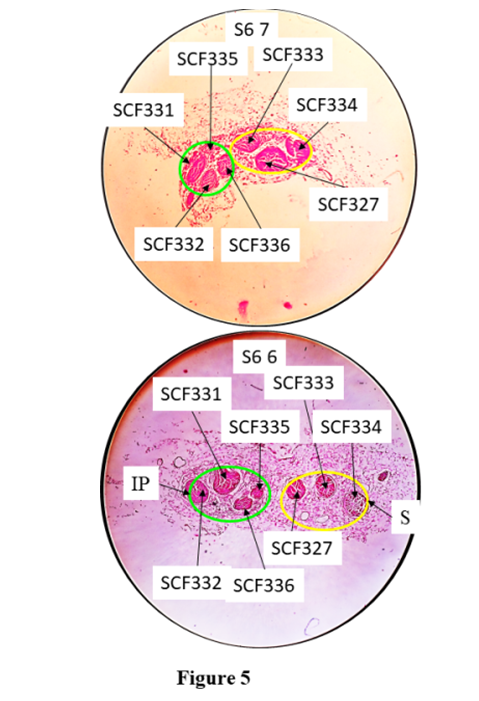 | Figure 5. Correlation of slides, S6 7 and S6 6 |
SCF 335 in S6 7 split into SCF338 and 339 in S6 6. The CFs changed their location and orientation. Then saphenous nerve gave infrapatellar (IP Green) branch and saphenous (yellow) proceeded downward for innervation. S= saphenous nerve.b). Correlation of fascicles in sub-cutaneous trunk:The CFs, 303 and 304 corresponding to sub-cutaneous trunk at the point of separation were identified, grouped and correlated back to A1 8. These CFs were found to be contributed by nerve fibres from 6+17*+7*. Here CFs 7 and 17 were partially contributing while the CF 6 was fully contributing to this trunk.The CFs 303 and 304 constituted subcutaneous trunk which was separated laterally from cutaneous trunk after C6 1. In the sub-cutaneous trunk, the CFs 303 and 304 in slide C6 1 of which CF303 split into SCCF 305 and 306 in SCT1 1 slide. After the slide SCT1 1, the CFs 304, 305 and 306 were traceable from SCT1 1 up to SCT3 1. As the fascicles were identified at the point of bifurcation of sub-cutaneous trunk into MC and IC nerves so these CFs were grouped again corresponding to MC and IC nerves and then tracked back up to A1 8. The nerve fibres from CFs17*+7*were partially contributing to MC and from 6 was fully contributing to IC. The sub-cutaneous trunk bifurcated into IC and MC nerves after SCT3 1 slide. The CF304 constituting MC split into MCCF307, 308, 309, 310 in MC1 3 slide. The SCCF305 split into ICCF307, 308, 309 and SCCF306 into ICCF310, 311 in IC1 1 respectively (Fig. 6). The CFs of these nerves further split in smaller/tiny CFs in IC2 1 ready for innervation. 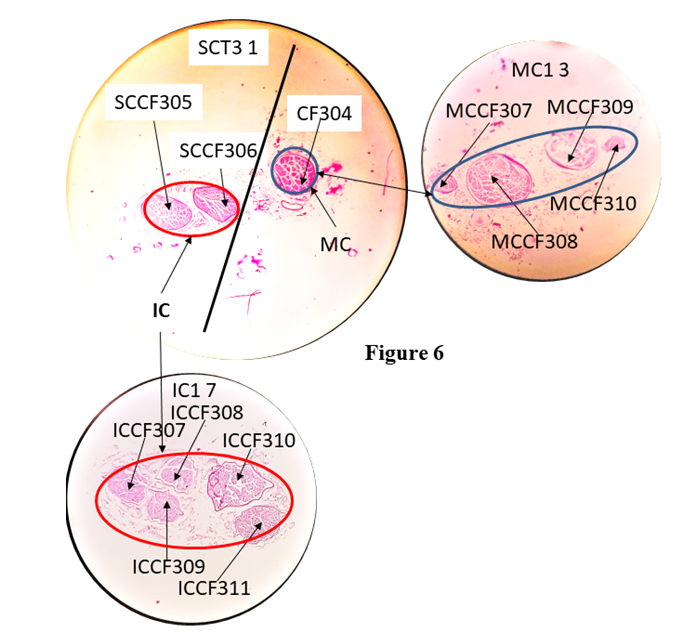 | Figure 6. Correlation of slides, SC3 1 with IC1 1 continuing to IC2 1 and MC1 3 |
The subcutaneous trunk bifurcated into MC (blue) and IC (red) after SC3 1. CF304 in SC3 1 split into MCCF307, 308, 309 and 310 forming MC in MC1 3. SCCF305 and SCCF306 in SC3 1 split into ICCF307, 308, 309 and ICCF310 and 311 in IC1 7.3D Schematic models: The fascicular and group correlation data of pathways and configuration of identified fascicles have been used to map the trunks/nerves and presented as 3D schematic models of cutaneous trunk, S nerve and subcutaneous together with MC and IC nerves (Fig. 7A-7C). The emergence of branches if any were also shown in the models. Thus the models bring out comprehensible and simple 3D picture of identified pathways of fascicle. All the slides have been calibrated for distances and so the CFs were not only identified at all the distance levels but also their straight undisturbed path could also be computed.Model showing fascicles in cutaneous trunk (Fig. 7A): As cutaneous trunk separates out after A24 1 so the model of cutaneous trunk consists of the 7 slides namely, A 24 1, C1 13/C2 12, C2 11/C2 1, C3 3/C3 1, C4 6/C4 1, C5 5/C5 1, C6 5/C6 1 from cranial to caudal direction. The top slide of this diagram is A24 1 which possesses the CFs, 303,304; 280, 257, 270, 312, 313 corresponding to cutaneous trunk which is ready to emerge. The next slide represents two slides. C1 13 on the upper face and C2 12 at lower face as the fascicles present in C1 13 are traceable continuous and consistent up to slide C2 12. After passing through 5 transformational processes, the CCFs of cutaneous trunk were found to be surrounded by internal epineuriums in two groups in slide C5 5 in the process of formation of S nerve and subcutaneous trunk. These fascicles were bifurcated into subcutaneous trunk {CFs (303, 304)} and S nerve {CCFs (319, 320, 323, 326, 327)} after the slide C6 1.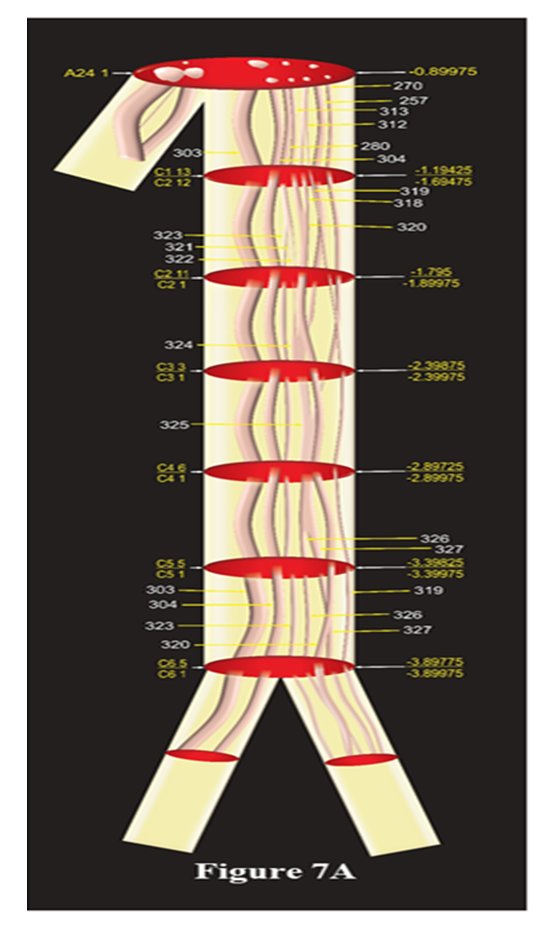 | Figure 7A. The Schematic model of Cutaneous Trunk |
This model presents correlated, identified and tracked fascicular pathways of cutaneous trunk consisting of CFs 303, 304, 257, 270, 280, 312, 313 at the cranial end which are transformed as CFs 303, 304; CCFs 319. 320, 323, 326, 327 at caudal end in C6 1 ready to emerge as subcutaneous trunk and saphenous nerve after this slide. All the slides presented as C1 13/C2 12 represent the continuity, consistency and traceability of all the group of fascicles between C1 13 and C2 12. The upper face of slide, C1 13 is matching with A24 1 whereas after lower face C2 12, the fascicles undergo transformational process (split, fusion or multiplexing). The position of all the slides has also been computed and labelled to locate the discontinuity in straight, consistent and traceable path of fascicles.Model showing fascicles in S nerve (Fig. 7B):S nerve, emerging out of cutaneous trunk medially after C6 1, possessed CCFs, 319, 320, 323, 326, 327. This model is constructed involving C6 1, S1 1/S2 1, S3 16/S3 1, S4 12/S4 1, S5 18/S5 1, S6 6, S6 1 slides. The CCFs of S nerve have been distributed between a branch (CCF330) contributing to subsartorial plexus in the middle of thigh and S nerve (CCFs 323, 326, 327 and SCF 329) continuing downward. This branch is emanating after the slide S4 1 for local innervation. Travelling downward, the S nerve again gave another infrapatellar branch after the slide S6 1. The fascicles have been distributed between S nerve (CCF327, SCFs 333, 334) and this branch (SCFs 331, 332, 335, 336). It is observed that these fascicles undergo 5 transformational processes then two reorganizations of fascicles due to positional migration. The slides marked by two names such as S1 1/S2 1 in the model represent two slides at both faces, top and bottom.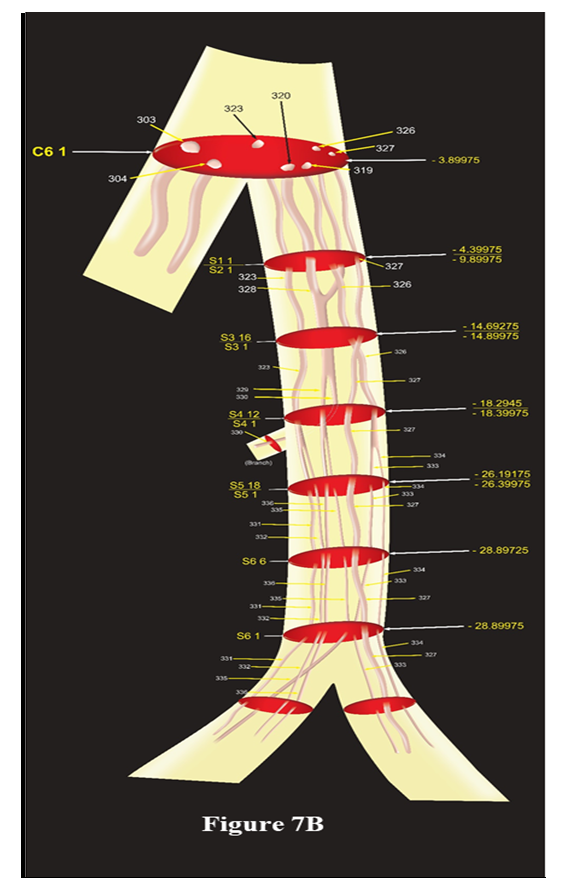 | Figure 7B. The Schematic model of saphenous nerve |
This model represents the 3D pathways of saphenous nerve having CCFs 319, 320, 323, 326, 327 at emergence from cutaneous trunk of femoral nerve undergoing suitable transformation to give branches for innervation. Saphenous gave one branch having SCF 330 undergoing downwards and giving another infrapatellar branch (SCFs 331, 332, 335, 336) and proceeding further downwards to leg and ankle having CCF327 and SCFs 333, 334.Model showing fascicles in subcutaneous trunk, MC, IC nerves (Fig. 7C): The cutaneous trunk bifurcated into S nerve medially and subcutaneous trunk laterally, the subcutaneous trunk was found to possess SCCF305, 306 and CF304 after passing through 1 transformational process between C6 1 and SC1 1. The fascicles are traceable from SC1 1 to SC3 1. After SC3 1 the subcutaneous trunk bifurcated into MC (CF304) medially and IC (SCCFs 305, 306) laterally instantly splitting into MCCF307, MCCF308, MCCF 309 and ICCF307, ICCF308, ICCF309; ICCF310, ICCF311 in IC1 1 after passing through 1 split in MC and two splits in IC. This can be better understood as CF304= MCCF307+ MCCF308+ MCCF 309 in MC1 1, SCCF305= ICCF307 + ICCF308+ ICCF309 and SCCF306= ICCF310 + ICCF311 in IC1 1.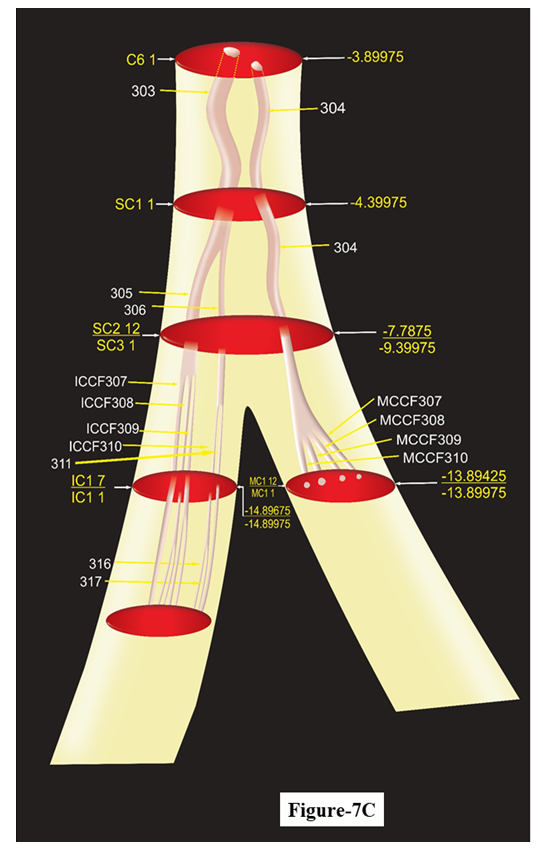 | Figure 7C. The Schematic model of subcutaneous, medial cutaneous, intermedius cutaneous nerves |
The interval of continuous, consistent, and traceable pathways can be computed from these models as all the slides have been calibrated in distance from inguinal ligament. This model explains the plexiform [7] arrangement of nerve fibers but not somatotopic [8].This model is manifestation of fascicular configuration of subcutaneous trunk and its branches medial cutaneous, intermedius cutaneous nerves. After SC3 1 the subcutaneous trunk bifurcated into MC (CF304) medially and IC (SCCFs 305, 306) laterally.
4. Discussion
The correlation, tracking and interpretation of CFs in slides brought out the intervals of continuity, consistency and traceability, position of discontinuity at the occurrence of split, fusion and multiplexing besides identification of longitudinal pathways of fascicles in cutaneous, subcutaneous trunks and S, MC, IC nerves. The mapping of these pathways of fascicles and correlated groups of CFs corresponding to these nerves and trunks brought out the 3D schematic models. These models explain the controversy of somatotopy and plexiform structures [8]. The slides would provide shape, size and directivity of fascicle for matching of nerve fibres for repair, grafting and regeneration. The knowledge of the transitional processes is of paramount importance to neurosurgeons to place electrode preoperatively for stimulation to confirm injured CFs.Models of cutaneous trunk (Fig. 7A), S nerve (Fig. 7B) and subcutaneous trunk along with MC and IC nerves (Fig. 7C):The pathways of all the fascicles in each nerve/trunk were mapped and presented in the 3D models of cutaneous, S nerve, subcutaneous trunks along with MC and IC nerves cranially from top of cutaneous trunk up to the terminal slides of nerves/trunks. The model of cutaneous trunk (Fig. 7A) presents the identified pathways of fascicles in various slides. Similar picturesque view of pathways of fascicles of S nerve, subcutaneous trunk, MC and IC has been presented. From these models the continuous, traceable and consistent interval of the fascicles and position of transformational processes can be computed. The correlation data and models will tremendously help to neurosurgeon for deciding the placement of stimulating electrode to confirm the injured fascicle and location of the injury.Clinical Significance: This small piece of experimental research work, pertaining to microneuroanatomy of femoral nerve and its cutaneous branches namely, cutaneous, subcutaneous trunks, S, MC and IC nerves, is focused on internal morphology involving shapes, sizes, location, orientation, configuration and longitudinal pathways of fascicles. This resulted in evolution of 3D schematic models for these nerves and trunks. If the fascicles of S, MC and IC are damaged, the communication of sensory information from the innervated area will be interrupted leading to aggravation of clinical problems. So it is not merely nerve where injury should be investigated rather injured fascicles should be targeted for diagnosis for complications which may occur anywhere in entire fascicular path from origin to point of innervation. The diagnosis of neural insults requires not only the location and degree of injury but also identification, isolation, orientation, directivity, shape and size of injured nerve CFs for planning surgical repair, grafting and regeneration [9].S may be injured anywhere in its course from thigh to foot due to superficial and long course. In addition to this, procedures such as saphenous vein cut down, vein harvest for bypass surgery or orthopaedic surgery can result in damage to the saphenous nerve resulting in loss of cutaneous sensation in the medial leg. To repair this nerve at fascicular level requires exact location of injury and identification of fascicles. The schematic model of S nerve provides exact location, configuration and orientation of fascicles. Hence this model will be of utmost use to repair the injured S.The location and degree of injury is investigated by the high resolution MRI advanced neurography [4,10]. But this has its own limitations of recording and interpretation. This generate uncertainty in diagnosis and thereby in treatment. Thus the radiologists and neurosurgeons face the impediments of pinpointing the probable position of injury and identification of fascicles. Therefore, the imagery coupled with our internal morphological study together can refine the interpretation for identification of injured CF and location of injury. Methodically, it can be done by one to one correlation between images of transverse histological and high resolution MRI advanced neurographic sections at the same position from inguinal ligament. The distance of location of injured fascicle from inguinal ligament may be computed in MRI neurography and then the calibrated histological section in the evolved models of cutaneous, subcutaneous trunks, S, MC and IC nerves at the same level may be compared and examined for confirmation of identified injured fascicle. After identification of injured fascicle, the idea of shape, size, location and orientation can also be derived from our model more so from histological section for matching, alignment and directivity of nerve fibres for repair and grafting. The neurosurgery at fascicular level is currently uncommon however, with upcoming science and technology in tomorrow’s world, present study will be highly useful. Fascicles were mapped in pudendal nerve [11], femoral nerve [12] and in sciatic nerve [13]. But the fascicles were tracked in the slides at the interval of 5 mm in these studies. But we correlated the fascicles in the slides more precisely at the interval of one mm. The larger sampling interval may cause missing of the transformational processes which are very important for suitable placement of stimulating electrodes. An attempt has been made to carry out 3D peripheral nerve fascicle reconstruction in median nerve [14]. Gustafson proposed design and development of nerve cuff electrode for targeted stimulation [12]. In the same continuity, with this pioneer work, the stimulation electrode if designed focused on fascicles, will have minimal invasion on surrounding structure.
5. Conclusions
The models of cutaneous, subcutaneous trunks, S, MC and IC nerves brought out by correlation of fascicles and grouped fascicles together with their configuration data present the straight, continuous and identified pathways of CFs interrupted by transformational processes calibrated in distance from inguinal ligament. This data will be of utmost importance to imagery guided microneurosurgical interventions more precisely at fascicular level together with the imagery interpretation to radiologists and neurosurgeons to assess injury and its location in an identified fascicular pathway to plan for its repair and surgical access. Fascicular electrode may be designed and developed like nerve cuff electrode [6] to improve neural microsurgery tremendously at fascicular level.
References
| [1] | Singh R, Tubbs RS, Singla, M. Classification and fascicular analysis of variant branching pattern of femoral nerve for microsurgical intervention. A series of thirteen cadavers. Int J Morphol 2016; 34(2):561-569. |
| [2] | Mc Glone F. 2009. The cutaneous sensory system Available from: https://www.researchgate.net/publication/223490107 The cutaneous system [accessed Nov, 2019]. |
| [3] | Ebenezer GJ, McArthur JC, Thomas D. Denervation of skin in neuropathies: the sequence of axonal and Schwann cell changes in skin biopsies. Brain 2007; 130: 2703-2714. |
| [4] | Chhabra Avneesh, Lianxin Zhao, John A Carrino, Eo Trueblood, Saso Koceski, Filip Shteriev et al. 2013. MR Neurography: Advances. Radiology Research and Practice. Volume 2013, 14 pages Article ID 809568. |
| [5] | Singh R, Rajkumar, Chandra N, Gupta Kavita, Ansari MS. Discovery of microanatomy of nerve to iliacus and its clinical significance. Int J Morph. 2019; 37(3): 1150-1163. |
| [6] | Chandra Naresh, Singh Rajani. Tracking of Fascicles of Sartorius and Pectineus Nerves-A key to Neurosurgery. Journal of Clinical and Diagnostic Research 2019; 13(1): AC01-AC08. |
| [7] | Sunderland S. A classification of peripheral nerve injuries producing loss of function. Brain 1951; 74 (4): 491–516. |
| [8] | Stewart JD. Peripheral nerve fascicles: anatomy and clinical relevance. Muscle & Nerve 2003; 28:525–541. |
| [9] | Payne S Houston. Nerve Repair and Grafting in the Upper Extremity. J South Orthop Assoc 2001; 10(2). https://www.medscape.com/viewarticle/423216_6. |
| [10] | Bäumer Philipp, Sabine Heiland, Martin Bendszus, Mirko Pham. MR Neurography - Diagnostic Criteria to Determine Lesions of Peripheral Nerves. Clinical Neurology 2012; page 10-14. Available at Magnetom Flash · 2/2012 www. siemens. com/magnetom-world. |
| [11] | Gustafson KJ, Zelkovic PF, Feng AH et al. Fascicular anatomy and surgical access of the human pudendal nerve. World J Urol 2005; 23(6):411-8. |
| [12] | Gustafson KJ, Pinault GCJ, Neville JJ et al. Fascicular anatomy of human femoral nerve: implications for neural prostheses using nerve cuff electrodes. J Rehab Res Dev. (JRRD) 2009; 46 (7): 973-984. |
| [13] | Gustafson KJ, Grinberg Y, Joseph S et al. Human distal sciatic nerve fascicular anatomy: implications for neural prostheses using nerve cuff electrodes. J Rehab Res Dev. (JRRD) 2012; 49 (2): 309-322. |
| [14] | Zhong Y, Wang L, Dong J et al. Three dimensional reconstruction of peripheral nerve internal fascicular groups. Sci Rep 2015; 5: 171-68. |











 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML


