-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Journal of Nuclear and Particle Physics
p-ISSN: 2167-6895 e-ISSN: 2167-6909
2012; 2(5): 119-125
doi: 10.5923/j.jnpp.20120205.03
Radioactive Tracer Technique in Characterization of Nuclear Grade Anion Exchange Resins Indion-102 and Indion GS-300
Pravin U. Singare
Department of Chemistry, Bhavan’s College, Munshi Nagar, Andheri (West), Mumbai, 400 058, India
Correspondence to: Pravin U. Singare , Department of Chemistry, Bhavan’s College, Munshi Nagar, Andheri (West), Mumbai, 400 058, India.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
In the present study, attempt is made to characterize two strongly basic nuclear grade anion exchange resins Indion-102 and Indion GS-300. The characterization of the resins is made based on kinetics study by application of radioactive tracer technique using 131I and 82Br isotopes. It is observed that the iodide ion-isotopic exchange reaction take place at a faster rate than bromide ion-isotopic exchange reaction which is mainly due to solvation effect. The results indicate that both the iodide and bromide ion-isotopic exchange reactions are greatly influenced by the ionic concentration and the exchange increases with increase in ionic concentration. However the same ion-isotopic exchange is observed to decrease with rise in temperature. It is also observed that specific reaction rate (min-1), amount of ion exchanged (mmol) and percentage of ions exchanged for Indion GS-300 resins are higher than that for Indion-102 resins under identical experimental conditions.
Keywords: Indion-102, Indion GS-300, Nuclear Grade Ion Exchange Resins, Anion Exchange Resins, Tracer Applications, 131I, 82 Br, Reaction Kinetics, Ion-Isotopic Exchange Reactions, Distribution Coefficient, Reaction Rate
Cite this paper: Pravin U. Singare , "Radioactive Tracer Technique in Characterization of Nuclear Grade Anion Exchange Resins Indion-102 and Indion GS-300", Journal of Nuclear and Particle Physics, Vol. 2 No. 5, 2012, pp. 119-125. doi: 10.5923/j.jnpp.20120205.03.
Article Outline
1. Introduction
- Ion exchange materials are insoluble polymeric substances containing loosely held ions which are exchanged with other ions in solutions when they come in contact with each other. These exchanges take place reversibly without any physical alteration to the ion exchange material. The synthetic resins are not only used for purifying water, but also for various other applications including separation of some rare earth elements. In the past decade inorganic ion exchange materials have emerged as an increasingly important replacement or complement for conventional organic ion exchange resins. However in number of cases, for specific physical and chemical reasons, organic resins cannot be replaced by inorganic ion exchangers[1-3]. Also, in nuclear power plant operations the currently available inorganic exchangers cannot entirely replace conventional organic ion exchange resins, especially in high purity water applications or in operations in which the system chemistry must be controlled through the maintenance of dissolved species such as lithium ions or boric acid[3]. Nuclear power plant process water systems have typically used organic ion exchange resins to control system chemistry, to minimize corrosion or the degradation of system components and to remove radioactive contaminants. Organic resins are also used in a number of chemical decontamination or cleaning processes for the regeneration of process water by reagents and for radionuclide removal[1, 2]. Organic ion exchange resins has been developed over a much longer period of time than the selective inorganic ion exchangers that are now becoming available in commercial quantities and they can now meet the demands of the nuclear industry. The organic ion exchange resins are the wave of the present research and are considered as the material of next generation. For proper selection of ion exchange resin, it is essential to have adequate knowledge regarding their physical and chemical properties, which forms the complementary part of resin characterization study. Generally the selected ion exchange materials must be compatible with the chemical nature of the liquid waste such as pH, type of ionic species present as well as the operating parameters, in particular temperature[3]. Hence in the present investigation, it is proposed to test the performance of industrial grade ion exchange resin under different experimental conditions like temperature and concentration of ionic species present in the external exchanging medium.
2. Experimental
2.1. Conditioning of Ion Exchange Resins
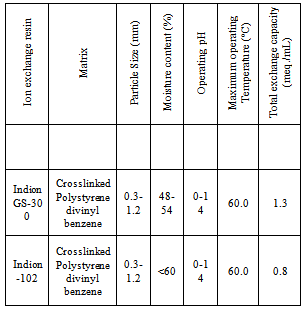
- Ion exchange resin Indion GS-300 and Indion-102 (Ion Exchange India Ltd., Mumbai) are nuclear grade strongly basic anion exchange resin in hydroxide form having quaternary ammonium -N+R3 functional group. Details regarding the properties of the resins used are given in Table 1. These resins were converted separately in to iodide / bromide form by treatment with 10 % KI / KBr solution in a conditioning column which is adjusted at the flow rate as 1 mL / min. The resins were then washed with double distilled water, until the washings were free from iodide/bromide ions as tested by AgNO3 solution. These resins in bromide and iodide form were then dried separately over P2O5 in desiccators at room temperature.
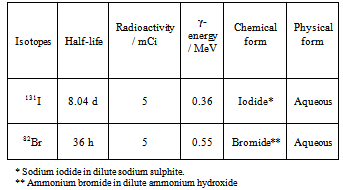
2.2. Radioactive Tracer Isotopes
|
|
2.3. Study on Kinetics of Iodide Ion-isotopic Exchange Reaction
- In a stoppered bottle 250 mL (V) of 0.001 M iodide ion solution was labeled with diluted 131I radioactive solution using a micro syringe, such that 1.0 mL of labeled solution has a radioactivity of around 15,000 cpm (counts per minute) when measured with γ -ray spectrometer having NaI (Tl) scintillation detector. Since only about 50–100 μL of the radioactive iodide ion solution was required for labeling the solution, its concentration will remain unchanged, which was further confirmed by potentiometer titration against AgNO3 solution. The above labeled solution of known initial activity (Ai) was kept in a thermostat adjusted to 30.0℃. The swelled and conditioned dry ion exchange resins in iodide form weighing exactly 1.000 g (m) were transferred quickly into this labeled solution which was vigorously stirred by using mechanical stirrer and the activity in cpm of 1.0 mL of solution was measured. The solution was transferred back to the same bottle containing labeled solution after measuring activity. The iodide ion-isotopic exchange reaction can be represented as:
 | (1) |
2.4. Study on Kinetics of Bromide Ion-Isotopic Exchange Reaction
- The experiment was also performed to study the kinetics of bromide ion- isotopic exchange reaction by equilibrating 1.000 g of ion exchange resin in bromide form with labeled bromide ion solution in the same concentration and temperature range as above. The labeling of bromide ion solution was done by using 82Br as a radioactive tracer isotope for which the same procedure as explained above was followed. The bromide ion-isotopic exchange reaction can be represented as:
 | (2) |
3. Results and Discussion
|
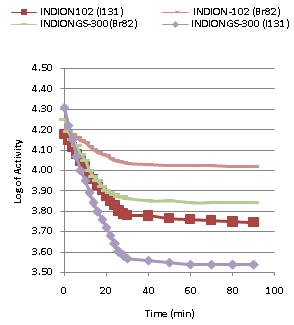 | Figure 1. Kinetics of Ion-Isotopic Exchange Reactions Amount of ion exchange resin = 1.000 g, Concentration of labeled exchangeable ionic solution = 0.001M, Volume of labeled ionic solution = 250 mL, Temperature = 30.0℃ |
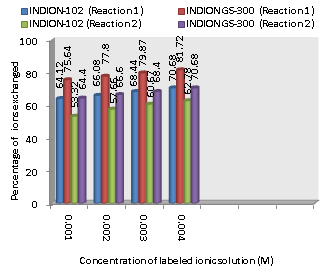 | Figure 2. Variation in Percentage Ions Exchanged with Concentration of Labeled Ionic Solution Amount of ion exchange resin = 1.000 g, Volume of labeled ionic solution = 250 mL, Temperature = 30.0℃ |
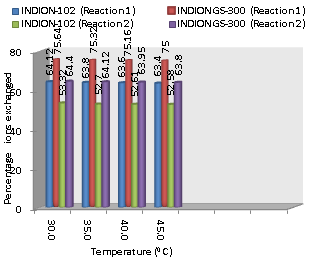 | Figure 3. Variation in Percentage Ions Exchanged with Temperature of Labeled Ionic Solution Amount of ion exchange resin = 1.000 g, Concentration of labeled exchangeable ionic solution = 0.001M, Volume of labeled ionic solution = 250 mL, Amount of exchangeable ions in 250 mL labeled solution = 0.250 mmol |
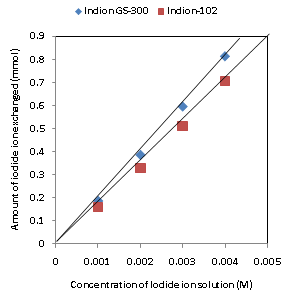 | Figure 4. Correlation between concentrations of iodide ion solution and amount of iodide ion exchanged Amount of ion exchange resin = 1.000 g, Volume of labeled ionic solution = 250 mL, Temperature = 30.0℃ Correlation coefficient (r) for Indion-102 = 0.9996 Correlation coefficient (r) for Indion GS-300= 0.9998 |
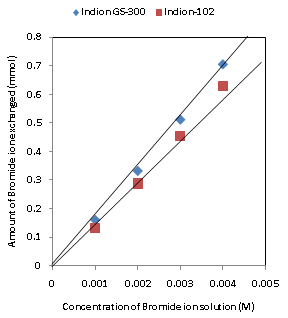 | Figure 5. Correlation between concentrations of bromide ion solution and amount of bromide ion exchanged Amount of ion exchange resin = 1.000 g, Volume of labeled ionic solution = 250 mL, Temperature = 30.0℃ Correlation coefficient (r) for Indion-102 = 0.9997 Correlation coefficient (r) for Indion GS-300= 0.9996 |
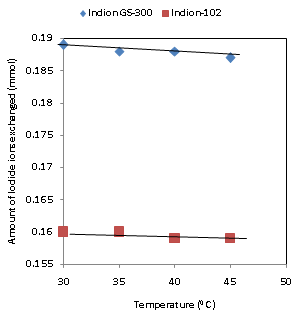 | Figure 6. Correlation between Temperatures of exchanging medium and amount of iodide ion exchanged Amount of ion exchange resin = 1.000 g, Concentration of labeled exchangeable ionic solution = 0.001M, Volume of labeled ionic solution = 250 mL, Amount of exchangeable ions in 250 mL labeled solution = 0.250 mmol Correlation coefficient (r) for Indion-102 = -0.8944 Correlation coefficient (r) for Indion GS-300= -0.9487 |
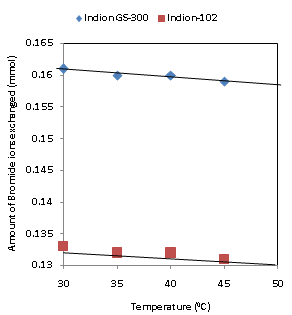 | Figure 7. Correlation between Temperatures of exchanging medium and amount of bromide ion exchanged Amount of ion exchange resin = 1.000 g, Concentration of labeled exchangeable ionic solution = 0.001M, Volume of labeled ionic solution = 250 mL, Amount of exchangeable ions in 250 mL labeled solution = 0.250 mmol Correlation coefficient (r) for Indion-102 = -0.9487 Correlation coefficient (r) for Indion GS-300= -0.9487 |
3.1. Comparative Study of Ion-isotopic Exchange Reactions
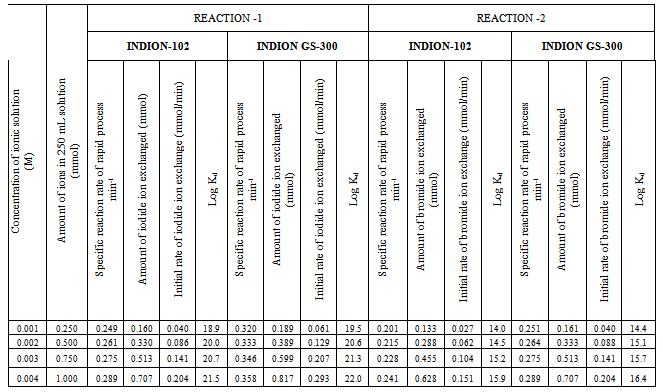
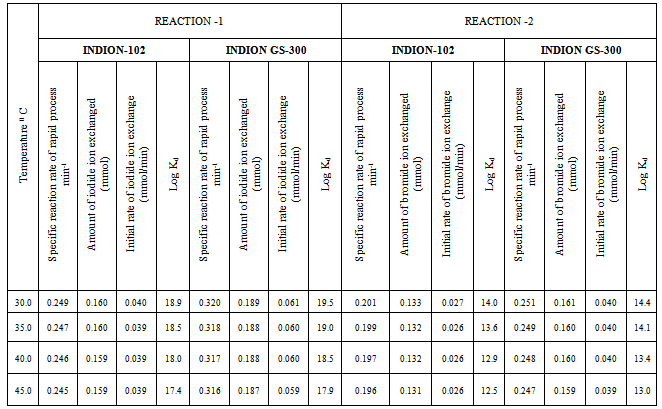
- In the present investigation it was observed that due to the rapid ion-isotopic exchange reaction taking place, the activity of solution decreases rapidly initially, then due to the slow exchange the activity of the solution decreases slowly and finally remains nearly constant. Preliminary studies show that the above exchange reactions are of first order[12, 13]. Therefore logarithm of activity when plotted against time gives a composite curve in which the activity initially decreases sharply and thereafter very slowly giving nearly straight line (Figure 1), evidently rapid and slow ion-isotopic exchange reactions were occurring simultaneously[5-11]. Now the straight line was extrapolated back to zero time. The extrapolated portion represents the contribution of slow process to the total activity which now includes rapid process also. The activity due to slow process was subtracted from the total activity at various time intervals. The difference gives the activity due to rapid process only. From the activity exchanged due to rapid process at various time intervals, the specific reaction rates (k) of rapid ion-isotopic exchange reaction were calculated. The amount of iodide / bromide ions exchanged (mmol) on the resin were obtained from the initial and final activity of solution and the amount of exchangeable ions in 250 mL of solution. From the amount of ions exchanged on the resin (mmol) and the specific reaction rates (min-1), the initial rate of ion exchanged (mmol/min) was calculated.Because of larger solvated size of bromide ions (310 pm) as compared to that of iodide ions (300 pm), it was observed that the exchange of bromide ions occurs at the slower rate than that of iodide ions[14]. Hence under identical experimental conditions, the values of specific reaction rate (min-1), amount of ion exchanged (mmol) and initial rate of ion exchange (mmol/min) are calculated to be lower for bromide ion-isotopic exchange reaction than that for iodide ion-isotopic exchange reaction as summarized in Tables 3 and 4. For both bromide and iodide ion-isotopic exchange reactions, under identical experimental conditions, the values of specific reaction rate increases with increase in concentration of ionic solution from 0.001M to 0.004M (Table 3). However, with rise in temperature from 30.0℃ to 45.0℃, the specific reaction rate was observed to decrease (Table 4). From the results, it appears that iodide ions exchange at the faster rate as compared to that of bromide ions which was related to the extent of solvation (Tables 3 and 4).From the knowledge of Ai, Af, volume of the exchangeable ionic solution (V) and mass of ion exchange resin (m), the Kd value was calculated by the equation
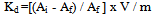 | (3) |
3.2. Comparative Study of Anion Exchange Resins
- From the Table 3, it is observed that for iodide ion-isotopic exchange reaction by using Indion GS-300 resin, the values of specific reaction rate (min-1), amount of iodide ion exchanged (mmol), initial rate of iodide ion exchange (mmol/min) and log Kd were 0.320, 0.189, 0.061 and 19.5 respectively, which was higher than 0.249, 0.160, 0.040 and 18.9 respectively as that obtained by using Indion-102 resins under identical experimental conditions of 30.0℃, 1.000 g of ion exchange resins and 0.001 M labeled iodide ion solution. The identical trend was observed for the two resins during bromide ion-isotopic exchange reaction.From Table 3, it is observed that using Indion GS-300 resins, at a constant temperature of 30.0℃, as the concentration of labeled iodide ion solution increases 0.001 M to 0.004 M, the percentage of ions exchanged increases from 75.64 % to 81.72 %. While using Indion-102 resins under identical experimental conditions the percentage of ions exchanged increases from 64.12 % to 70.68 %. Similarly in case of bromide ion-isotopic exchange reaction, the percentage of ions exchanged increases from 64.40 % to 70.68 % using Indion GS-300 resin, while for Indion-102 resin it increases from 53.32 % to 62.78 %. The effect of ionic concentration on percentage of ions exchanged is graphically represented in Figure 2.From Table 4, it is observed that using Indion GS-300 resins, for 0.001 M labeled iodide ion solution, as the temperature increases 30.0℃ to 45.0℃, the percentage of ions exchanged decreases from 75.64 % to 75.0 %. While using Indion-102 resins under identical experimental conditions the percentage of iodide ions exchanged decreases from 64.12 % to 63.40 %. Similarly in case of bromide ion-isotopic exchange reaction, the percentage of ions exchanged decreases from 64.40 % to 63.80 % using Indion GS-300 resin, while for Indion-102 resin it decreases from 53.32 % to 52.58 %. The effect of temperature on percentage of ions exchanged is graphically represented in Figure 3. The overall results indicate that under identical experimental conditions, as compared to Indion-102 resins, Indion GS-300 resins shows higher percentage of ions exchanged. Thus Indion GS-300 resins show superior performance than Indion-102 resins.
3.3. Statistical Correlations
- The results of present investigation show a strong positive linear co-relationship between amount of ions exchanged and concentration of ionic solution (Figures 4, 5). In case of iodide ion exchange using Indion GS-300 and Indion-102 resins, the values of correlation coefficient (r) were found to be 0.9998 and 0.9996, while for bromide ion exchange the values of r were 0.9996 and 0.9997 respectively. There also exist a strong negative co-relationship between amount of ions exchanged and temperature of exchanging medium (Figures 6, 7). For Indion GS-300 and Indion-102 resins, the respective values of r were found to be -0.9487 and -0.8944 during iodide ion exchange, and -0.9487 during bromide ion exchange for both the resins.
4. Conclusions
- The experimental work carried out in the present investigation will help to standardize the operational process parameters so as to improve the performance of selected nuclear grade ion exchange resins. The radioactive tracer technique as used in the present investigation can also be applied further for characterization of different nuclear as well as non-nuclear grade ion exchange resins.
ACKNOWLEDGEMENTS
- The author is thankful to Professor Dr. R.S. Lokhande for his valuable help and support in carrying out the experimental work in Radiochemistry Laboratory of Department of Chemistry, University of Mumbai, Vidyanagari, Mumbai -58.The author is extremely thankful to SAP Productions for developing and maintaining the manuscript template.
References
| [1] | International Atomic Energy Agency, 1967, Operation and Control of Ion exchange Processes for Treatment of Radioactive Wastes, Technical Report Series No. 78, IAEA, Vienna. |
| [2] | International Atomic Energy Agency, 1984, Treatment of Low and Intermediate-level Liquid Radioactive Wastes, Technical Report Series No. 236, IAEA, Vienna. |
| [3] | International Atomic Energy Agency, 2002, Application of Ion exchange processes for the treatment of Radioactive Waste and Management of Spent Ion Exchangers, Technical Report Series No.408, IAEA, Vienna. |
| [4] | Sood, D.D., 1998, Proc. Int. Conf. on Applications of Radioisotopes and Radiation in Industrial Development, ed. Sood, D.D., Reddy, A.V.R., Iyer, S.R.K., Gangadharan, S., and Singh, G., (B.A.R.C., India) 35–53. |
| [5] | Singare, P.U., and Lokhande, R.S., 2012, Studies on Ion-Isotopic Exchange Reactions Using Nuclear Grade Ion Exchange Resins., Ionics, 18(4), 351–357. |
| [6] | Singare,P.U., Lokhande, R.S., and Patil, A.B., 2008, Application of Radioactive Tracer Technique for Characterization of some Strongly Basic Anion Exchange Resins., Radiochim. Acta, 96(02), 99-104. |
| [7] | Lokhande, R.S., and Singare, P.U., 2008, Comparative Study on Iodide and Bromide Ion-Isotopic Exchange Reactions by Application of Radioactive Tracer Technique., J.Porous Mater., 15(03), 253-258. |
| [8] | Lokhande, R.S., Singare, P.U., and Patil, A.B., 2007, Application of Radioactive Tracer Technique on Industrial Grade Ion Exchange Resins Indion-830 (Type-1) and Indion-N-IP (Type-2)., Radiochim. Acta, 95(01), 111-114. |
| [9] | Lokhande, R.S., and P.U. Singare, 2007, Comparative Study on Ion-Isotopic Exchange Reaction Kinetics by Application of Tracer Technique., Radiochim. Acta, 95(03), 173-176. |
| [10] | Lokhande, R.S., Singare, P.U., and Kolte, A.R., 2007, Study on Kinetics and Mechanism of Ion-Isotopic Exchange Reaction Using Strongly Basic Anion Exchange Resins Duolite A-101 D and Duolite A-102 D., Radiochim. Acta, 95(10), 595-600. |
| [11] | Lokhande, R.S., Singare, P.U., and Dole, M.H., 2006, Comparative Study on Bromide and Iodide Ion-Isotopic Exchange Reactions Using Strongly Basic Anion Exchange Resin Duolite A-113., J.Nuclear and Radiochemical Sciences, 7(02), 29-32. |
| [12] | Lokhande, R.S., and Singare, P.U. 2003, Study of reversible ion-isotopic self diffusion reaction using 82 Br as a radioactive tracer isotope., Asian J. Chem., 15(1), 33-37. |
| [13] | Lokhande, R.S., and Singare, P.U., 2005, Study on kinetics of self diffusion reaction by application of 82 Br as a radioactive tracer isotope., Asian J. Chem., 17(1), 125-128. |
| [14] | Shannon, R.D., 1976, Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides., Acta Crystallographica, A32, 751-767. |
| [15] | Heumann, K.G., and Baier, K., 1982, Chloride distribution coefficient on strongly basic anion- exchange resin: Dependence on co-ion in alkali fluoride solutions., Chromatographia, 15(11), 701-703. |
| [16] | Adachi, S., Mizuno, T., and Matsuno, R., 1995, Concentration dependence of the distribution coefficient of maltooligosaccharides on a cation-exchange resin., J. Chromatography A, 708(2), 177-183. |
| [17] | Shuji, A., Takcshi, M., and Ryuichi, M., 1996, Temperature dependence of the distribution coefficient of maltooligosaccharides on cation-exchange resin in Na+ form., Bioscience, Biotechnology, and Biochemistry, 60(2), 338-340. |
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-Text HTML
Full-Text HTML