-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Materials Science
p-ISSN: 2162-9382 e-ISSN: 2162-8424
2018; 8(1): 1-5
doi:10.5923/j.materials.20180801.01

Surface Modification of High Performance Polymer and Analysis of Surface Properties. -A Brief Review Article
Md. Rasel1, Md. Shah Alam1, Mohammad Tofayel Ahmed2, Jarin Akter3, Habibur Rahman Abir1, Mohammad Sohel Rana1
1Department of Textile Engineering, Southeast University, Dhaka, Bangladesh
2Department of Organic Material Science and Engineering, Pusan National University, Busan, Korea
3Faculty of Textile and Clothing Technology, Hochschule Niederrhein University, Monchengladbach, Germany
Correspondence to: Md. Rasel, Department of Textile Engineering, Southeast University, Dhaka, Bangladesh.
| Email: |  |
Copyright © 2018 Scientific & Academic Publishing. All Rights Reserved.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Nowadays polymer applications have been a vital issue in the digital world. Their main sources are both natural and synthetic. A wide range of polymers are available those have physical, chemical and biological properties to match the requirements of specific applications. Besides initial bulk properties of the polymer or materials, their surface finish properties which have paramount importance for surface-living system interactions such as surface chemistry, tension, roughness, wettability. We have taken PEEK filament polymer in this study for surface modification via wet-chemical, plasma & Diazonium chemical treatment. Nowadays plasma treatment (corona plasma) for surface modification of materials is being more popular to various fields for various applications for its easier & promising technique. We have described the modification of the polymer (PEEK filament) with the purpose of surface preparation to improve the adhesion between PEEK and silver particle also. First of all, the main target of this works is the modification through plasma than wet treatment by coating or metalizing by cationic Ag particle on high performance filament as PEEK surface. Finally, our targets are observation the effect of Acetic acid on modified PEEK surface, observation of filaments Surfaces via electro microscopic tester (SEM) and observation of Ag coated PEEK filaments effect on physical properties or observation of properties of Ag coated surface. Observation the wettability, surface free energy and roughness on the surface morphology of metallic filament conductivity or electrical resistance. Another approach has been included in this study which is modification of PEEK materials via Diaz onium chemistry and purpose of this is polymer surface (PEEK) modification for hydrophilic, biocompatible properties to use PEEK as biomaterial, electrical fields e.g. orthopedics implants, electrical circuits purposes.
Keywords: Plasma, Surface, PEEK, Biocompatibility
Cite this paper: Md. Rasel, Md. Shah Alam, Mohammad Tofayel Ahmed, Jarin Akter, Habibur Rahman Abir, Mohammad Sohel Rana, Surface Modification of High Performance Polymer and Analysis of Surface Properties. -A Brief Review Article, American Journal of Materials Science, Vol. 8 No. 1, 2018, pp. 1-5. doi: 10.5923/j.materials.20180801.01.
Article Outline
1. Introduction
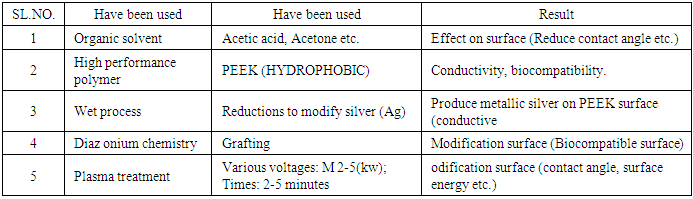
- High performance polymers are hydrophobic in nature and the main purpose of this study is modification of high performance polymer (PEEK) two different ways surface modification via plasma method & another is surface modification by Diazonium chemistry but this study includes biocompatibility, contact angle, conductivity & surface energy measurement. After surface modification PEEK surface is coated by Ag particle with the help of wet chemical process [1] where ionic silver is fixed on the surface of filaments then reduced to metallic silver. Solvents as Acetic acid has no significant effects on PEEK surfaces but acetone reduce the contact angle. On the other hand, plasma voltages have significant effects because with the increase of voltages compare to time it has effects on polymer surface properties e.g. conductivity, contact angle etc. We have been studied 2kw to 5kw voltages & 2-5min which reduced water contact angle & produced roughness also on the surface. Roughness or surface properties also have been investigated via Microscopic tester whereas conductivities measured by conductive tester machine, Water contact angle has measured using the sessile drop technique with a video-based contact angle measurement device. There are many techniques can be used to modify or alter the surface properties of materials by addition of particles; coatings and functional groups [2-7], pulsed vacuum arcs initiated on the surface of a consumable cathode [8-10], and laser [11] are liable to alter the surfaces also. It has been also developed to operate at near ambient temperature and atmospheric pressure eliminating the expensive vacuum systems [12, 13]. Most of polymers have outstanding physiochemical properties & easier to process depending on requirements of applications e.g. polymeric materials in recent years like surface modification of biomaterials [14-17] but due to the disadvantage of chemical treatments, physical surface treatments are preferred to modify the surface of polymeric materials [12] such as orthopedic and dental implants, heart valves, joint replacements, bone plate/cements, blood vessel prostheses, coronary stents, artificial ligaments/tendons, skin replacements and contact lenses [18]. whereas silver coated PEEK possible to build complete switching circuits to construct textile- based sensors and to produce self-luminous textiles where textile solar cells are under way [19]. Metallic modified materials e.g. E-textiles, intelligent clothing is the part of development of wearable technology but enable to compute digital components and electronics embedded because these are allowed technological elements in everyday textiles and clothes [20]. Therefore it depends on the physical or mechanical properties (such as viscosity, hardness), permeability, optical, tensile/compression strength, properties, corrosion resistance, wear resistance, elastic modulus, process ability, fatigue resistance, weight and costs [21] e.g. (PEEK) as hard material for orthopedic application [22]. To treat the surfaces by Plasma is often preferred way as it offers longer lasting and more stable surface energy increment than any other treatments [13]. Therefore, atmospheric plasma treatment has been employed to modify the polymer and composite surfaces. To enhance the efficiency of cell production and maintenance.PEEK is Non-biodegradable polymers but only limit its inertness, due to inertness no adhesion occurs with bone tissue but perfect to substitute load bearing metallic orthopedic implants for biomedical applications. Diaz onium chemistry methods which can modify the plastic used in biomedical applications e.g., cell culture plates to.
2. Experimental Parts
- In these experimental parts we have been studied contact angle, surface free energy, conductivity etc. via various techniques and mentioned discussion part.
2.1. Material
- PEEK is a semi-crystalline thermoplastic polymer [23] possessing the service temperature -250°C to +300°C, tensile strength 90 to 120 MPa [24] and elongation of 16% to 80% [25] for that it can be applied in textiles, medical, aerospace and automotive etc. industries also. It’s hydrophobic & has chemical structure which containing aryl rings and interconnected via ketene and ether groups located at opposite ends of the rings. It has excellent thermal, chemical & mechanical properties. PEEK dimensions 20x20x1.5 mm have been prepared by cutting from a square shaped PEEK bar (2x2cm) by using an IsoMet 5000 linear precision saw (BUEHLER, Whit by, ON, Canada) [26].
2.2. Methods
- Plasma is a treatment that happened with current flows from an electrode with a high potential into a neutral fluid or air, via ionizing that fluid for creation a region of plasma around the electrode. On the other hand, wet- chemical process has been produced metallic surface on the PEEK filament polymer surface via various chemical reactions such as AgNO3 + 2NH4OH [Ag (NH3)2] +NO3- + 2H2O (1) then ionic silver are reduced to metallic silver 2Ag+ + C6H8O6 2AgO + C6H6O6+ 2H+ [27]. Plasma is very popular methods for modification surfaces of materials by containing various reactive groups, ions & radicals, electrons but functional groups also formed. On the other hand, reactions of polymer chain degradation occur on polymer surfaces and produces ions by generation high kinetic energies from electrical field in the plasma but functional groups & degrading reactions of polymer chain happened on the surfaces when irradiated with plasma because ions always present with radicals in plasma [28]. The surface treatment on PEEK improves adhesion properties which is attributed due to the formation of polar groups on the polymer surfaces [29]. Hopefully the polymer (PEEK) filament surfaces has been become more hydrophilic & wet ability after modification. Diaz onium chemistry which is able to graft organic compounds to the surface & this technique has been used to bender many different groups to materials surfaces; General chemical structure of aryl Diaz onium salt is R−N2 X, R stands for a variety of functional groups, which are organic or organometallic substituents, and X is the counter anion groups including halides, carboxylic acid groups, nitro and alkyls [30], redox species [31], even dendrimers [32], and per fluorinated chains [33].
|
3. Result & Discussion
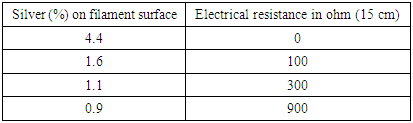
- Acetic acid has been used to modify PEEK filaments to increase adhesion between the surface of filaments & silver (Ag) which has been done at room temperature and near about 18/20minites then washed by fresh water & finally has been dried at room temperature. It shows that there is no effect of plasma treatment on breaking force & elongation at various Kw for 2/5 minutes on both treated & untreated PEEK filaments surfaces with acetic acid. So, less influence on tensile properties of modified surfaces filaments also.Through SEM tester has been investigated both single and monofilament & multifilament for determination plasma treatment effects on these filaments surfaces & to determinate water contact angle on surfaces. We have been studied to finding out the contact angle of water on materials surfaces by SEM but failed to take images. The SEM images clearly shows no obvious things on undesized PEEK multifilament surface but on the desized or modified surface shows uneven & rough surface compare to undersize. On the other hand, Monofilament PEEK undesized shows clear but cling on some particles to the surface. After plasma treatment via various powers it shows grooves & bulges to the surface of PEEK monofilaments also.Water Contact angle & surface energy have been studied from PEEK filament surfaces. Several samples (8-10) have been taken to investigate or finding out the surface properties. The finding of this study is acetone wash reduces the contact angle of water which also produces hydrophilic surfaces but this change is insignificant during acetic acid treatment. On the other hand, surface modification of PEEK filaments via atmospheric pressure plasma the contact angle decreases undoubtedly & resulting lower contact angle as surface become more oxidized & finally produces wetting properties. Plasma treatment with different power on undesized materials that boosting or increasing polar part & surface energies as polar part and dispersive parts including with surface free energy. Further plasma treatment has been done on desized materials but resulting less increased polar parts compare to undesized materials. Polar parts/dispersive parts/surface free energy can be increased by plasma treatment by changing power that means reduce water contact angle through this way. Water contact angle has measured using the sessile drop technique with a video- based contact angle measurement device (OCA 25, Future Digital) equipped with an auto dispenser. A sessile water droplet (30 µl) was placed on the surface of PEEK samples; the image of droplet was recorded after 30 seconds and the tangent angle at the point of contact between water, PEEK and air was measured by the software. The test was conducted on three spots on the surface of six samples for each condition [26].We have been investigated that surface modification of PEEK with Diaz onium chemistry methods & by this way has been strongly enhanced biocompatibility, hydrophilic which possible for grafting e.g. propionate groups on the surface of PEEK.
|
4. Conclusions
- Modified PEEK has been introduced having conductive & hydrophilic Properties (low water contact angle) by wet & plasma treatment. Coating gives good surface resistivity and improved mechanical or physical characteristics. Plasma treatment has developed the properties of the surface such as wettability or surface free energy, roughness but roughness caused no problem to get conductive surface and can be used in various applications e.g. E-textile or smart textile, medical textile etc. according to demand. Surface roughness has been analyzed by SEM on the filament surface morphology. Diaz onium chemistry produced biocompatible modified PEEK material surface for medical applications e.g. perfect to substitute load bearing metallic orthopedic implants for biomedical applications [26].
ACKNOWLEDGEMENTS
- We would like to thank Mr. A.B.M FAISAL, Senior lecturer of Department of Textile Engineering, Southeast University for his motivation, especially grateful to almighty ALLAH for helping us to complete this study.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML