Nasr A. Belacy MD 1, 2, Tawfik M. Ghabrah MD 3, Mostafa H. Abdelsalam1, 4, Magdi A. El-Damarawi MD 1, 5, Basem M. Elsawy MD 1, 6, Noha A. Nasif MD 1, 7, Eman A. El-Bassuoni MD 1, 8
1Department of Physiology, School of Medicine, University of Tabuk, Saudi Arabia
2Department of Physiology, School of Medicine, Benha University
3Department of Community Medicine, School of Medicine, University of Tabuk, Saudi Arabia
4Department of Physiology, School of Medicine, Zagazig University
5Department of Physiology, School of Medicine, Tanta University
6Department of Physiology, School of Medicine, Beni Swaif University
7Department of Physiology, School of Medicine, Ain Shams University
8Department of Physiology, School of Medicine, El Minia University
Correspondence to: Nasr A. Belacy MD , Department of Physiology, School of Medicine, University of Tabuk, Saudi Arabia.
| Email: |  |
Copyright © 2014 Scientific & Academic Publishing. All Rights Reserved.
Abstract
Conventional oral spirometry is a commonly used test for respiratory functions. However, the nasal passages are the primary pathway for regulating ventilation and modulating ventilated air. Here, we tested the validity of using the nasal route (nasal spirometry) for the evaluation of respiratory functions. 250 healthy young adults (150 males and 100 females; 17 to 23 years of age) were subjected to two spirometry tests: oral spirometry by using a mouth piece and nasal spirometry by using a face mask. Measurement parameters included: Vital capacity (VC), forced vital capacity (FVC), forced expiratory volume first second (FEV1), FEV1/FVC%, Forced expiratory flow (FEF25/75%). and maximum voluntary ventilation (MVV). In both males and females, only VC was significantly higher in nasal than oral spirometry, while FVC, FEV1, FEV1/FVC % FEF25/75% and MVV were significantly higher in oral than nasal spirometry. Prediction equations for different measurements of nasal spirometry were derived by multiple regression analysis using sex, height, and weight as independent variables. We conclude that nasal spirometry could be a valid procedure which may be more real in expressing normal respiratory functions.
Keywords:
Nasal spirometry, Respiratory functions, Vital capacity, FEV1, MVV, Pulmonary function
Cite this paper: Nasr A. Belacy MD , Tawfik M. Ghabrah MD , Moustafa H. Abdelsalam MD , Magdi A. El-Damarawi MD , Basem M. Elsawy MD , Noha A. Nasif MD , Eman A. El-Bassuoni MD , Nasal Spirometry: A New Approach for Spirometry to Evaluate Respiratory Functions, Basic Sciences of Medicine , Vol. 3 No. 2, 2014, pp. 30-35. doi: 10.5923/j.medicine.20140302.03.
1. Introduction
Spirometry is the most useful and commonly available test of pulmonary function [1]. Prediction values are available only for conventional lung function devices using the mouth piece. Although some studies have described the use of a face mask as an alternative for subjects that cannot hold the mouth piece correctly [2] and others have described the use of nasal spirometry to assess nasal patency [3] and nasal resistance [4], no prediction equations are available for normal pulmonary function tests during nasal The nose is the major portal of air exchange between the internal and external environment [5] and many anatomical and physiological differences have been described between the nasal and oral routes for breathing, including volume of dead space and airway resistance [6-8]. The nasal mucosa secretes nitric oxide which induces broncho-dilation, decreases pulmonary resistance, and increases dynamic lung compliance [9]. In the current study, we examined the validity of nasal spirometry (using a face mask) in assessing pulmonary function.
2. Methods
2.1. Subjects
Subjects consisted of 150 male and 100 female healthy, adult students of the age range 17-23 years. Participants (randomly chosen from the University of Tabuk in Saudi Arabia) were nonsmokers and were free from any acute or chronic conditions that could affect the validity of the tests. All subjects gave their informed consent after explanation of all the test procedures. The study was approved by the Research Committee of the University of Tabuk. Data were obtained for each subject regarding age, sex, height, and weight.
2.2. Spirometry
A computerized Spirolab II® spirometer (Medical International Research MIR, Rome, Italy) with a mouth piece connection was used for this study. This Spirolab is based on a turbine sensor which works on the infra-red interruption principle. Spirolab makes breathing tests and calculates an index of test acceptability (test quality control) and a measure of reproducibility. It also gives functional interpretation with 11 possible levels following the latest ATS/ERS (American Thoracic Society/European Respiratory Society) classification [10-12].For oral spirometry; each test subject was connected to the spirometer with the conventional cardboard mouthpiece supplied with the spirometer. Nasal spirometry requires a face mask which is non-compressible and well fitted to the face, with sealed side slits. The breathing tube must have the same diameter as the mouth piece so that it fits properly and can be easily connected to the turbine of the spirometer. After many trials, we chose the face mask (Adult No. 5) supplied by Shinmed Reusable Resuscitator® (autoclavable), model SW72200A (Wellkang Ltd, London, UK). Measured parameters of both oral and nasal spirometry consisted of: Vital capacity (VC), forced vital capacity (FVC), forced expiratory volume first second (FEV1), FEV1/FVC%, forced expiratory flow between 25% and 75% of forced vital capacity (FEF25/75%) and maximum voluntary ventilation (MVV).
2.3. Instructions to Subjects
All subjects received the same instructions. They were allowed to do multiple trials before the start of the experiment in order to be familiar with all of the test procedures.
2.4. Oral Spirometry
A cardboard mouthpiece without teeth grip was used, whereby the subject was required to hold the mouth piece and the nose was closed tightly with the nose clip. The subject was allowed to complete at least 3 trials and the highest one was considered the right one. At least 3 minutes for rest were allowed between any two trials.
2.5. Nasal Spirometry
Each subject held the face mask tightly and was instructed to close their mouth firmly. All subjects were young educated and cooperative and all experiments were attended by a member of the research team. Any condition where mouth leakage occurred during nasal spirometry either reported by the subject or noticed by the research team the test was discarded and repeated all over again. Before the nasal spirometry trials, the subject washed his/her nose with 0.9 NaCl solutions to clear it from any secretions or crusts, and to relieve any nasal obstruction. The rest of the instructions were the same as for oral spirometry. All measurements were done in the sitting position with the guidelines for spirometry rigidly followed [11, 12].
2.6. Statistical Analysis
The results of VC, FVC, and FEV1, FEV1/FVC%, FEF25/75% and MVV measured by oral and nasal spirometry were compared using paired t-test with a significance level of p < 0.05. For generating prediction equations for nasal spirometry, correlations were made for different parameters measured against age, height, weight, and sex as independent variables in all subjects. Only the significant correlations were included in the equations. Multiple regression analyses were then performed to derive separate prediction equations for each Spirometric measurement. For all lung indices examined, simple linear models provided more acceptable fits to the data may be due the narrow age range. Therefore, linear models were chosen as the basic format for evaluating the relationships between the dependent variables and the independent variables.IBM SPSS® for Windows (v. 21) was used for all statistical analysis.
3. Results
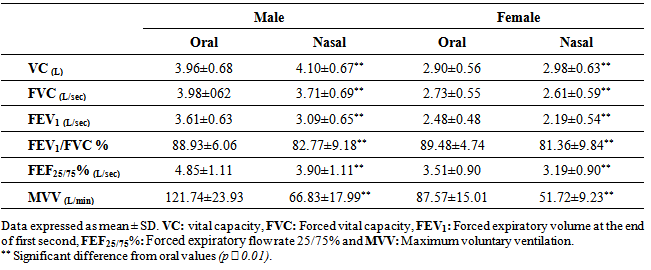
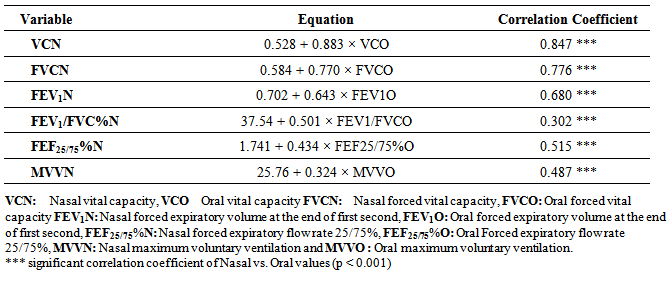
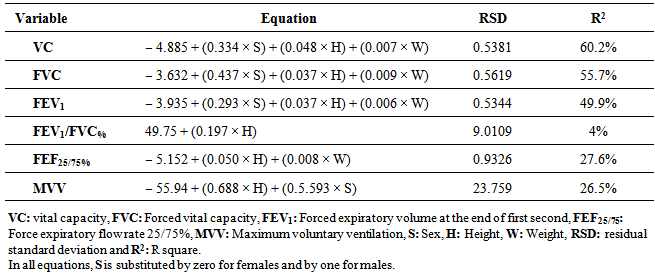
In this study, 250 subjects (150 male and 100 females) were included. Table (1) shows the means and standard deviation of age, height and weight of the included subjects. All subjects were examined by the two spirometric measurements: By traditional oral spirometry with a cardboard mouth piece and by nasal spirometry with face mask. Table (2) shows the comparison of the VC, FVC, FEV1, FEV1/FVC%, FEF25/75% and MVV as measured by both oral and nasal spirometry. Values obtained by oral spirometry were significantly higher than those obtained by nasal spirometry in both male and female groups, except for (slow) VC which was significantly higher in nasal than oral spirometry (Figures 1 to 6). These results indicate that the values obtained by oral spirometry do not reflect the actual values that result when a person respires normally via the nasal passages. However, table (3) shows a significant correlation between oral and nasal spirometry, so prediction equations were derived for nasal spirometry based on the oral values.The next step in this study was to derive prediction equations using multiple regression analysis for the different values obtained by nasal spirometry. The effects of age, height, weight, and sex on different parameters were evaluated. Non-significant variables were excluded and regression analysis was repeated including only the significant variables. Height was found to affect all dependent variables, while sex had an effect (not shown in the figures) on VC, FVC, FEV1 and MVV. Although significant, the effect of weight on VC, FVC, FEV1 and FEF25/75% showed the least effect in all independent variables. All of the data for male and female subjects are shown in Figures 1 to 10. In all regression models, age was not significant, likely due to the narrow age range of the subjects included in this study. Age was therefore not included as a variable in any of the regression equations. Table (4) shows the prediction equations for all parameters of nasal spirometry. For simplicity, one equation is used for both male and female subjects with the sex (S) substituted by zero in case of female subjects and by one in male subjects. It is clear from this table that VC was the parameter most affected by the different independent variables, while FEV1/FVC was the least affected.Table 1. Characteristics of male and female subjects included in the study
 |
| |
|
Table 2. Parameters of spirometry measured via oral and nasal routes in male and female subjects
 |
| |
|
Table 3. Prediction equations to calculate different nasal respiratory parameters based on their respective oral values
 |
| |
|
Table 4. Prediction equations for parameters measured by nasal spirometry
 |
| |
|
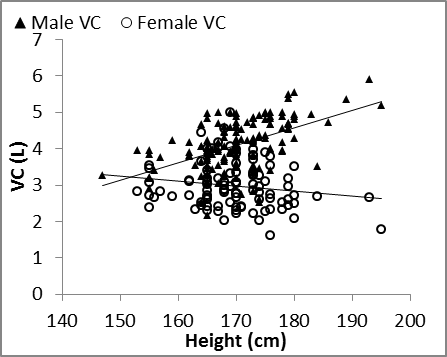 | Figure 1. Nasal VC vs. height in males and females |
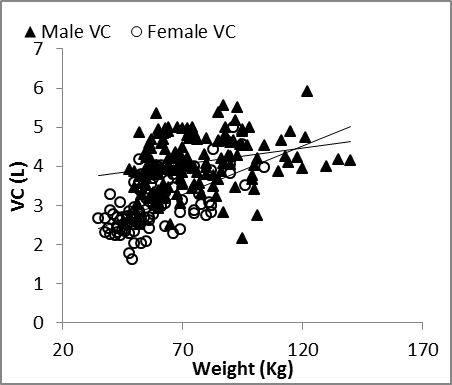 | Figure 2. Nasal VC vs. weight in males and females |
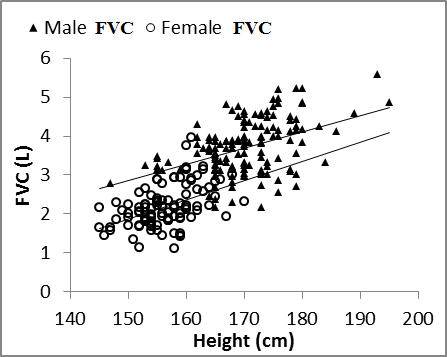 | Figure 3. Nasal FVC vs. height in males and females |
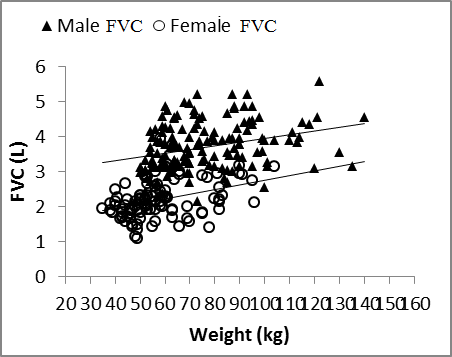 | Figure 4. Nasal FVC vs. weight in males and females |
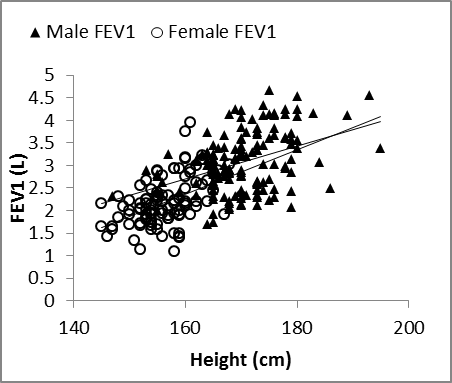 | Figure 5. Nasal FEV1 vs. height in males and females |
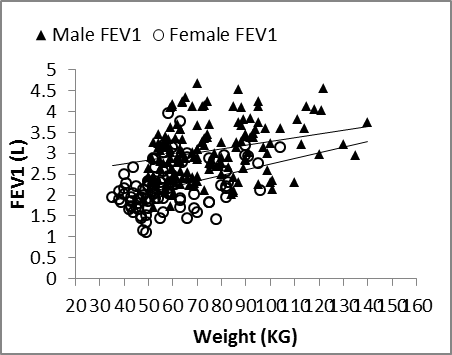 | Figure 6. Nasal FEV1 vs. weight in males and females |
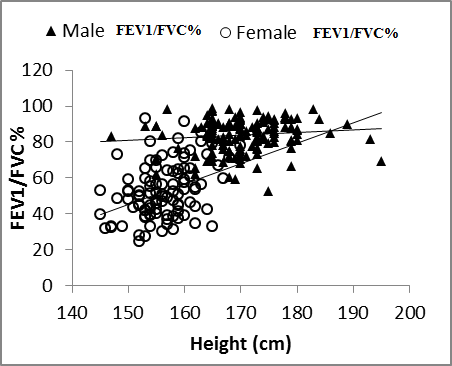 | Figure 7. Nasal FEV1/FVC% vs. height in males and females |
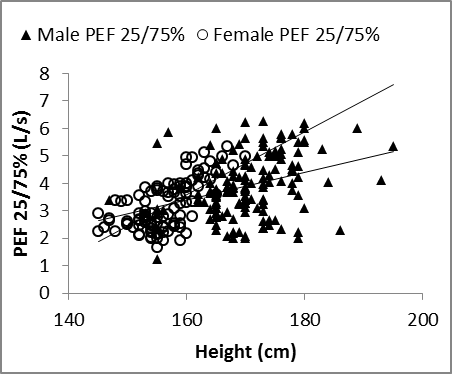 | Figure 8. Nasal FEF25/75%vs. height in males and females |
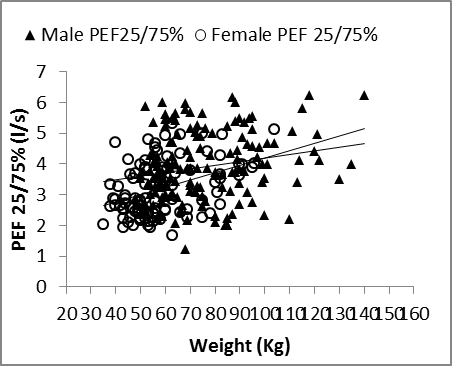 | Figure 9. Nasal FEF25/75% vs. weight in males and females |
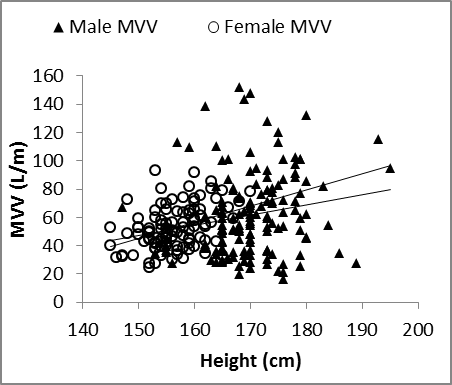 | Figure 10. MVV vs. Height in males and female |
4. Discussion
Inhalation of a deep breath through the nasal passages permits deeper air intake into the lungs than oral breathing. This well-known simple fact raises a fundamental question as to the efficacy of oral spirometry as the ideal method to measure respiratory volumes and capacities and hence, evaluate respiratory function. The current study focused on the potential of using nasal spirometry as a valid tool for assessing pulmonary function. We first compared the results of measurements of nasal spirometry with the same parameters measured by oral spirometry. Our data clearly showed that all the forced parameters (FVC, FEV1, FEV1/FVC, FEF25/75% and MVV) were significantly higher in oral as compared to nasal spirometry, except the vital capacity (slow vital capacity) which was significantly higher in nasal than oral spirometry. These results indicate that when breathing is slow (which is the case in normal at rest breathing) nasal breathing provides volumes larger than the oral breathing, while in situations of forced breathing (as seen during conditions of increased effort and activity) the oral breathing route provides larger volumes. Yilmaz and Naclerio [5] reported that the normal nose is capable of sustaining 20-30 L of airflow per minute during eupnoea, while larger volumes require oral breathing to supplement nasal breathing. The slow vital capacity showed a significant (although small) increase in nasal as compared to oral spirometry (140 ml in males and 80 ml in females). It has been shown in many previous studies that the nasal passages have multiple influences on the breathing process that make the nasal breathing superior to oral breathing. Tanaka et al. [7] reported that the dead space and airway resistance were significantly greater during nasal than during oral breathing. Other researchers have reported that the nasal mucosa secrets nitric oxide, which has been shown to have potent bronchodilator and vasodilator effects [9, 13], decrease pulmonary resistance, and increase dynamic lung compliance [9]. Also, it was reported that during sleep, nasal breathing has a stimulant effect on breathing so that ventilation increases during nasal breathing as compared with oral breathing [8]. All of these factors may contribute to the significant difference between nasal and oral slow vital capacity. Also, the slow pattern of nasal breathing may provide for more inflation of the lung. On the other hand, the higher values of dynamic lung parameters in oral spirometry compared to nasal spirometry could be explained by the fact that the oral passage is wider and shorter than the nasal passages. Another important factor is that most normal subjects are not familiar with applying enough force to expel maximum air volume through the nasal passages as is possible during forced oral respiration. Under such conditions of increased force, the results may be changed. In a similar trial, Wohlgemuth et al. [2] compared between the use of a face mask and the traditional oral mouth piece in spirometry. They reported that FVC and FEV1 were 200 ml lower with mask than with mouth piece, which is similar to our results. However, in their study it was not clear whether the subjects used pure nasal or naso-oral breathing during the use of the face mask.In the next step, we derived prediction equations based on the presence of significant correlation between corresponding nasal and oral measurements. These equations will help to predict the values expected for nasal spirometry based on oral spirometry measurements. Also, these equations are important as they indicate that the factors affecting oral and nasal spirometry are the same.In the present study, we also generated prediction equations for the different parameters of nasal spirometry based on our results in Tabuk University. The effects of the independent variables: age, sex, height and weight on the different parameters of the nasal spirometry were evaluated. The effect of age was not obvious, due to the narrow age range in our study (17-23 years); thus, it was excluded from all the equations. In a study by Korotzer et al. [14], the age range of the subjects was between 22 and 33 years and they also reported that the coefficient of age was not significant. However, age was an effective independent variable in a study by Hankinson et al. [15] on a sample of the U.S. population that covered a wide age range (8-80 years). On the other hand, we found that sex was an important factor affecting most of the parameters of nasal spirometry. VC, FVC, FEV1, and MVV were significantly lower in females than males, while the percentages of FEV1/FVC and FEF25-75% showed no significant difference. The lower values of VC, FVC, FEV1, and MVV in females than males could be attributed to the lower values of height (156.4±5.1 cm in females and 170.2±7.3 cm in males) and weight (58 ±14.8 kg for females and 75.8±19.5 kg males). When the different parameters of nasal spirometry were evaluated against the factor of height, it was found to affect all the parameters by a positive relationship. In several other studies, height was also shown to be an important variable affecting the parameters of oral spirometry [15-17]. In contrast, the effect of weight was small, but significant. Weight significantly affected VC, FVC, FEV1, and FEF25-75%. From Table 3, it can be seen that all of the parameters affected by weight were affected primarily by height. So, the positive effect of weight could be explained by the positive effect of height. In a study by Falaschetti et al. [17], they reported no relationship between weight and lung function, although height was effective. This may have been due to the large number of subjects in their study which may have included some obese persons.
5. Conclusions
We conclude that nasal spirometry could be an easy and reliable method for valid evaluation of respiratory functions specially in cases in which oral spirometry cannot be performed as in children, elder persons and some neurological conditions. The prediction equations derived in this study can be used as a guide for future studies that apply this method of spirometry.
6. Conflict of Interest
The authors declare no conflict of interests.
References
| [1] | Hayes D Jr., Kraman SS. The physiologic basis of spirometry. Respir Care. 2009; 54(12): 1717-26 |
| [2] | Wohlgemuth M, van der Kooi EL, Hendriks JC, Padberg GW, Folgering HT. Face mask spirometry and respiratory pressures in normal subjects. Eur Respir J. 2003; 22(6): 1001-6. |
| [3] | Nagle SK and Kelkar RS. Spirometry as an objective tool for nasal patency. Indian Journal of Otolaryngologyand Neck Surgery 2006;59(1):41-42. |
| [4] | Goyal M, Verma NS, Goel A, Kumar P and Tiwari S. Nasal spirometry. Indian J Physiol pharmacol. 2010; 54(3):271-6. |
| [5] | Sahin-Yilmaz A, Naclerio RM. Anatomy and physiology of the upper airway. Proc Am Thorac Soc. 2011; 8(1): 31-9. |
| [6] | Fitzpatrick MF, McLean H, Urton AM, Tan A, O'Donnell D, Driver HS. Effect of nasal or oral breathing route on upper airway resistance during sleep. Eur Respir J. 2003; 22(5): 827-32 |
| [7] | Tanaka Y, Morikawa T, Honda Y. An assessment of nasal functions in control of breathing. J Appl Physiol. 1988; 65(4): 1520-4. |
| [8] | McNicholas WT, Coffey M, Boyle T. Effects of nasal airflow on breathing during sleep in normal humans. Am Rev Respir Dis. 1993; 147(3): 620-3. |
| [9] | Dupuy PM, Shore S A, Drazen JM, Frostell C, Hill WA, Zapol WM. Bronchodilator action of inhaled nitric oxide in guinea pigs. J Clin Invest. 1992; 90(2): 421–428. |
| [10] | Standardized lung function testing. Official statement of the European Respiratory Society. Eur Respir J Suppl. March 1993; 16: 1-100. |
| [11] | Miller MR, Crapo R, Hankinson J, V. Brusasco, F. Burgos, R. Casaburi, A. Coates, P. Enright, C.P.M. van der Grinten, P. Gustafsson, R. Jensen, D.C. Johnson, N. MacIntyre, R. McKay, D. Navajas, O.F. Pedersen, R. Pellegrino, G. Viegi and J. Wanger General considerations for lung function testing. Eur Respir J. 2005; 26: 153-161. |
| [12] | Miller MR, Hankinson J, Brusasco V, Burgos F, Casaburi R, Coates A, Crapo R, Enright P, van der Grinten CP, Gustafsson P, Jensen R, Johnson DC, MacIntyre N, McKay R, Navajas D, Pedersen OF, Pellegrino R, Viegi G, Wanger J; Standardisation of spirometry. Eur Respir J. 2005; 26(2): 319-38. |
| [13] | Ichinose F, Roberts JD, Jr, Zapol WM. Inhaled Nitric Oxide: A Selective Pulmonary Vasodilator: Current Uses and Therapeutic Potential. Circulation. 2004; 109: 3106-11. |
| [14] | Korotzer B, Ong S, Hansen JE. Ethnic differences in pulmonary function in healthy nonsmoking Asian-Americans and European-Americans. Am J Respir Crit Care Med. 2000; 161(4 Pt 1): 1101-8. |
| [15] | Hankinson JL, Odencrantz JR, Fedan KB. Spirometric reference values from a sample of the general U.S. population. Am J Respir Crit Care Med. 1999; 159(1): 179-87. |
| [16] | Ip MS, Karlberg EM, Karlberg JP, Luk KD, Leong JC. Lung function reference values in Chinese children and adolescents in Hong Kong. I. Spirometric values and comparison with other populations. Am J Respir Crit Care Med. 2000; 162(2 Pt 1): 424-9. |
| [17] | Falaschetti E, Laiho J, Primatesta P, Purdon S. Prediction equations for normal and low lung function from the Health Survey for England. Eur Respir J. 2004; 23(3): 456-63. |











 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML


