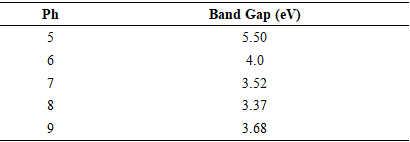
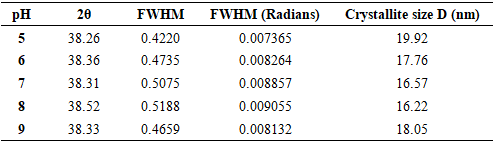
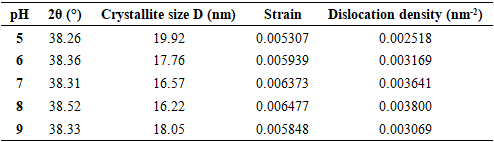
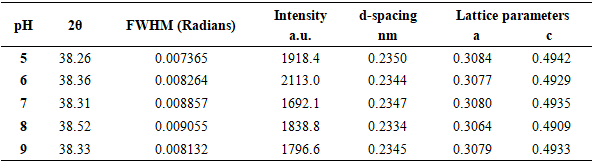
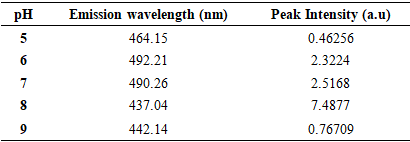
| [1] | Haleem, A., Javaid, M., Singh, R. P., Rab, S., & Suman, R. (2023). Applications of nanotechnology in medical field: a brief review. Global Health Journal, 7(2), 70-77. |
| [2] | Afolalu, S. A., Ikumapayi, O. M., Oloyede, O. R., Ogedengbe, T. S., & Ogundipe, A. T. (2022). Advances in Nanotechnology and Nanoparticles in the 21st Century–an overview. Proceedings of the International Conference on Industrial Engineering and Operations Management. Nsukka, Nigeria, |
| [3] | Aldeen, T. S., Mohamed, H. E. A., & Maaza, M. (2022). ZnO nanoparticles prepared via a green synthesis approach: Physical properties, photocatalytic and antibacterial activity. Journal of Physics and Chemistry of Solids, 160, 110313. |
| [4] | Droepenu, E. K., Wee, B. S., Chin, S. F., Kok, K. Y., & Maligan, M. F. (2022). Zinc oxide nanoparticles synthesis methods and its effect on morphology: A review. |
| [5] | Malik, S., Muhammad, K., & Waheed, Y. (2023). Nanotechnology: a revolution in modern industry. Molecules, 28(2), 661. |
| [6] | Patil, N., Bhaskar, R., Vyavhare, V., Dhadge, R., Khaire, V., & Patil, Y. (2021). Overview on methods of synthesis of nanoparticles. International Journal of Current Pharmaceutical Research, 13(2), 11-16. |
| [7] | Pulingam, T., Foroozandeh, P., Chuah, J.-A., & Sudesh, K. (2022). Exploring various techniques for the chemical and biological synthesis of polymeric nanoparticles. Nanomaterials, 12(3), 576. |
| [8] | Latolla, N. S. (2022). Phytochemical Investigation and Antidiabetic Activity of Cissampelos capensis and Strychnos henningsii the Eastern Cape Medicinal Plants. |
| [9] | Srujana, S., & Bhagat, D. (2022). Chemical-based synthesis of ZnO nanoparticles and their applications in agriculture. Nanotechnology for Environmental Engineering, 7(1), 269-275. |
| [10] | Alharbi, F. N., Abaker, Z. M., & Makawi, S. Z. A. (2023). Phytochemical substances—mediated synthesis of zinc oxide nanoparticles (ZnO NPS). Inorganics, 11(8), 328. |
| [11] | Radulescu, D.-M., Surdu, V.-A., Ficai, A., Ficai, D., Grumezescu, A.-M., & Andronescu, E. (2023). Green synthesis of metal and metal oxide nanoparticles: a review of the principles and biomedical applications. International Journal of Molecular Sciences, 24(20), 15397. |
| [12] | Souza, T. A., Menezes, A. C., Santos, C. K., Jesus, F. G., Rocha, E. C., & Araújo, M. S. (2024). Toxicity of Bioactive Compounds of Strychnos pseudoquina (Loganiaceae) in Spodoptera frugiperda (Noctuidae). Sustainability, 16(11), 4430. |
| [13] | Nair, G. M., Sajini, T., & Mathew, B. (2022). Advanced green approaches for metal and metal oxide nanoparticles synthesis and their environmental applications. Talanta Open, 5, 100080. |
| [14] | Ismail, S. M. M., Ahmed, S. M., Abdulrahman, A. F., & AlMessiere, M. A. (2023). Characterization of green synthesized of ZnO nanoparticles by using pinus brutia leaves extracts. Journal of Molecular Structure, 1280, 135063. |
| [15] | Karam, S. T., & Abdulrahman, A. F. (2022). Green synthesis and characterization of ZnO nanoparticles by using thyme plant leaf extract. Photonics, |
| [16] | Abdelbaky, A. S., Abd El-Mageed, T. A., Babalghith, A. O., Selim, S., & Mohamed, A. M. (2022). Green synthesis and characterization of ZnO nanoparticles using Pelargonium odoratissimum (L.) aqueous leaf extract and their antioxidant, antibacterial and anti-inflammatory activities. Antioxidants, 11(8), 1444. |
| [17] | Singh, S., Gade, J. V., Verma, D. K., Elyor, B., & Jain, B. (2024). Exploring ZnO nanoparticles: UV–visible analysis and different size estimation methods. Optical Materials, 152, 115422. |
| [18] | Abdelghani, G. M., Ahmed, A. B., & Al-Zubaidi, A. B. (2022). Synthesis, characterization, and the influence of energy of irradiation on optical properties of ZnO nanostructures. Scientific Reports, 12(1), 20016. |
| [19] | Indumathi, T., Theivarasu, C., Pradeep, I., Rani, M. T., Magesh, G., Rahale, C. S., & Kumar, E. R. (2021). Effects of Nd doping on structural, optical, morphological and surface-chemical state analysis of ZnO nanoparticles for antimicrobial and anticancer activities. Surfaces and Interfaces, 23, 101000. |
| [20] | Ghosh, S., Ghosh, A., Pramanik, S., Kuiri, P. K., Sen, R., & Neogi, S. K. (2022). Synthesis of ZnO nanoparticles by co-precipitation technique and characterize the structural and optical properties of these nanoparticles. Journal of Physics: Conference Series, |
| [21] | de Almeida, W. L., Ferreira, N. S., Rodembusch, F. S., & de Sousa, V. C. (2021). Study of structural and optical properties of ZnO nanoparticles synthesized by an eco-friendly tapioca-assisted route. Materials Chemistry and Physics, 258, 123926. |
| [22] | Almarhoon, Z. M., Indumathi, T., & Kumar, E. R. (2022). Optimized green synthesis of ZnO nanoparticles: evaluation of structural, morphological, vibrational and optical properties. Journal of Materials Science: Materials in Electronics, 33(30), 23659-23672. |
| [23] | Jayachandran, A., Aswathy, T., & Nair, A. S. (2021). Green synthesis and characterization of zinc oxide nanoparticles using Cayratia pedata leaf extract. Biochemistry and Biophysics Reports, 26, 100995. |
| [24] | Sedefoglu, N. (2023). Characterization and photocatalytic activity of ZnO nanoparticles by green synthesis method. Optik, 288, 171217. |
| [25] | Sarkar, T., Kundu, S., Ghorai, G., Sahoo, P. K., & Bhattacharjee, A. (2023). Structural, spectroscopic and morphology studies on green synthesized ZnO nanoparticles. Advances in Natural Sciences: Nanoscience and Nanotechnology, 14(3), 035001. |
| [26] | Ramalingam, R., Sadasivuni, K., Allohedan, H., Sunderajan, M., & Yuvaraj, S. (2025). Photoluminescent, optical and magnetic property characterization of green chemistry method prepared calcium doped ZnO nanoparticles. Journal of Ovonic Research, 21(1). |
| [27] | Ramesh, P., Saravanan, K., Manogar, P., Johnson, J., Vinoth, E., & Mayakannan, M. (2021). Green synthesis and characterization of biocompatible zinc oxide nanoparticles and evaluation of its antibacterial potential. Sensing and Bio-Sensing Research, 31, 100399. |
| [28] | Al-Harbi, H. F., Awad, M. A., Ortashi, K. M., Al-Humaid, L. A., Ibrahim, A. A., & Al-Huqail, A. A. (2025). Green Synthesis of Zinc Oxide Nanoparticles: Physicochemical Characterization, Photocatalytic Performance, and Evaluation of Their Impact on Seed Germination Parameters in Crops. Catalysts, 15(10), 924. |
| [29] | Wijesinghe, U., Thiripuranathar, G., Iqbal, H., & Menaa, F. (2021). Biomimetic synthesis, characterization, and evaluation of fluorescence resonance energy transfer, photoluminescence, and photocatalytic activity of zinc oxide nanoparticles. Sustainability, 13(4), 2004. |
| [30] | Dey, A., & Somaiah, S. (2022). Green synthesis and characterization of zinc oxide nanoparticles using leaf extract of Thryallis glauca (Cav.) Kuntze and their role as antioxidant and antibacterial. Microscopy research and technique, 85(8), 2835-2847. |
| [31] | Pathak, J., Akhani, S. B., & Rathore, M. S. (2024). Structural and photoluminescence properties of green synthesized ZnO nanoparticles from Calotropis gigantea leaves. Materials Today: Proceedings. |
| [32] | Tabassam, L., Khan, M. J., Hussain, S., Khattak, S. A., Shah, S. K., & Bhatti, A. S. (2022). Structural, optical and antimicrobial characteristics of ZnO green nanoparticles. Journal of Sol-Gel Science and Technology, 101(2), 401-410. |



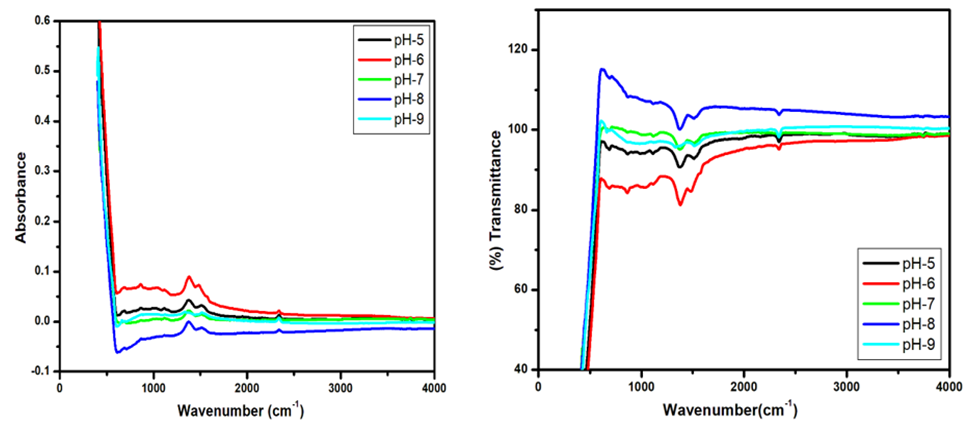
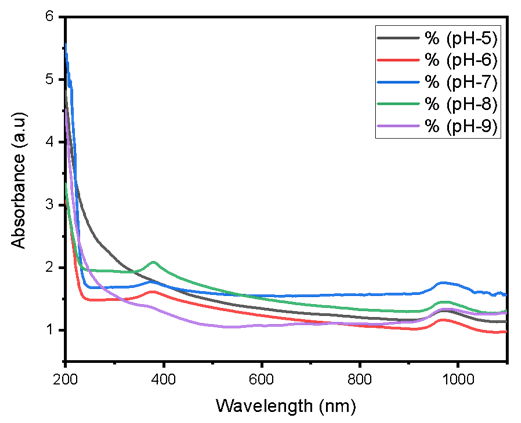
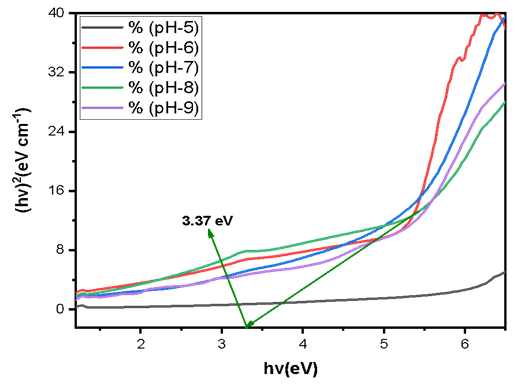
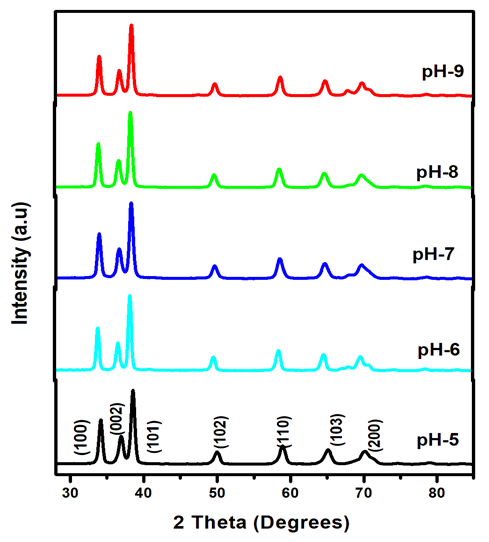
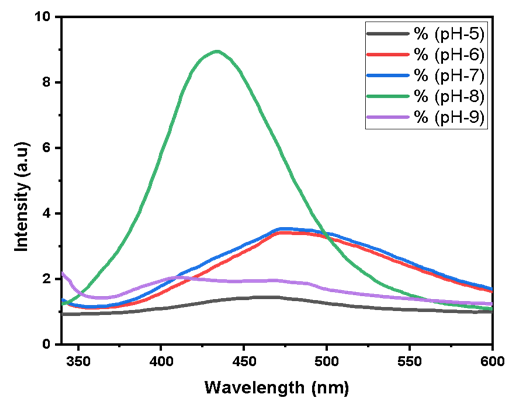
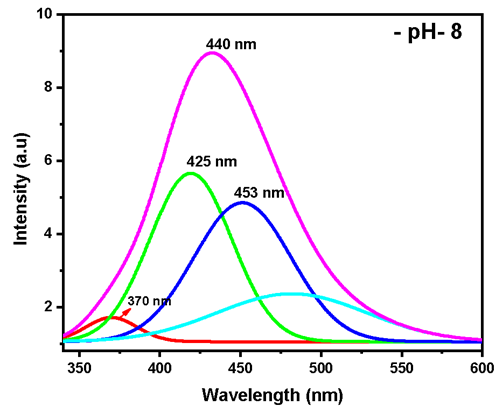
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML