-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Materials Science
p-ISSN: 2162-9382 e-ISSN: 2162-8424
2012; 2(2): 1-5
doi: 10.5923/j.materials.20120202.01
Natural Rubber/ Organoclay Nanocomposite from Tea (Camellia Sinensis) Seed Oil Derivative
L. E Yahaya 1, K. O Adebowale 2, A. R. R Menon 3, B. I Olu-Owolabi 2
1Cocoa Research Institute of Nigeria, Ibadan, Nigeria.
2Chemistry Department, University of Ibadan, Nigeria
3National Institute for Interdisciplinary Science and Technology, Trivandrum, India
Correspondence to: L. E Yahaya , Cocoa Research Institute of Nigeria, Ibadan, Nigeria..
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
Sodium salt of tea seed oil (TSO-Na) was organomodified with kaolin (China clay). The organo-kaolin was characterized by determining the FTIR and XRD. Results indicated that TSO-Na was successfully intercalated onto the kaolin surface. Nanocomposite of rubber copolymer with tea seed oil organomodified kaolin was prepared by incorporating 5phr organo-kaolin. The mechanical testing of the rubber vulcanizate involved the determination of tensile and tear property. The performance characteristics showed that there was considerable improvement in the tensile strength, tear strength and elongation at break properties of the organoclay filled vulcanizate compared to the unmodified polymer. Scanning electron microscopy (SEM) studies revealed that the incorporation of 5phr organoclay has remarkable effect on the failure mechanism of the NR filled vulcanizate. This is an indication that the failure mechanism is strongly dependent on the reinforcement effect of the modified filler marked by a higher extent of rubber-filler matrix interaction. There is therefore indication that tea seed oil can be organomodified with kaolin to serve as nanofiller.
Keywords: Tea Seed Oil, Natural Rubber, Organoclay, Nanofiller, Modulus, Vulcanizate
Article Outline
1. Introduction
- Polymer nanocomposites have attracted much attention in recent times. This is because of their improved mechanical, thermal, barrier as well as flame retardant properties at very low dosage loading of fillers (1, 2). Polymer/organoclay nanocomposites have emerged as a new class of material for reinforcing polymers. These are materials in which the particle size of the dispersed phase was nanometer sized at least in one dimension. Organoclay is a filler containing layered silicate that are occupied by ions such as Ca+, Al+3, or Na+ and this are usually replaced by organic cations such as alkyl ammonium cation through ion exchange reaction to obtain surfaces that are hydrophobic (3, 4).Ordinarily, clays have been used as filler in polymer reinforcement, but for better improvement in compatibility of polymers and organoclay layers which consequently leads to increased interlayer spaced, the clay for the nanocomposite need to be organomodified. These organoclays as reinforcing materials have found wide use in various polymers such as in plastics, polypropylene (5, 6), polystyrene (7), polyethylene terepthalate (8) and the rubbers (3, 9-14).The interlayer cations are usually replaced by alkyl ammonium cations , thus making the layered silicate organophillic. The large onium cation increases the initial interlayer distance. The interlayer distance of commercially available montmorillonite (bentonite), the commonly used filler is between 1.2-2.5nm and this distance is usually enough to produce intercalated and exfoliated polymer structures. Clays can thus be made organophillic by reacting their hydroxyl groups with organic silane. Literature search on organomodified filler-rubber nanocomposites revealed that montmorillonite clays have been extensively studied as the ideal mineral for the preparation of organomodified filers.As the natural deposits of bentonites are rather limited and the demand for same goes ever increasing, there has been considerable investigation for finding substitute for this type of product. In this regard, there is dearth of data on the relatively, inexpensive and abundantly available mineral kaolin (15). Furthermore, the use of organomodifiers such as octyl amine, octyldecylammine, dodecyl ammine, dimethyl benzyl dehydrogenated tallow, quaternary ammonium etc have been reported. Many of these are rather exorbitant in price because of the depleting nature of there petrochemical source (16), there is need therefore to look inward for alternate source. In this respect, derivatives of fatty acids based on low cost, renewable natural resources as organomodifier for silicate fillers need special mention.Tea seed oil (TSO) is a by product of camellia cultivation and is a rich source of oil that can be employed in this respect. This study therefore reports the use of Tea (camellia sinensis) seed oil derivative as a modifier of kaolin and the effect of the modified filler on the mechanical properties of natural rubber.
2. Materials and Methods
2.1. Materials
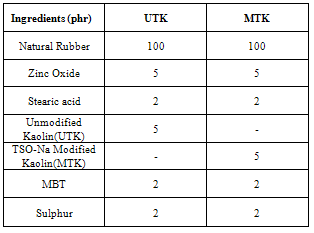
- Tea seed oil (TSO) was obtained from the Mambilla substation of the Cocoa Research Institute of Nigeria. Kaolin, grade BCK was obtained from M/s English, India clays Ltd, Veli, Thiruvananthapuram, Kerala, India. Natural rubber was of the Indian standard ribbed smoke sheet (grade RSSV); sodium hydroxide (LR Fischer), hydrazine hydrate (Aldrich), zinc oxide, stearic acid, sulphur and mercaptobenzothiazole (MBT) were obtained from the open market.
2.2. Preparation of Sodium Salt of TSO
- Sodium salt of tea seed oil (TSO-Na) was synthesized by reacting 28 ml of TSO with approximately 27.5 ml of 20 % NaOH in an ice bath with constant agitation for 12hours. The pH of the resulting solution was maintained at 8 - 9. TSO-Na was then introduced into a separating funnel and washed with water to remove excess base. This was then oven-dried to remove any moisture and powdered.
2.3. Modification of Kaolinite Surface
- 2 g of TSO-Na was treated with 9.8g kaolin, 7 ml of hydrazine and 50 ml water under vigorous agitation at 20 oC. The mixture was homogenized using an ultra Schallprozessor(HIELSCHER, GMbH, UP 100 H) and the sample was dried using a freeze drier (HetroTrap-CT600e, JOUAN).The sample was powdered and labeled.
2.4. Characterization of Tea Seed Oil
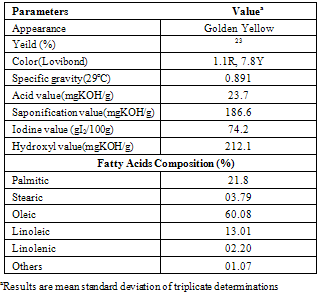
- The physicochemical properties and fatty acid profile of TSO was carried out according to IUPAC standard procedure.
2.5. Characterization of Unmodified and TSO-Na Modified Kaolin
2.5.1. FTIR
- The IR spectroscopy of both the modified and modified sample were.determined using the Fourier Transform Infra red spectrometer (MAGNA, 560, NICOLET). In a typical experiment, about 10mg of sample was thoroughly mixed with finely ground KBr and made into pellet to obtain a transparent disc.
2.5.2. XRD
- The X-ray diffraction of the samples was determined using a Philips -1710 X-ray diffractometer using monochromatic Ni-filtered Cu K alpha radiation 1.5418Ao at 40Kv and 20mA. All the readings were taken at room temperatures. The d-spacing was calculated using Braggs equation, nλ =2d sinθ , where λ is the wavelength of the monochromatic x-ray source, d is the spacing between two similar planes, θ is the angle at which x-ray falls on the sample, and n is the order of reflection.
2.6. Preparation of Natural Rubber Organo-Kaolin Mixes
- Natural rubber-organokaolin mixes of the samples were mixed on a two roll open mill for 5-10 min and the homogenized mixes were sheeted out according to the recipe in table 1.
|
2.7. Preparation of Vulcanizates
- Vulcanizate sheets of dimension 90 mm × 90 mm ×1.5 mm were prepared by compression molding of the mixes at 140 oC for 10 min on an electrically heated, semi-automatic laboratory press (model PF-A15) at 3torr pressure.
2.8. Measurement of Tensile and Tear Properties
- Dumb-bell shaped and crescent shaped samples were cut out of the molded sheet for tensile and tear strengths measurements respectively. The vulcanizates were measured according to standard ASTM D624-86 using a universal testing machine (Hounsfield H5 KS)
2.9. Scanning Electron Microscopy
- The fracture surface of the vulcanizates was studied with JEOL 5600LV scanning electron microscope. The fracture surface was coated with gold to prevent electrostatics charging during examination
3. Results and discussions
3.1. Characterization of Tea Seed Oil
- Table 2 shows the physicochemical characteristics and fatty acid profile of tea seed oil. From the table, it can be seen that tea seed oil possesses long hydrocarbon chain, C18 which is a requirement for its use as an organomodifier.
|
3.2. Characterization of Unmodified and Modified TSO-Na Kaolin
3.2.1. FTIR Spectroscopy
- The FTIR spectra of unmodified and TSO-Na modified kaolin are depicted in figures 1 and 2.The unmodified kaolin spectrum shows a band at 3620cm-1 and 3695cm-1 which are characteristics of inner hydroxyls and vibrations of outer surface hydroxyls respectively(17, 18), but for modified sample, there is the occurrence of bands at 2846cm-1 and 2919cm-1 showing the occurrence of intercalation of TSO-Na on kaolin surfaces (19).This intercalation process decreases the electrostatic attraction between the lamellae by causing an increase in the dielectric constant when the compound penetrate between the layers.
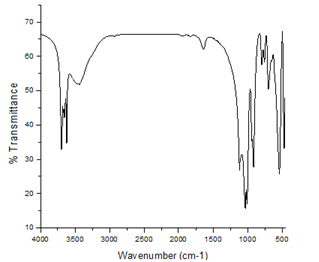 | Figure 1. FTIR Spectrum of Unmodified Kaolin |
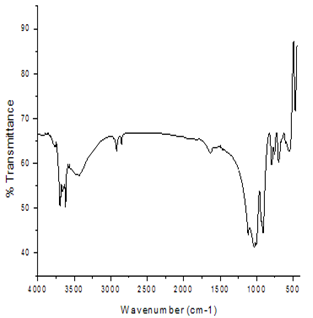 | Figure 2. FTIR Spectrum of TSO-Na Modified kaolin |
3.2.2. X-Ray Diffraction
- The XRD patterns of the neat kaolin and modified kaolin are shown in figures 3 and 4 respectively. The interlayer distance was determined from the diffraction peak position in the XRD using the Braggs equation. The basal distance of the neat kaolin and the interlayer distance of the organokaolin are respectively 7.15 and 14.6Å. It is thus evident that the intercalation of organic modifier enlarges the distance of the kaolin layers thus suggesting the successful preparation of organokaolin using tea seed oil derivative. The peak value of TSO-Na is greater than the d-spacing (13.89Å) reported for caesium acetate- kaolinites intercalate (17) and sodium montmorillonite (12.5-12.6Å) (20, 21).
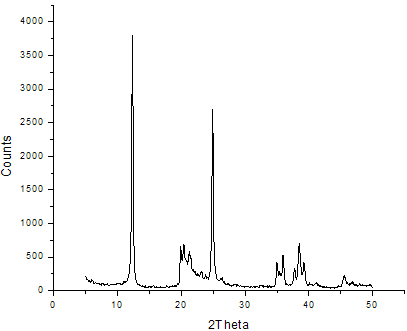 | Figure 3. XRD Spectrum of Unmodified Kaolin |
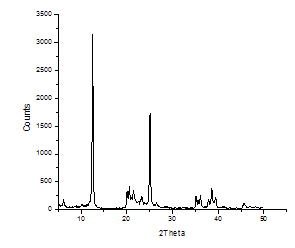 | Figure 4. XRD Spectrum of TSO-Na Modified Kaolin |
3.3. Mechanical Properties of Natural Rubber Vulcanizate
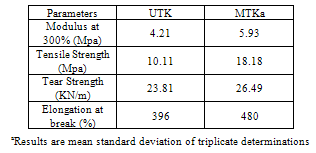
- Table 3: shows the mechanical properties of TSO-Na modified kaolin rubber vulcanizate compared with the unmodified sample. It is evident that MTK shows higher values of modulus (at 300% elongation), tensile strength and tear strength. The increased value of parameters can be related to the reinforcing effect of the filler. Similar increase in mechanical properties has been documented for various natural rubber nanocomposites containing some amount of organomodified clays (16, 21, 23, 24).The implication of this is that there is the possibility of higher degree of rubber-filler interaction resulting from the role of the organomodifier as a coupling agent between the duos.
|
3.4. Scanning Electron Microscopy
- Figure 5 (a) and (b) shows the SEM micrographs of the tensile fracture surfaces of the neat and organoclay filled Natural rubber vulcanizates respectively. The modified NR vulcanizate exhibits rougher fracture surface than the unmodified vulcanizate. This indicates that the fracture mechanism is strongly dependent on the reinforcement effect of the modified filler marked by a higher extent of rubber-filler interaction (3, 24). According to Costa et al., 2000, (23) the larger specific surface area and smaller particle size of organokaolin is believed to provide better rubber-filler interaction. This explains why organoclay filled vulcanizate shows better mechanical properties than unmodified vulcanizate.
 | Figure 5. SEM Micrograph of surface fracture of: (a) unmodified NR vulcanizate and (b) modified NR vulcanizate |
4. Conclusions
- Tea seed oil derivative was organomodified with kaolin (China clay). The organomodified clay was characterized by FTIR and XRD. Evidence indicated that TSO-Na was successfully intercalated with the clay. Nanocomposite of Natural rubber copolymer with TSO-Na modified kaolin was prepared. Incorporation of 5phr organoclay resulted in a significant enhancement of the mechanical properties of the natural rubber vulcanizate. Further evidence of successful dispersion of the organoclay in the NR vulcanizate was obtained from the scanning electron micrographs as this showed rougher fracture surface for the modified vulcanizate when compared with the unmodified.
ACKNOWLEDGEMENTS
- One of the authors (LEY) is thankful to the Third World Academy of Science, (TWAS) Trieste, Italy for providing the fellowship and the Council for Scientific and Industrial Research (CSIR) India for providing the facilities for this work. Many thanks to the management of Cocoa Research Institute of Nigeria and the help of Messrs Chandran with the SEM measurements, Guruswamy with the XRD measurements.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-Text HTML
Full-Text HTML