-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Materials Science
p-ISSN: 2162-9382 e-ISSN: 2162-8424
2012; 2(2): 28-33
doi: 10.5923/j.materials.20120202.06
On the Primary Phase Microstructure of Solidifying Ti-46Al-8Nb Refractory Intermetallic Alloy
A. V. Kartavykh
Dept. for Functional and Structural Nanomaterials, Technological Institute for Superhard and Novel Carbon Materials (TISNCM), Troitsk, 142190, Russia
Correspondence to: A. V. Kartavykh , Dept. for Functional and Structural Nanomaterials, Technological Institute for Superhard and Novel Carbon Materials (TISNCM), Troitsk, 142190, Russia.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
The reported contradictory data on microstructure formation of the refractory intermetallic Ti–46Al–8Nb (at.%) alloy and on the high-temperature phase transformations proceeding within the Ti–Al–Nb phase diagram are analysed and clarified experimentally. To determine the primary solidifying phase, a set of experiments is performed on melting of the alloy specimens with low oxygen contamination in a high purity argon atmosphere using crucibles made of oxygen-free ceramics (99.99% AlN), and subsequent rapid solidification. Volumetrically-isothermal cooling from 1943 K at rates of 5, 10, and 20 K/s and following quench of mini-ingots from 1763 K are used. Specimens were studied by scanning electron microscopy (SEM) in backscattered electron (BSE) mode. SEM-BSE micrographs demonstrate contrasting shadow regions of non-uniform niobium segregation, which are fixed by quench and decorate the primary polycrystalline microstructure formed within the temperature range between 1843 (liquidus) and 1773 K (solidus). The primary crystallizing phase is proven to be represented by β(Ti) dendrites, which have clearly pronounced fourfold (bcc-lattice) symmetry being formed with secondary dendrite arms development. The solidification path is shown to be described with single- phase scheme L→L+β(Ti)→β(Ti); no peritectic β(Ti)→α(Ti) bcc-hcp phase transformation revealed within the mushy state of alloy.
Keywords: Tial-Based Intermetallics, Phase Diagrams, Solidification, Microstructure, Electron Microscopy (SEM)
Article Outline
1. Introduction
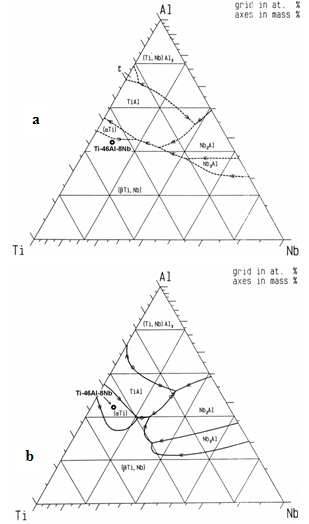
- The lightweight intermetallic Ti–46Al–8Nb (at.%) alloy is a new advanced material for the application in constructions and items operating under extreme high temperature and high pressure gas-plasma medium conditions. In particular in the EU countries, the alloy has received a major attention in developing pilot technologies of turbine blades manufacturing for gas-burning power plants and aero-engines within IMPRESS Integrated Project[1]. In contrast to most of TiAl-based alloys, the Ti–46Al–8Nb is characterized by the improved combination of ductility and high-temperature impact strength that is reached via the formation of a special phase polycrystalline microstructure of ingot or functional cast item[2]. The purposeful formation of a reproducible balanced dual-phase γ+α2 structure, which is additionally strengthened with niobium alloying and characterized by optimum dimensional and orientational grain parameters, is rather difficult problem[3,4]. During casting and cooling, the alloy undergoes successive multistage structural and phase transformations (including those in solid state), which are reflected in its complex phase diagram. Purposeful microstructural engineering of Ti–46Al–8Nb alloy requires reliable temperature-phase state diagrams, at least, for close chemical composition ranges. However, there are a number of substantially different high-temperature Ti–Al–Nb triple phase diagrams, even in the reputable specialized handbooks (see, e.g.[5]). It follows from these diagrams that both the β, and α phases could be the primary crystalline phase for the Ti–46Al–8Nb (at.%) composition (Figure. 1). It is highly crucial point, since these phases have different crystal lattices (bcc and hcp ones[6], respectively) and, therefore, they form the primary polycrystalline grains differing in sizes and symmetry. It is likely that one of the causes for the difference in the reported data is the uncontrollable dissolved oxygen content in the TiAl-based alloys used for the experimental phase diagram evaluations. According to[7,8], interstitial oxygen substantially affects the composition and structure of the primary solid phase in TiAl intermetallics whose compositions are close to equiatomic with respect to the Ti/Al ratio. To determine the fundamental solidification behaviour of the Ti–46Al–8Nb alloy more exactly, experiments should be performed under high purity conditions at reproducible temperature regimes using initial material minimally contaminated with oxygen. A primary microstructure cannot be revealed in industrially cast alloy owing to the diffusion averaging of the component concentrations and deep solid- phase transformations occurring in massive cast items under slow cooling conditions. The problem can be solved by applying a special laboratory test furnace (stand) that allows performing the processes of controlled rapid solidification and subsequent quenching of small sample from temperature slightly lower then solidus (to fix the structure). According to recent data of[9], the distribution coefficients of niobium and aluminum in Ti–46Al system at the liquidus temperature are 1.154 and 0.923, respectively. Therefore, the primary solid phase and inter-crystalline melt in mushy state should be enriched in niobium and aluminum, respectively. Areas characterized by a high niobium concentration exhibit a bright contrast in scanning electron microscopy (SEM-BSE) micrographs. Thus, the nature of decorated primary dendrites can be determined visually taking into account their symmetry, because the β and α phases have four- and six-fold symmetry each, respectively. This approach was used in the present study aimed at experimental determination of the fundamental nature of the primary melt grown solid phase in the refractory Ti–46Al–8Nb alloy in order to eliminate the contradiction of the data on the phase transformations and structure formation during solidification and subsequent heat treatment.
2. Experimental
- The pilot batch of intermetallic alloy was produced by GfE Metalle und Materialien GmbH (Germany, Nuremberg, http://www.gfe.com) via direct synthesis using initial components and an induction furnace with a “cold” copper crucible, and subsequent triple homogenizing arc remelting (hereinafter, initial alloy GfE). In accordance with the manufacturer certificate, the oxygen content in the alloy is 520 wt.ppm and the real alloy composition corresponds to the 45.78Ti–46.35Al–7.87Nb (at.%).Our experiments were performed using a boat crucibles 2 cm3 in volume manufactured of AlN 99.99% purity by GIREDMET Ltd. (). The technique of TiAl-based intermetallics solidification/casting in AlN non-oxide crucibles/moulds to decrease the interstitial oxygen contamination in ingots was suggested, developed, and firstly performed in[10, 11] and was patented by us in[12]. We used technical argon subjected to triple purification, namely, drying of the gas in a silica gel column and the absorption of O2 and CO2 with zeolite filters. This gas refining procedure is a standard technology used in the semiconductor industry; the Ar purity was checked by the dew point of the gas; the latter did not exceed –70℃.
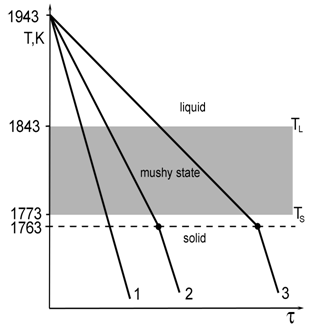 | Figure 2. Schematic of experimental solidification/quench thermal cycles for specimens 1, 2 and 3 of Ti–46Al–8Nb (at.%) alloy |
3. Results and Discussion
- Figures 3a–3c show SEM-BSE micrographs taken from the polished sections of experimental Ti–46Al–8Nb specimens. The use of a progressively decreasing cooling rate dT/dτ and the decorating Nb-alloying effect allowed us to trace the dynamics of the nucleation and morphological development of primary phase polycrystals. The liquidus (TL) and solidus (TS) temperatures of the alloy under study are known to be 1843 and 1773 K, respectively[14]. This allows us to estimate the solidification time τ for each experiment. When cooling sample 1 at the rate dT/dτ = 20 K/s (τ = 3.5s), only an equiaxed granular primary microstructure characterized by grain sizes of 80–100μm (Figure. 3a) has time to form as a result of multiple volumetric nucleation of the solid phase; no habit of grains has been formed yet. When cooling sample 2 at the rate dT/dτ = 10 K/s (τ = 7s), a dendritic primary phase structure forms as a result of competitive growth of polycrystalline grains; therewith, some dendrites even have the developed secondary arms (see Figure. 3b). Further morphological development of dendrites is observed in sample 3, where dT/dτ = 5 K/s. In this case (τ = 14s), the most clearly pronounced dendritic microstructure forms that characterized by a high degree of fourfold symmetry with developed arms of the second and even the third order (see Figure. 3c). Since a dendritic crystal grows preferably along the thermal gradient direction, the most symmetric and developed dendrites, being characterized by a minimum deviation of cross-arms angle from 90°, form at the volumetrically-isothermal cooling conditions applied in the laboratory benchmark furnace.
 | Figure 4. Idealistic theoretical shape of a dendrite solidified with the bcc β(Ti) lattice in accordance with the simulation data[15] (for comparison with the real dendrite shown in Figure. 3c) |
 | Figure 6. SEM-BSE micrograph of α(Ti)-phase dendrite nucleated on TiB2 seed micro-particle (the latter is seen at the center of dendrite) during solidification of Ti–49Al–1B (at.%) alloy[18] |
4. Conclusions
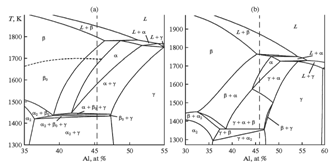
- (1) As a result of targeted experiments on controlled solidification and subsequent quenching of high-purity specimens and SEM studies, the nature of the primary crystalline phase in the intermetallic Ti–46Al–8Nb (at.%) alloy was determined. It is shown that β(Ti) dendrites, which have clearly pronounced fourfold symmetry, is the primary solid phase that forms within the liquidus–solidus temperature range.(2) The solidification path of the alloy is shown to be described with single-phase scheme L→L+β(Ti)→β(Ti); no peritectic L→L+β(Ti)→L+β(Ti)+α(Ti) bcc-hcp phase transformation was revealed within the mushy state.(3) In accordance with the experimental results, the phase diagram calculated by Witusiewicz et al. in Ref.[16] was selected from conflicting reported data. At present, it most correctly reflects the sequence of high-temperature phase transformations during solidification and successive cooling of the alloy under study.
ACKNOWLEDGEMENTS
- This work was performed in terms of Federal program “Research and scientific-pedagogical brainpower of innovated Russia 2009–2013” (State contract no. 02.740.11.0505) and was supported by the Russian Foundation for Basic Research (grants 10-03-00338 and 12-01-97514).
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-Text HTML
Full-Text HTML