-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Materials Science
p-ISSN: 2162-9382 e-ISSN: 2162-8424
2026; 16(1): 11-18
doi:10.5923/j.materials.20261601.02
Received: Feb. 17, 2026; Accepted: Mar. 6, 2026; Published: Mar. 17, 2026

Synthesis and Application of Casein Carbon Quantum Dots on Cotton Textiles for Improving Flame Retardancy, Thermal Stability, and Abrasion Resistance
Monique Cotrim1, Patrícia Patrício2, Artur Mottin3, Eliane Ayres1
1Design School, State University Of Minas Gerais, Belo Horizonte, Brazil
2Department of Chemistry, CEFET-MG, Belo Horizonte, Brazil
3Department of Mechanical Engineering, CEFET-MG, Belo Horizonte, Brazil
Correspondence to: Monique Cotrim, Design School, State University Of Minas Gerais, Belo Horizonte, Brazil.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In this work, eco-friendly carbon quantum dots derived from casein protein (CCDs) were synthesized through a simple caramelization route and applied to cotton fabric. The CCDs exhibited strong blue photoluminescence at 365 nm and a nitrogen- and oxygen-rich surface that enabled their interaction with cellulose. Cotton fabrics modified with CCDs (0.5% and 2.5% w/w) showed improved thermal stability, retaining key cellulose vibrational features even after exposure to 330°C, while the unmodified fabric underwent extensive degradation. Cigarette ignition tests demonstrated reduced surface deformation compared to pristine cotton, indicating enhanced flame retardancy. Abrasion tests revealed that the 0.5% CCD treatment yielded the best wear resistance, attributed to uniform nanoparticle dispersion and formation of a protective layer.
Keywords: Eco-friendly textile finishing, Casein carbon dots, Flame retardancy, Cotton abrasion resistance
Cite this paper: Monique Cotrim, Patrícia Patrício, Artur Mottin, Eliane Ayres, Synthesis and Application of Casein Carbon Quantum Dots on Cotton Textiles for Improving Flame Retardancy, Thermal Stability, and Abrasion Resistance, American Journal of Materials Science, Vol. 16 No. 1, 2026, pp. 11-18. doi: 10.5923/j.materials.20261601.02.
Article Outline
1. Introduction
- The increasing demand for environmentally friendly functional textile materials has intensified research on bio-derived materials that can enhance fabric performance while minimizing environmental impact. Cotton, the most widely used natural fiber, is highly flammable due to its cellulosic nature and exhibits rapid flame propagation once ignited [1]. Conventional flame-retardant (FR) finishes for cotton textiles are typically based on halogenated compounds, phosphorus-based additives, or nitrogen–phosphorus systems [2]. Halogenated FRs have historically been effective in suppressing flame propagation; however, their use has declined due to concerns regarding toxicity, environmental persistence, and the generation of hazardous combustion by-products [3]. Phosphorus-based treatments, such as organophosphates and phosphonates, are widely used due to their ability to promote char formation in cellulose during thermal degradation. Nevertheless, many of these finishes require complex synthesis routes and involve non-renewable precursors [3]. Additionally, some commercial FR formulations rely on formaldehyde-based crosslinking systems, which raise health and environmental concerns. Consequently, there is increasing interest in developing bio-based flame-retardant systems derived from renewable resources that can be effective while reducing environmental impact [3-5].Among bio-derived materials, casein has attracted particular attention as a sustainable flame-retardant agent for cellulose-based textiles [3-5]. Casein is a phosphoprotein naturally rich in nitrogen and phosphorus functional groups, which are well known to promote char formation and inhibit flame propagation in cellulosic substrates [3-5]. Compared with other nitrogen- or phosphorus-based additives, casein offers several advantages: it is renewable, biodegradable, widely available as a by-product of the dairy industry, and can be processed without the use of toxic reagents. These characteristics make casein a particularly promising precursor for developing environmentally friendly flame-retardant systems. Beyond its intrinsic FR properties, casein also represents a valuable carbon and nitrogen source for the synthesis of carbon quantum dots (CDs): nanometric structures with outstanding optical properties, high stability, biocompatibility, and functional surface groups [6-7]. The conversion of casein into CDs through hydrothermal carbonization yields nitrogen-doped structures with abundant carboxyl, hydroxyl, and amine moieties, facilitating their binding to cellulose and other biopolymers [8-9]. By incorporating nanoparticles alongside organic or inorganic compounds, it is possible to substantially modify fabric surfaces, enabling finishes that enhance abrasion resistance, water repellence, and protection against UV, electromagnetic, and infrared radiation [10]. Additionally, from a lubrication and microstructural perspective, CDs exhibit strong potential for tribological applications. Their nanoscale dimensions, large surface-to-volume ratio, and tailored surface chemistry enhance their performance. At the same time, their improved dispersibility and storage stability in polar media confer clear advantages over traditional carbon materials [11]. In this context, the present study aims to investigate the effect of casein-derived carbon quantum dots (CCDs) on the thermal stability and abrasion resistance of cotton. Although several studies have investigated carbon quantum dots for sensing, catalysis, and biomedical applications, their potential as functional finishes for textiles remains largely unexplored. In particular, limited research has examined the ability of CDs to enhance both thermal stability and mechanical durability of cotton fabrics. Furthermore, the use of casein-derived carbon quantum dots (CCDs) as a bio-based nanomaterial for textile finishing has not yet been systematically investigated. To the best of our knowledge, this is the first paper that investigates the effect of CDs on cotton abrasion resistance. CCDs were synthesized without any harsh chemicals by using a simple caramelization method [12]. The obtained material was characterized by UV-Vis and FTIR. CCDs cotton fabric samples were investigated by FTIR, Thermogravimetric analysis, flame resistance test, and Martindale abrasion test. This approach aims to merge the known charring ability of casein with the nanoscale reactivity and surface functionality of CDs, hypothesizing a cooperative mechanism of thermal protection and wear resistance.
2. Experimental Section
2.1. Materials
- Bovine Milk Casein (Sigma Aldrich, USA) and Citric Acid (Nox, Brazil) were used without purification. 100% Cotton Fabric (twill 3x1, 0,260 g/ m2; dyed; Cedro Têxtil, Brazil) was previously washed with 2 gL-1 with non-ionic detergent (Oxiteno, Brazil) and dried at ambient temperature. A commercial 100% cotton FR textile (twill 3x1, 290 g/ m2, dyed; Cedro Têxtil, Brazil) was used as received.
2.2. Methods
2.2.1. Casein-Derived Carbon-Dots (CCDs) Obtention
- Casein-derived carbon quantum dots (CCDs) were prepared using a simple caramelization method [12] of casein in citric acid. Briefly, casein powder (5% w/w) was added to an aqueous citric acid solution (80 gL-1) under agitation with the aim of a magnetic stirrer and kept at 70°C until its complete dissolution. The solution was then heated to 140°C and stirred until it turned into a viscous, dark-brown caramel solution. After that, deionized water was added to it at room temperature. The supernatant and solid residue were separated by filtration. The supernatant solution was filtered (0,22 µm and 0,045 µm). The following equation calculated the concentration of CCDs in solution,
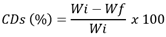 | (1) |
2.2.2. Casein Carbon-Dots Addition to Textile
- As mentioned above, CCDs solutions were used in two concentrations (0,5% and 2,5% w/w). The solutions were heated at 60°C, and then textile samples were immersed for 15 minutes, passed through a lab-pad-roller, with a wet pick-up of 100%, and then air dried.
2.2.3. Characterization
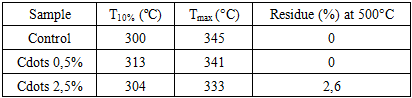
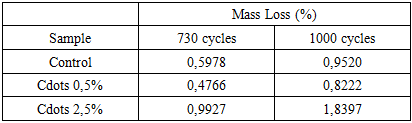
- CCD's absorption spectra were investigated using a UV/Vis spectrophotometer (Shimadzu UV-2600i) at a range of 185 to 800 nm. A 365 nm light source was used to investigate the photoluminescence of the CCD solution. The Fourier transform infrared (FTIR) spectra of CCDs and textile samples were recorded on an Infrared Spectrum instrument (Agilent Technologies, USA), with a 4 cm-1 resolution, 80 scans, and a wavenumber range of 4000-600 cm-1, using the ATR mode. Textile samples (modified and control) were examined before and after being burned in a muffle furnace for 5 minutes at 220°C, 330°C, and 500°C as suggested elsewhere [13].The effect of the addition on cotton fabric was investigated using a Martindale tester (Mathis, Brazil) in accordance with ISO 12947:1-2007. Briefly, circular samples (40 mm) were subject to several rubbing cycles (100, 200, 500, 730, and 1000 cycles) against an abrasive wool cloth and a 9 kPa load. Sample mass variation between cycles was verified by weighing samples with an analytical scale (± 0,0004g). One-way analysis of variance was used to determine statistically significant differences (p <0,05) between groups. All statistical analyses were performed using PAST software.Thermogravimetric analysis (TGA, Shimadzu equipment, model DTG-60H) was conducted under an air atmosphere, with a flow rate of 50 mL min-1, at a heating rate of 10°C min-1, from ambient temperature to 600°C.Flammability property was investigated in accordance with BS 5852 Part 1, using a cigarette as the heat source (source 0), as reported by Manasoglu et al. [14]. Briefly, the textile sample was affixed to polyurethane foam, and a lit cigarette was placed on top until it burned along its entire length. After removing the cigarette ash, the surface deformations were measured with a ruler and a caliper.
3. Results and Discussion
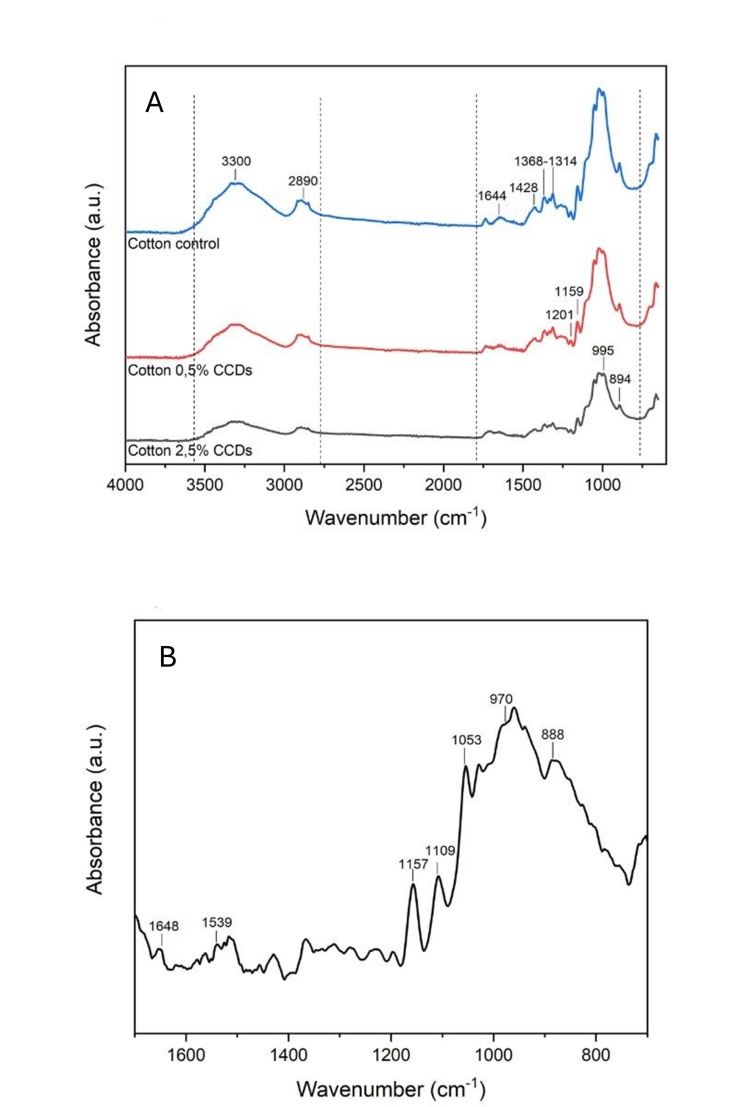
- In our work, we investigated the production of environmentally friendly CDs using casein as a natural precursor (CCDs), without the use of any harsh chemicals. The as-prepared CCDs aqueous solution was yellowish and transparent under daylight and emitted strong blue fluorescence under UV light (365 nm), as shown in Figure 1A. (inset). The UV-vis spectra of CCDs (Figure. 1A) showed two absorption peaks at 250 and 280 nm, which were attributed to the π–π* transition of C=C and n–p* transition of C=O, respectively [12,15]. FTIR spectra of CCDs (Figure. 1B) indicate the presence of nitrogen and oxygen functional groups.
 | Figure 1. A) CCDs UV-vis absorption spectra and CCDS blue fluorescence at 365 nm (inset); B) FTIR spectra of CCDs, casein diluted in citric acid solution, and casein powder |
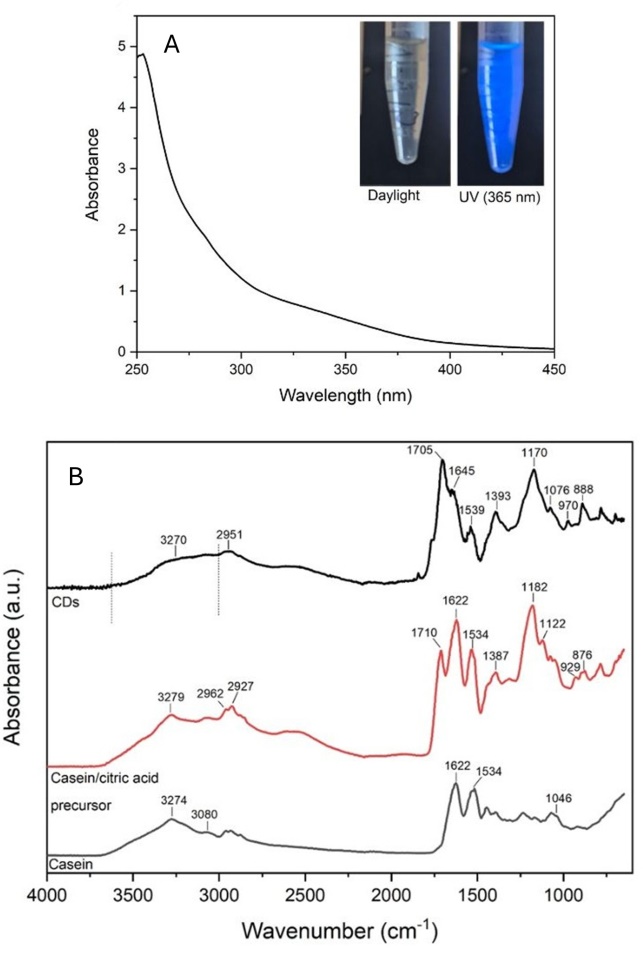
 | Figure 3. A) FTIR spectra of control (unmodified) and CCDS cotton modified samples; B) Subtracted FTIR spectra of 2,5% w/w CCDs cotton |
|
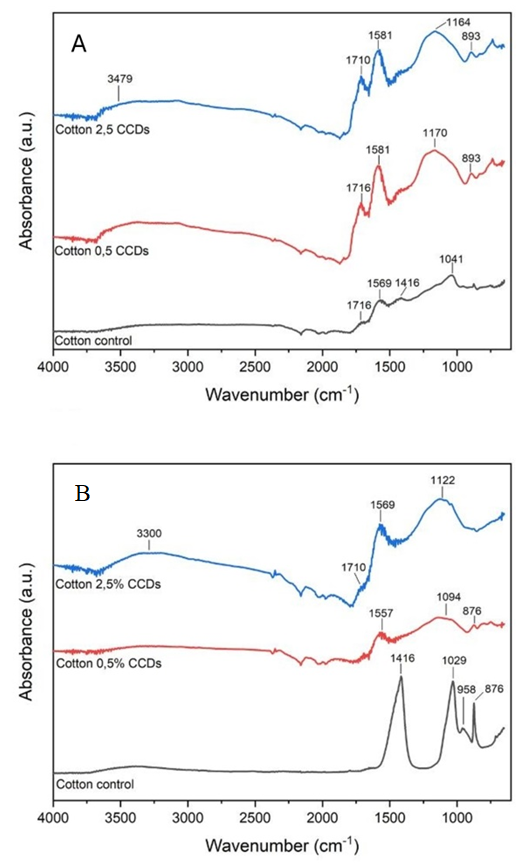
 | Figure 4. TGA curves of CCDs modified samples and control (unmodified cotton) |
|
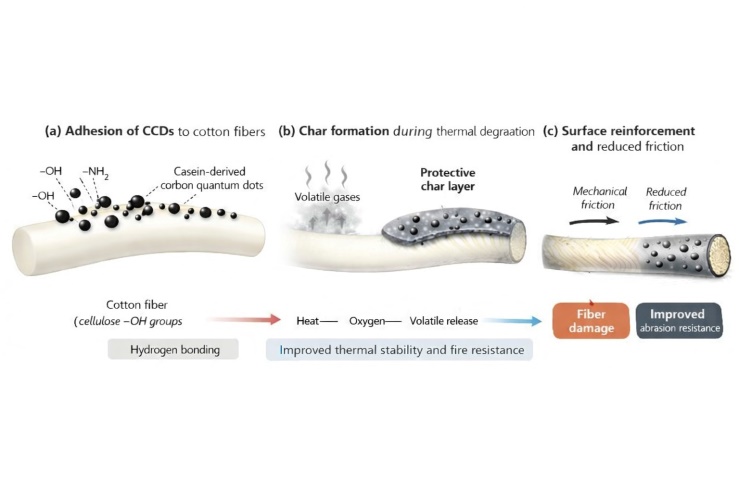 | Figure 5. Proposed mechanism for the enhancement of thermal stability, flame retardancy, and abrasion resistance of cotton textile treated with casein carbon dots (CCD) |
4. Conclusions
- This study demonstrated that casein-derived carbon quantum dots can be synthesized through a green, straightforward method and effectively incorporated into cotton textiles to enhance their functional performance. The presence of CCDs resulted in a synergistic improvement in thermal stability and flame retardancy, delaying cellulose degradation up to 500°C and reducing surface deformation under smouldering-cigarette ignition. The protective effect is attributed mainly to the phosphorus- and nitrogen-rich composition of casein-derived CDs, which promotes char formation and preserves the cellulose backbone at elevated temperatures. Furthermore, abrasion testing revealed that a low CCD concentration (0.5% w/w) significantly improved wear resistance, likely due to the formation of a uniform nanoscale lubricating layer capable of reducing friction and protecting the fiber surface. Although higher CCD loading possibly led to nanoparticle aggregation and reduced abrasion performance, the overall results highlight the potential of CCDs as a versatile, bio-based nanostructured coating for sustainable textile engineering. These findings open new pathways for the development of environmentally responsible, thermally stable, and mechanically resilient cotton materials without resorting to toxic or non-renewable additives.
ACKNOWLEDGEMENTS
- This paper was supported by Fundação de Amparo a Pesquisa de Minas Gerais (FAPEMIG) grant number BPD-00673-22. The authors also acknowledge the support from CNPq (Grant No. 308994/2022-1, Call 08/2022).
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML