-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Materials Science
p-ISSN: 2162-9382 e-ISSN: 2162-8424
2013; 3(5): 130-135
doi:10.5923/j.materials.20130305.04
The Manufacture of a Novel 3D Hydroxyapatite Microstructure Derived from Cuttlefish Bones for Potential Tissue Engineering Applications
Gérrard Eddy Jai Poinern, Derek Fawcett
Murdoch Applied Nanotechnology Research Group. Department of Physics, Energy Studies and Nanotechnology School of Engineering and Energy, Murdoch University, Murdoch, Western Australia 6150, Australia
Correspondence to: Gérrard Eddy Jai Poinern, Murdoch Applied Nanotechnology Research Group. Department of Physics, Energy Studies and Nanotechnology School of Engineering and Energy, Murdoch University, Murdoch, Western Australia 6150, Australia.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
Porous three-dimensional hydroxyapatite scaffolds were synthesised from aragonitic cuttlefish bones via a microwave based technique. Due to its close similarity to the mineral phase found in the natural human bone matrix, synthetically produced hydroxyapatite is considered a viable apatite substitute for natural bone in a number of biomedical applications. The X-ray diffraction data confirms the conversion of crystalline formations of calcium carbonate, the main component of the cuttlefish, to hydroxyapatite. The synthesis process is straightforward, efficient and creates a highly porous 3 dimensional microstructure that has the potential to be used in a wide range of hard tissue engineering applications such as bone repair, filling material and bone augmentation.
Keywords: Hydroxyapatite, Microstructure, Microwave Irradiation, Regenerative Medicine
Cite this paper: Gérrard Eddy Jai Poinern, Derek Fawcett, The Manufacture of a Novel 3D Hydroxyapatite Microstructure Derived from Cuttlefish Bones for Potential Tissue Engineering Applications, American Journal of Materials Science, Vol. 3 No. 5, 2013, pp. 130-135. doi: 10.5923/j.materials.20130305.04.
Article Outline
1. Introduction
- The human skeletal system is composed of a hard tissue called bone and more flexible cartilaginous materials which provide the structural support and movement capability of the body[1]. In the event of an accident, where the bone is fractured or damaged due to diseases, bone substitutes are needed to repair the fracture or replacement the infected bone tissue[2]. The demand for natural bone and bone substitutes is high. In Australia alone, the Australian Orthopaedic Association’s National Joint Replacement Registry recorded more than 75,000 hip and knee replacements being carried out annually[3]. This health issue is not unique to Australia and has been identified by World Health Organization as one of the most serious health problems facing the world today[4].In spite of being the most preferred option, with its excellent biocompatibility and osteogenic capacity, autografts have problems associated with donor site morbidity and limited supply[5, 6]. Given an adequate blood supply and no infection, autographs are successful donor tissues due to their identical nature. However, following the bone replacement procedure, patients receive immunosuppressive therapy which involves taking drugs designed to suppress the immune system. During the therapy, many of these drugs used tend to kill rapidly dividing cells and cause serious side effects. Therefore, to be a successful autograft procedure, right balance of immunosuppressive drugs and antibiotics to keep infection under control are needed to prevent implant site and donor morbidity. The autograft bone is usually harvested from the patient’s iliac crest in the form of cancellous bone. This means that the patient has only a limited number of potential harvest sites capable of supplying a suitable autograft. It is due to this limited supply that has led to the search for alternative sources of bone materials such as xenogenic and allogenic bone grafts. However, both of these replacement bone sources do solicit a significant detrimental response from the immune system. In spite of this allogenic bone grafts are routinely used due to the high clinical demand[7, 8]. Nevertheless, concerns regarding the immune response and the possibility of pathogen infection from bone sources have resulted in the search for alternative synthetic biocompatible materials have been investigated [9].Inorganic materials such as calcium phosphate ceramics, porous coralline, and calcium sulphate’s have all been investigated in the past and demonstrated both good biocompatibility and bioactivity properties[10-12]. One particular material that has been extensively studied for a variety of bone repair and replacement procedures is hydroxyapatite (HAP), which is a member of the calcium phosphate family[13-15]. The stoichiometric chemical formula of HAP is[Ca10(OH)2(PO4)6] and is highly biocompatible due to its close chemical similarity to the mineral component of natural bone[16]. There are four significant advantages of using HAP for bone tissue engineering: 1) it has been found to have good biocompatibility with surrounding body tissues; 2) it is highly stable under a variety of physiological conditions and its biodegradability in situ is slow; 3) it provides good osteoconductivity, and 4) it has good osteoinductivity capabilities[17-22]. It is due to these favourable advantages that has resulted in HAP being used as the major component in bone cements and is routinely used to coat titanium based medical implants[23]. In the case of metallic implants, the HAP coating improves bone fixation, bone in-growth and increases the life span of the implant in the body.This paper reports the rapid chemical conversion of a 3-D porous CaCO3 micrometre scale structure found in cuttlefish bone to a HAP structure. A wet chemical technique, incorporating microwave heating was used during the conversion process. The advantage of using microwave heating in this process arises from the volumetric distribution of energy throughout the material. This is fundamentally different to the conventional thermal conduction mechanism normally found in ovens and furnaces[24]. Unlike other hydrothermal based conversions of cuttlefish bone this microwave based conversion process is rapid in comparison[25, 26]. Thus, allowing a greater throughput of material and improving the overall efficiency of the microwave based conversion process. Advanced characterisation techniques such as x-ray diffraction (XRD) and field emission scanning electron microscopy (FESEM) were used to investigate the effectiveness of the conversion process. XRD was used to verify the chemical conversion of CaCO3 to HAP, while FESEM microscopy was used to confirm the integrity of the 3-D micrometre scale structure after processing. The results of the XRD and FESEM analysis indicate that a 3-D micrometre scale HAP structure is formed at the end of the conversion process.
2. Materials and Methods
2.1. Materials
- All analytical grade reagents used in this work were supplied by Chem-Supply (Australia) and the cuttlefish bone samples were collected from a local beach. The main reactants used to synthesis the nanometre sized HAP powder used for comparative analysis consisted of calcium nitrate tetrahydrate[Ca(NO3)2.4H2O] and potassium di-hydrogen phosphate[KH2PO4]. During the synthesis process the solution pH was maintained by the addition of ammonium hydroxide[NH4OH]. All aqueous solutions used throughout this study were made using Milli-Q® water (18.3 MΩ cm-1) produced by an ultrapure water system (Barnstead Ultrapure Water System D11931; Thermo Scientific, Dubuque, IA).
2.2. Preparation of HAP Powder Used for Comparison Purposes
- The synthesis of the HAP powder begins by adjusting the pH of a 40 mL solution of 0.32M calcium nitrate tetrahydrate to a value of 9.0 by adding and mixing approximately 2.5 mL of ammonium hydroxide. The solution was then exposed to ultrasound irradiation for 1 h using an UP50H Ultrasound Processor[50 W, 30 kHz, MS7 Sonotrode (7mm diameter, 80 mm length)] supplied by Hielscher Ultrasound Technology. During the second hour of ultrasonic irradiation a 60 mL solution of 0.19 M potassium di-hydrogen phosphate was slowly added drop-wise into the reactive mixture. Throughout the second hour the calcium:phosphate[Ca:P] ratio was maintained at 1.67 and the solution pH maintained at 9.0. At the end of the second hour, the solution was filtered using centrifugation (15,000g) for 20 minutes at room temperature. The resultant white precipitate was then transferred to a fused silica crucible ready for thermal treatment. The thermal treatment consisted of placing the crucible into an LG® (Australia) household microwave rated at 1100 W (2450 MHz) for 40 minutes. After cooling, the resulting white agglomerated mass was ground to form a fine powder and then characterised using XRD. A schematic presentation of the procedure is presented in Figure 1.
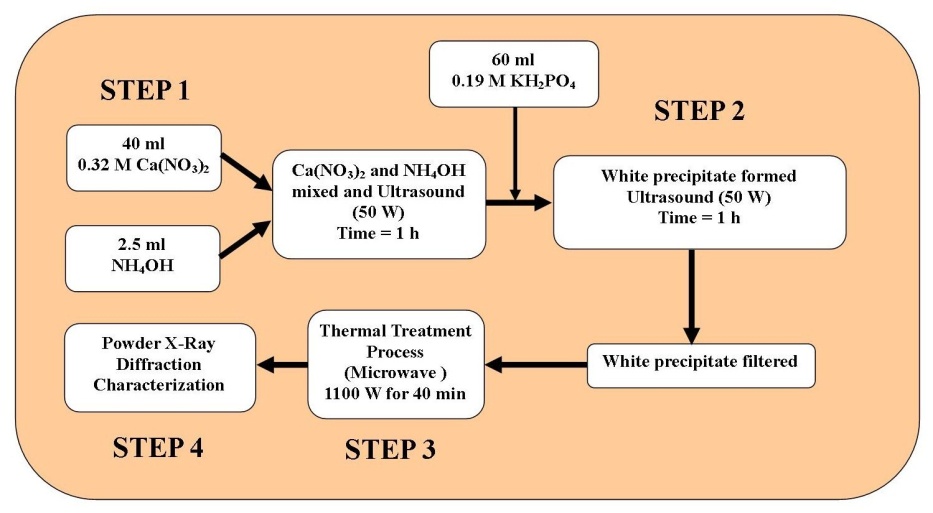 | Figure 1. Schematic of the synthesis procedure used to produce nanometre scale HAP powders used for comparative analysis |
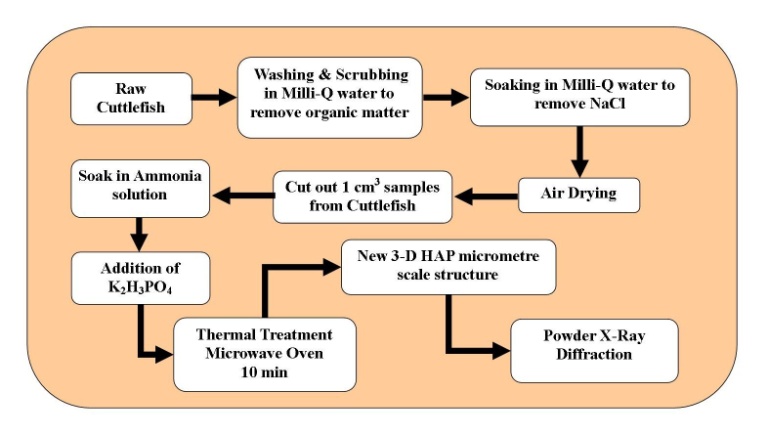 | Figure 2. Schematic of cuttlefish conversion process to produce a 3-D HAP micrometre scale scaffold structure. |
2.3. Microwave Based Conversion of Cuttlefish Samples
- The conversion process begins with a raw cuttlefish bone being washed and scrubbed to removal all traces of organic matter. In the next stage, the cuttlefish bone was soaked in a series of Milli-Q® water solutions to progressively remove any sea salt (NaCl) content. After the salt had been removed, the cuttlefish bone was given a final wash in Milli-Q® water before being allowed to air dry. The dried cuttlefish bone was then cut up into 1 cm2 portions, with the thickness of the portions being 1 cm for samples being used for conversion. Each sample then underwent a pre-treatment procedure that consisted of two steps. In the first step each sample was placed into a 30% solution of ammonia (Chem-Supply, Australia) and permitted to soak for 30 minutes. Following the initial soaking, a 0.19 M solution of K2H3PO4 was added to the solution. Then the solution containing the cuttlefish bone was then placed into a microwave bomb (45 ml, 4782 Parr Instrument Company) and sealed. The bomb was then placed into an LG® (Australia) household microwave oven rated at 1100 W (2450 MHz). The microwave was set to full power and the thermal treatment was carried out over 10 minutes. The conversion procedure is presented schematically in Figure 2 and images of the sample during processing are shown in Figure 3. After processing samples were examined using XRD and FESEM.
2.4. Characterisation of Materials
- Analysis of the XRD spectra was used to identify the HAP peaks in synthesised powders and examine the cuttlefish sample before and after conversion. The HAP powder pattern and the converted cuttlefish pattern were then compared and peak identification was carried out. XRD spectra were recorded at room temperature, using a Siemens D500 series diffractometer[Cu Kα = 1.5406 Å radiation source] operating at 40 kV and 30 mA.The diffraction patterns were collected over a 2θ range from 20° to 60° with an incremental step size of 0.04° using flat plane geometry. The acquisition time was set at 2 seconds for each scan. The morphological features of the converted cuttlefish samples were investigated using FESEM. All micrographs were taken using a high resolution FESEM[Zeiss 1555 VP-FESEM] at 3 kV with a 30 µm aperture operating under a pressure of 110-10 Torr. In addition, the FESEM micrographs were also used to estimate the size of major structure features seen in the images.
3. Results and Discussions
- The XRD technique was used to identify and confirm the presence of crystalline HAP both in the synthesised powder used for comparative purposes and the converted cuttlefish samples. A typical XRD pattern of the synthesised ultra fine HAP powder is presented in Figure 4 and reveals the main (h k l) indices associated with HAP: (002), (211), (300), (202), (130), (002), (222) and (213). The (002) reflection peak from the XRD pattern was used to calculate the crystallite size of the powder using the Debye-Scherrer equation (Insert Figure 4)[27, 28]. The crystallite size was estimated to have a mean value of 30 ± 5 nm. The HAP XRD spectra as then used to in a comparative study between the pre-treated cuttlefish and the converted cuttlefish.The cuttlefish sample spectrum (b) prior to treatment is presented in Figure 5 and reveals an XRD pattern with numerous peaks indicating the presence of a CaCO3 based structure. The post-treatment sample spectrum (c) reveals a pattern that is similar to the synthesized HAP sample spectra (a). The peaks in the left hand side of the pattern are dominant, can be easily seen and clearly indicate the presence of the (002), (211), (300) and (202) indices. In the right hand side of the spectrum the peaks are not as prominent, but can still be identified as the (130), (222), (213) and (004) indices. In the pre-treated sample the CaCO3 peaks on the right hand side of the spectrum were quite dominant. This is not the case after treatment where the peaks are significantly smaller. The results of the XRD study have clearly shown that the cuttlefish sample has been effectively converted into HAP during the treatment process.
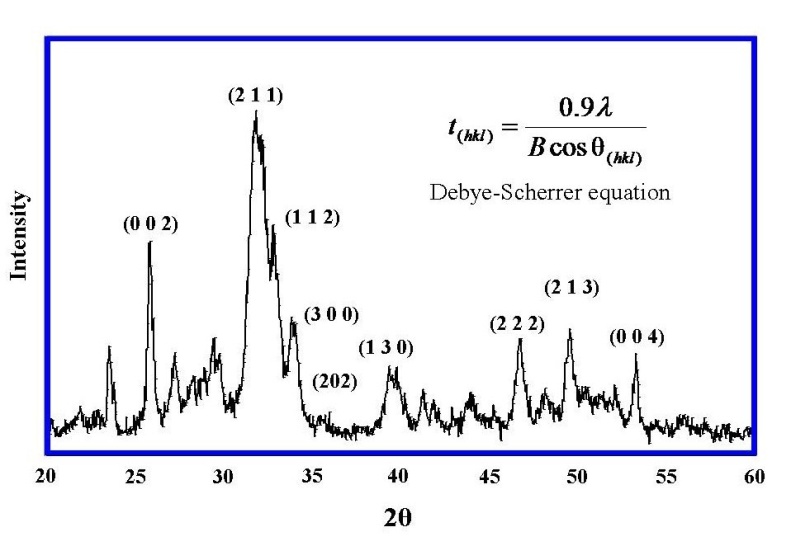 | Figure 4. A typical experimental XRD spectrum of the synthesized HAP powder used for comparative purposes. |
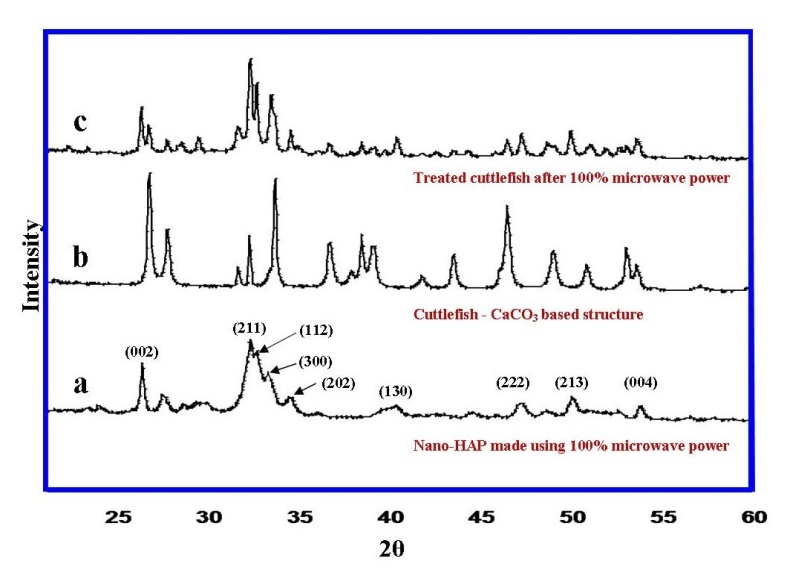 | Figure 5. Comparisons between the XRD spectra of synthesised HAP powder (a), pre-treated cuttlefish bone sample (b) and converted cuttlefish bone sample (c) |
 | Figure 6. A typical FESEM micrograph of the converted cuttlefish bone sample showing structural damage caused during the removal of organic material in the pre-treatment stage of processing |
4. Conclusions
- HAP is an important biocompatible material used in biomedical and tissue engineering applications. The results of this study have shown that the conversion of CaCO3 based cuttlefish bones to a 3-D HAP micrometre scale structure is simple and straightforward. The resultant HAP material resembles the mineral phase found in natural bone tissue, with the advantage of being modelled into a definite shape for a specific application. This highly porous HAP structure has the potential to be used as a cell scaffold for tissue engineering applications.
ACKNOWLEDGEMENTS
- The authors would also like to thank Mr R. Brundavanam for his assistance with the X-ray diffraction investigation.
Disclosure
- The authors report no conflict of interest in this work.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML