-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Journal of Laboratory Chemical Education
2016; 4(1): 9-18
doi:10.5923/j.jlce.20160401.03

Exploring Nutmeg’s Intriguing Place in History Using Narrative and Project-Based Approaches in the Science Laboratory
Todd Pagano1, Mark Goik1, David C. Templeton1, Annemarie D. Ross1, Susan B. Smith2
1Department of Science & Mathematics, Laboratory Science Technology Program, National Technical Institute for the Deaf / Rochester Institute of Technology, Rochester, USA
2Thomas H. Gosnell School of Life Sciences, Rochester Institute of Technology, Rochester, USA
Correspondence to: Todd Pagano, Department of Science & Mathematics, Laboratory Science Technology Program, National Technical Institute for the Deaf / Rochester Institute of Technology, Rochester, USA.
| Email: |  |
Copyright © 2016 Scientific & Academic Publishing. All Rights Reserved.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Developing strategies to engage students in the science classroom and laboratory is a critical task of any post-secondary science instructor/professor. The use of narratives and project-based learning are two methods that can be effective in enhancing the learning process and maximizing student success in coursework. We were inspired by the book, Napoleon’s Buttons: How 17 Molecules Changed History by Couteur & Burreson [1], to develop a series of skill-building activities that could be used as individual or interrelated laboratory experiments or as a capstone project to teach/enforce a variety of chemistry (and some associated biology-related) content in the post-secondary science laboratory. The book discusses the importance of nutmeg and its active component, isoeugenol, within the historical context of potentially protecting individuals against the Black Plague and also nutmeg’s role in the Treaty of Breda [1]. Using this narrative, the essential oil of nutmeg formed the topic for the series of project-based laboratory activities. The essential oil of nutmeg was extracted, isoeugenol was identified and quantified using a variety of analytical methods, and zone of inhibition microbial tests and antioxidant capacity measurements were performed on nutmeg. Analytical instrumentation traditional to the chemistry curriculum; including high performance liquid chromatography (HPLC), gas chromatography (GC), and infrared spectroscopy were employed, with a microplate spectrophotometer used as an extension activity. In addition, general laboratory skills such as sample/standard preparation, sterile technique, calibration curves, and the analytical process were enforced.
Keywords: Nutmeg, Isoeugenol, HPLC, Gas chromatography, Infrared spectroscopy, Microplate spectrophotometer, Antioxidant capacity, Zone of Inhibition, Historical narrative, Project-based learning
Cite this paper: Todd Pagano, Mark Goik, David C. Templeton, Annemarie D. Ross, Susan B. Smith, Exploring Nutmeg’s Intriguing Place in History Using Narrative and Project-Based Approaches in the Science Laboratory, Journal of Laboratory Chemical Education, Vol. 4 No. 1, 2016, pp. 9-18. doi: 10.5923/j.jlce.20160401.03.
Article Outline
1. Introduction
- Instructors are always looking for new and interesting ways to engage students in the science classroom and laboratory. Novel laboratory activities have been developed to capture student interest in the subject matter while teaching the necessary skills of the course curriculum. Such laboratory activities are plentiful and can be used in a variety of different courses and student skill levels. For example, laboratory experiments aimed at an Analytical Chemistry course that focus on instrumental analysis can range from examining art with Raman spectroscopy [2], measuring heavy metals in environmental samples using atomic absorption spectroscopy [3], [4], solving a mock arson trial with GC [5] and analyzing electronic cigarettes with mass spectrometry [6].The use of narratives to support the learning of science and laboratory skill-building can be another effective method for improving student engagement in the laboratory while enforcing important course concepts. In an educational setting, narratives are structured to follow a storyline or common-themed topic to teach desired material while relating back to the context of the narrative. In the science classroom, practitioners have used narratives to facilitate “meaning-making” and to “advance theoretical and analytical points” [7, p. 590]. Narratives in the science classroom can be valuable tools in that they facilitate learning with a constructivist approach [8], whereby the theoretical framework is based on previous understanding/beliefs about the subject content that the students bring to the classroom. The use of narratives has been demonstrated to improve student understanding of certain science concepts [9]. Further, narratives can support student participation while learning the challenging discourse that is required of students in science [10]. The particular narrative used in this study represents a unique way of supporting an interdisciplinary approach to learning by associating historical events to the sciences. When used together, the activities presented in this work can also take advantage of project-based learning (PBL) methods. PBL can motivate student learning by presenting them with a problem to solve using a solution they often originate themselves [11]. The process is student-centered, with the teacher often assuming the role of advisor or consultant, facilitating student development of self-guided activities that may occur over an extended period of time [11]. PBL often encourages student ownership of their scientific knowledge and skills through application to a real-life inquiry activity [12], [13]. PBL activities can sometimes be threaded throughout the entire semester of the course or can be used as a capstone project. Capstone projects can represent effective ways of incorporating curricular components into assignments that allow students to implement what they have learned throughout a program (or culmination of a course); a pedagogical tool sometimes utilized by instructors in the STEM fields [14], [15]. The series of activities presented in this work can be used as stand-alone laboratory activities or can be used as a capstone project that integrates the technical knowledge and critical laboratory skills that students learn throughout a course or through coursework in an academic program. The inspiration for this work came from the book, Napoleon’s Buttons: How 17 Molecules Changed History [1], and the chapter related to nutmeg (Myristica fragrans) introduces the intriguing storyline for the developed activities. The book discusses the possibility that nutmeg was used in Europe to ward off the Black Plague as early as the 14th century. In this historical narrative, people are described as wearing nutmeg necklaces to prevent infliction by the Plague. This doesn’t necessarily indicate that nutmeg (or its active component isoeugenol) functioned as an antibiotic against the bacteria that may have caused the Plague. However, it may have been a natural pesticide against the fleas that carried the disease. In fact, a Raid® product “Earth Options”™ contains the active component, eugenol (a structural isomer of isoeugenol), as an insect control product, and both isoeugenol and eugenol have been shown to have some insect control characteristics [16]. Still, isoeugenol has also been studied for its antimicrobial properties [17].A desire to establish a monopoly on the nutmeg trade may have also been a factor in the second Anglo-Dutch war, where the resolution was the 1667 Treaty of Breda. The Dutch agreed to secede control of the island of Manhattan in the Americas to the English in exchange for the Indonesian island of Run, an exclusive nutmeg growing location [1]. As a result, the English renamed the America’s city of New Amsterdam to New York [1]. Due to this geopolitical swap, one could postulate nutmeg as a hypothetical reason that people from New York are called “New Yorkers” and not “New Amsterdamers” [1]. Significant in spice trade, nutmeg has long been used as a flavorant, but has also been known to have beneficial antioxidant properties [18], possess food preservation qualities [19], and is often used in perfumes [20].The story of nutmeg is used to weave a narrative through a series of project-based laboratory activities in this work. The goal of these activities is to augment student engagement in the learning process while applying important laboratory skills in the post-secondary science laboratory. The series of laboratory activities were developed to systematically investigate the nature of isoeugenol in the oil of nutmeg. Experimental procedures began with the extraction of the essential oil of nutmeg, then samples and standards were prepared, followed by the identification and quantification of isoeugenol. Once the quantity of isoeugenol in the nutmeg oil was determined, testing for bacterial inhibition and antioxidant capacity was conducted. Instrumentation commonly used to analyze real-world samples, such as high performance liquid chromatography (HPLC), gas chromatography (GC), and infrared spectroscopy were utilized along with a microplate spectrophotometer as an extended activity. General laboratory skills were incorporated into the series of activities including solution/standard preparation, sterile technique, calibration curves, and the analytical process. The developed activities can be used as stand-alone laboratory experiments or several (or all) can be used in conjunction as part of an extended project or capstone.
2. Materials and Methods
2.1. Nutmeg Extraction and Sample/Standard Preparation
2.1.1. Essential Oil of Nutmeg Extraction
- A steam distillation method could be used to extract the essential oils from nutmeg (either whole seeds or from commercially purchased nutmeg powder) if an instructor wishes to add distillations as another laboratory technique that could be enforced by this project. A steam distillation/extraction method can be labor and time intensive. Su et al. [21] provide a simpler extraction method that could be used for the various activities in this project. We also completed procedures using a commercially available nutmeg oil. For this working nutmeg oil sample, 30 mg/mL of nutmeg oil (Nature’s Alchemy, 100% essential oil) was brought to volume with methanol (Fisher Scientific, HPLC grade) in a volumetric flask.
2.1.2. Isoeugenol Standards
- A stock solution of 0.8 mg/mL isoeugenol [2-methoxy-4-(prop-1-enyl)-phenol] stock standard was prepared by weighing isoeugenol (Alfa Aesar, 98%) in a 10 mL volumetric flask and bringing it to volume with methanol (Fisher Scientific, HPLC grade). This stock standard was then diluted to 800, 600, 450, 220, 88 and 44 ppm using serial dilutions.
2.2. Instrumental Techniques Applied to the Identification and Quantitation of Isoeugenol in Nutmeg Oil
2.2.1. HPLC
- A Hitachi L-7000 series isocratic HPLC instrument was used with a mobile phase of methanol:acetonitrile:water (25:25:50), a RP-18 column (Phenomenex), and a detection wavelength of 265 nm. The total run time was about 12 minutes when using a flow rate of 1 mL/minute. The 100 µL manual loop injector was sequentially filled with the nutmeg oil working solution and the series of isoeugenol standards that were injected into the instrument. A calibration curve based on the area under the analyte’s peaks was created for quantification purposes. The method could easily be adapted for use with a gradient HPLC system and an autosampler.
2.2.2. Gas Chromatography
- A Perkin Elmer Clarus 500 GC instrument with an autosampler and a mass spectrometer detector was used. The samples and standards were filtered and placed into GC sample vials and capped with a crimper. One µL of each sample and standard were injected into the instrument, which had a total runtime of 34 minutes. The oven was set to an initial temperature of 60°C and held for 1 minute and then slowly ramped at 5°C/minute to 220°C, followed by a 1 minute hold. The GC chromatograms could also be used to quantify isoeugenol in the nutmeg oil via calibration curves. In this work, a spectral library was not used in conjunction with the mass spectrometer detection for spectral identification of the isoeugenol, though with a proper database, this could certainly be done.
2.2.3. Infrared Spectroscopy
- A Perkin Elmer Spectrum BX Fourier Transform Infrared Spectrophotometer (FTIR) with a solid/liquid ZnSe attenuated total reflectance (ATR) sampling accessory was used to analyze 2 to 3 small drops of each nutmeg oil sample and isoeugenol standard for qualitative purposes. All spectra were background subtracted using a methanol blank. A 4000 to 500 cm-1 scan range was used and the sample and standard spectra were collected using the ratio mode at a resolution of 4.0 cm-1 and at a 2.0 cm-1 interval.
2.3. Project Extension: Bacterial Inhibition Testing
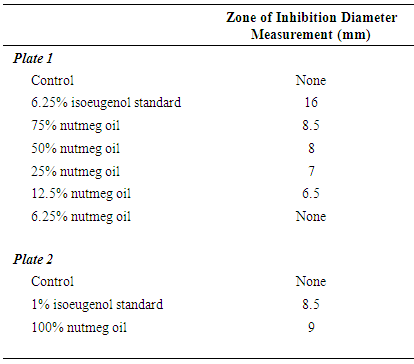
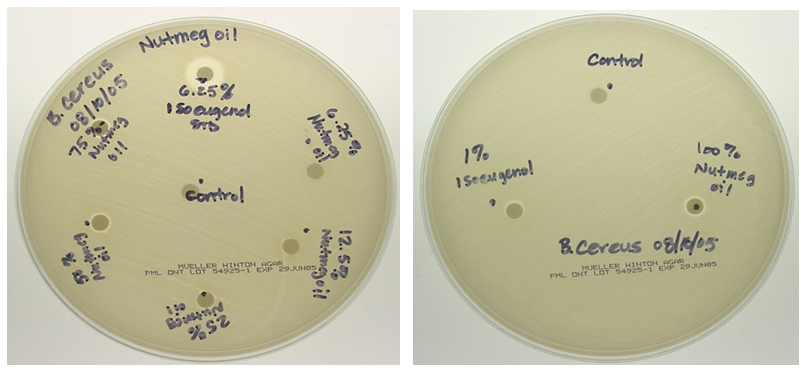
- The isoeugenol standards and nutmeg oil samples were diluted in acetone. Sterile 6 mm paper disks were then soaked in the acetone solutions, and upon removal, the solvent was allowed to evaporate off of the disks for a few minutes. Agar plates (150 mm) were streaked with Bacillus cereus bacteria (Ward’s Science, Rochester NY). Forceps were then used to place each disk onto the agar using sterile technique. The loaded plates were taped, inverted, and left at room temperature for 24 hours. The plates were then examined for the zones of inhibition, the clear halos of bacteria absence surrounding each disk. The zones of inhibition were then measured as the diameter of the clearing halo (which is inclusive of the disk itself) using a metric ruler.
2.4. Project Extension: Antioxidant Capacity of Nutmeg
- The procedures for the antioxidant capacity measurements of nutmeg are based on the procedure developed by Magalhães et al. [22].
2.4.1. ABTS Radical Solution Preparation
- An ABTS radical solution was prepared with 7.0 mM of 2,2’-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS; Sigma, ≥98%) and 2.45 mM of potassium persulfate (Acros Organics, 99+%) in nanopure water. The ABTS and potassium persulfate were allowed to react in solution for 12 to 16 hours in the dark at room temperature to produce the ABTS radical solution. Prior to testing, the ABTS radical solution was diluted 1:9 in nanopure water so that the final dilution had an absorbance of 0.800 ± 0.050 at 734 nm.
2.4.2. Sample Extraction and Preparation for Antioxidant Capacity Measurements
- Nutmeg was extracted according to a modified procedure of Su et al. [21] by adding 2.0 g of commercially available ground nutmeg powder (McCormick) to 20 mL of 80% methanol (Fisher Scientific, HPLC grade) and mixing vigorously on a shaker for 15 hours. The resulting extract was centrifuged for about 15 minutes and then the supernatant was filtered through a 0.45 µm regenerated-cellulose syringe filter (MicroSolv). The filtered extract was then diluted 1:200 in 80% methanol and was also protected from exposure to light prior to testing.
2.4.3. Trolox Standard Preparation
- A 1.0 mM Trolox(6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid) stock solution was prepared by adding 25 mg of Trolox (Acros Organics, 97%) to 100 mL of 50% (v/v) of absolute ethanol (Sigma). From this, five working standards were prepared in 50% ethanol at the following concentrations: 40, 20, 10, 5, and 2.5 µM. These standards were also protected from light by storing in the dark prior to testing.
2.4.4. Antioxidant Capacity Measurements
- The Trolox Equivalent Antioxidant Capacity (TEAC) of the nutmeg extract was measured over the course of a 15-minute reaction in a microplate using a Biotek Eon spectrophotometric microplate reader, whereby the change in absorbance at 734 nm was measured in comparison to a blank. Standards, samples, and nanopure water blanks (150 µL each) were pipetted into the wells of a microplate, and then 150 µL of the ABTS radical solution was added to each standard, sample, and blank to commence the reaction. The plate was immediately inserted into the microplate reader and the absorbance of each well was recorded every minute (with 8 replicate measurements per data point) for the 15 minutes of reaction.
3. Results
- The peak retention times from the HPLC and GC chromatograms for the nutmeg oil and isoeugenol standards can be qualitatively used to verify the presence of isoeugenol in nutmeg. The HPLC chromatograms show matching peaks with the isoeugenol standard for one of the nutmeg oil components at a retention time of approximately 5.5 minutes (Figure 1). Similarly, the GC chromatograms show matching peaks at 1.41 minutes for the nutmeg oil and isoeugenol standard (Figure 2).
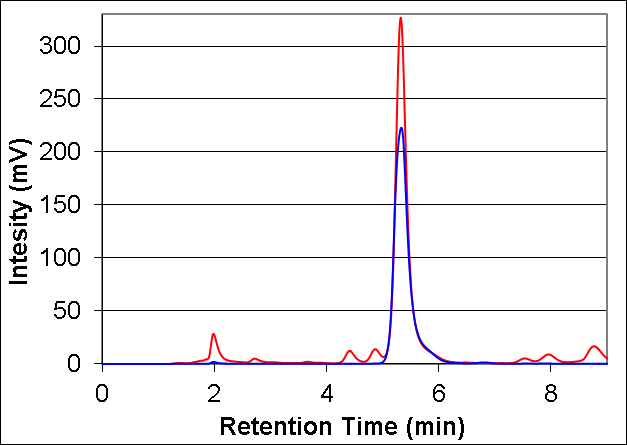 | Figure 1. HPLC chromatogram showing matching peaks for nutmeg oil working sample (red, 30 mg/mL in methanol) and the isoeugenol standard (blue, 800 ppm in methanol) |
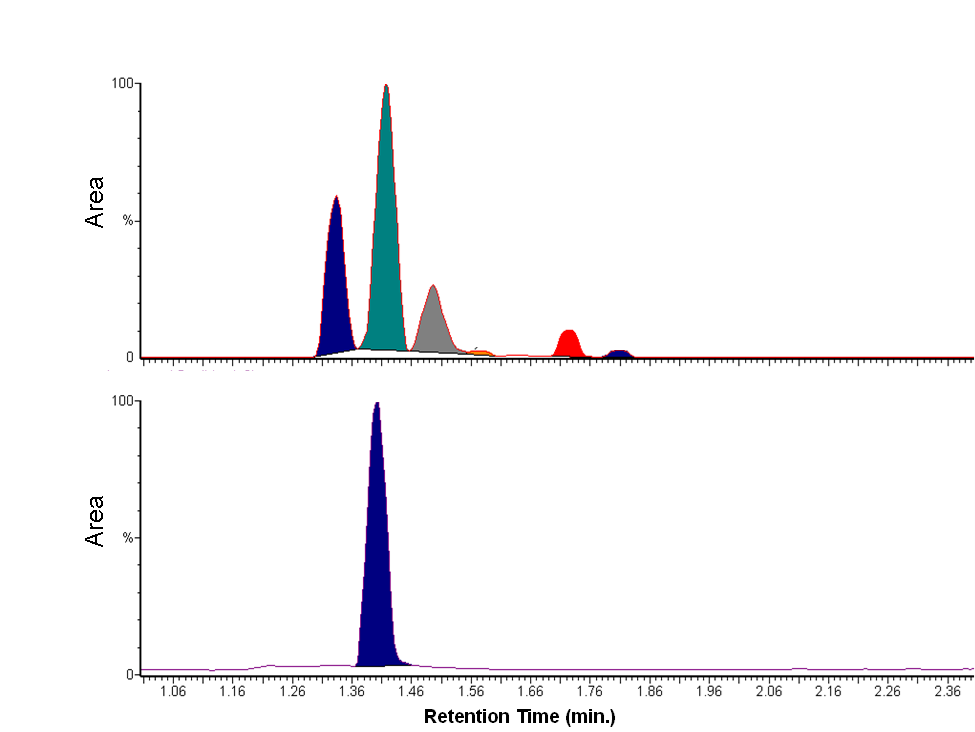 | Figure 2. GC chromatograms showing matching peaks for the nutmeg oil working sample (top, 30 mg/mL in methanol) and isoeugenol standard (bottom, 800 ppm in methanol) |
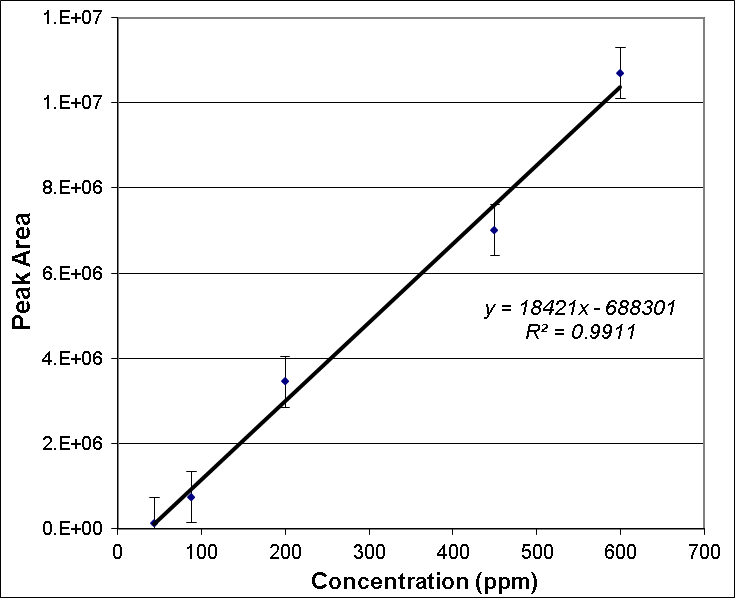 | Figure 3. Calibration curve based on HPLC analysis used to quantify isoeugenol in nutmeg oil. Data points represent average of three replicate injections and error bars are ± one standard deviation |
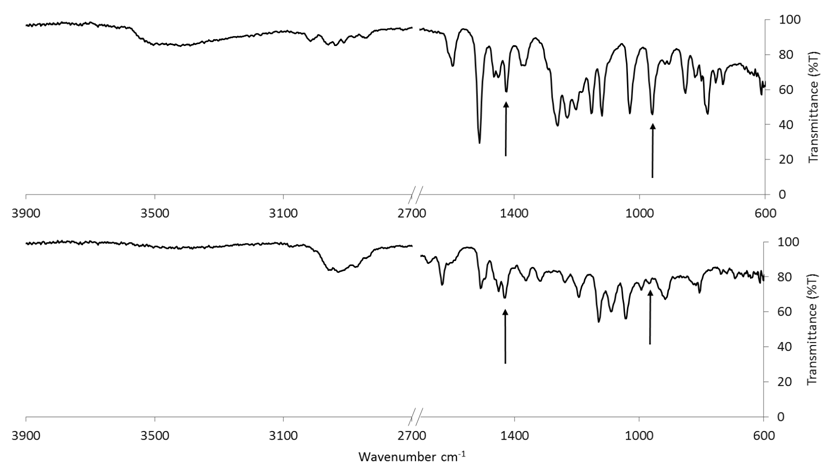 | Figure 4. Infrared spectra of 98% isoeugenol standard (top) and 100% nutmeg oil sample (bottom). Arrows show a couple characteristic frequencies of isoeugenol 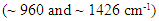 |
|
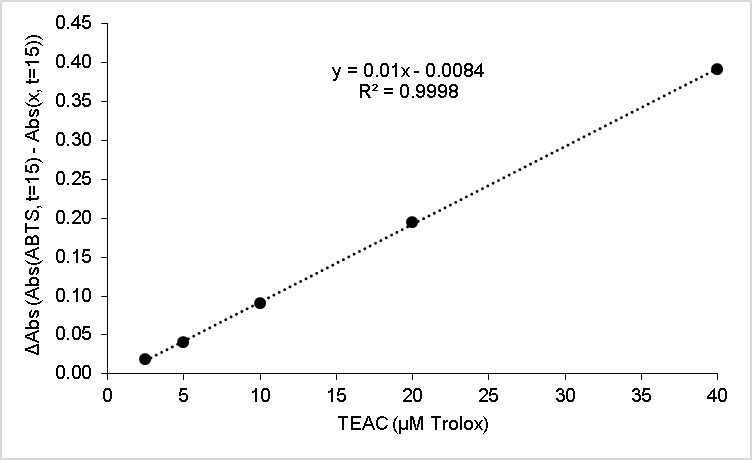 | Figure 6. ABTS Trolox standard curve |
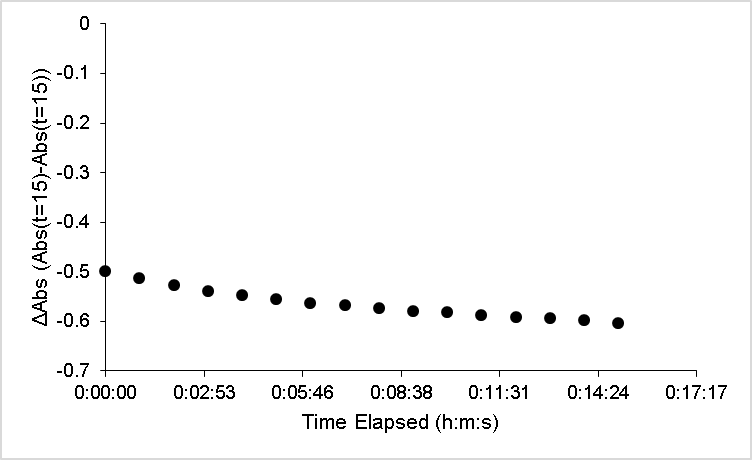 | Figure 7. ABTS kinetic profile for nutmeg over 15 minutes of reaction |
|
4. Discussion
- Calibration curves based on the HPLC analyses of the nutmeg oil determined that isoeugenol composed approximately 1% of the total essential oil profile. Along with the infrared spectroscopy, the chromatography methods (HPLC and GC) also helped to qualitatively identify the isoeugenol in the nutmeg oil. The zone of inhibition extension activity showed that isoeugenol had some antibiotic properties, as its biological activity was assessed in a sensitivity test to the bacteria, Bacillus cereus. Though this specific bacterium was not known to be the cause of the Plague, it can be relevant to show how nutmeg can have antibacterial properties, which might also have given nutmeg historical spice trade value due to food preservation properties. In fact, the seed of nutmeg has been shown to exhibit antibacterial activity against Bacillus cereus [20]. A further extension activity, antioxidant capacity testing, was used to profile the kinetics of the antioxidant properties of nutmeg and determine that it has about 90 µmol Trolox/g of sample using a spectrophotometric microplate reader. This indicates that nutmeg also has nutritional benefits due to its antioxidant capacity, a property that has been ascribed to isoeugenol [18]. Collectively, these results validate the value of nutmeg, and perhaps legitimize some of the historical implications of nutmeg that were introduced in this narrative. Future work into the detailed study of nutmeg could include testing its sensitivity to other bacteria and its applicability as a natural pest control agent. Given the aromatic molecular structure of isoeugenol, fluorescence spectroscopy might also provide another means of spectroscopic analysis, and when in the nutmeg oil form, multidimensional spectra can be coupled with chemometric analyses to characterize the phenolic portion of the essential oil, as has been done on environmental samples by this laboratory [28].Taking a virtual trip back in time, hundreds of years ago, to investigate whether or not the notion that nutmeg had the potential to protect people from the Black Death was based on scientific fact or myth can provide a narrative of interest to students. Although this project has roots in historical events, its educational value far exceeds an examination into manipulated treaties from the 17th century. Students can benefit from completing skill-building activities, working within the context of interdisciplinary fields, using advanced problem solving, and (if desired by the instructor) engaging in teamwork. A project such as this can entice students with backgrounds or interests in history, sociology, or the natural and physical sciences.An interdisciplinary, project-based learning experience allows both faculty and students to engage in more realistic applications of skills and knowledge taught or acquired in the classroom. Faculty from multiple disciplines should be encouraged to participate in various aspects of this project, not only as an opportunity to redirect learning goals from a course curriculum but to work directly with students engaged in inquiry. The developed laboratory activities could be introduced to students enrolled in courses such as Analytical Chemistry, Instrumental Analysis, and Organic Chemistry. The extension activities could be completed in General Biology, Microbiology, or Nutrition courses. As stated, another approach would be to group the activities into a capstone course or project. This particular series of activities places heavy emphasis on developing analytical instrumentation skills such as HPLC, GC, and infrared spectroscopy. Throughout the instrumental analyses students engage in the especially important skills of creating calibration curves, preparing samples and standards, and interpreting data.It should be noted that activities such as these present significant opportunities for students to develop writing skills. Scientific writing (including the writing of formal laboratory reports) provides a structured format that includes writing for detail and accuracy, analysis of data, evaluation of results, and knowledge transfer. All of the Bloom’s Taxonomy [29] levels can often be incorporated into one writing activity in the sciences. Scientific writing provides rich experiences to enhance writing skills at various cognitive levels including descriptive, analytical, evaluative, and transfer of knowledge. The fact that students have actually experienced the planning, preparation, process and data collection involved with a project such as this provides a meta-cognitive enhancement to the writing experience. The student writing components of these individual activities, or project when used together, can take the form of traditional laboratory reports, organized laboratory notebooks, memo writing, or wiki creation– to name a few types of writing assignments. These activities also lend themselves well to formal presentations by students or groups of students.Prior to being used in the classroom, this project began as an undergraduate research project conducted by students in their first two years of college study. The techniques used formed an appropriate level for the students and the context of the narrative provided a topic that they found interesting– two criteria for the successful integration of undergraduate research at this level [30], [31]. Whether the focus of undergraduate research projects or embedded in course curricula, these projects effectively engage students in laboratory learning through the use of real-world examples and topics that may pique the interest of post-secondary students.
5. Conclusions
- The book, Napoleon’s Buttons: How 17 Molecules Changed History [1], stimulated the use of two pedagogical strategies designed to engage and enhance student learning, motivation, and success in the science laboratory. A combination of a historical narrative focused on nutmeg’s role in history and project-based investigations into the chemical, biological, and nutritional properties of isoeugenol, an active component in the essential oil of nutmeg, is presented for use in a variety of post-secondary laboratory courses. Although the activities and demonstrated results were not intended to demonstrate that isoeugenol was effective in preventing infection from microorganisms that caused the Black Plague, they highlight its potential value for its antibacterial and antioxidant characteristics that might have provided beneficial health and nutriment properties during the spice trade. Future studies could investigate whether the chemicals in the profile of nutmeg have pest control properties that might have historically kept Plague-carrying fleas away from people. Serving as major contributions to the learning process were the development of a series of progressive, interrelated, skill-building activities for use in laboratory science curricula. Enforced skills include sample/standard preparation, instrumental analyses, and sample identification and quantification. These activities implemented the standard array of analytical instrumentation; HPLC, GC and infrared spectroscopy, while other extension activities (zone of inhibition and antioxidant capacity testing) were conducted to enhance the investigation. It is important to clarify that a series of interrelated activities such as these may be included in a capstone-like course or used in individual courses within a program of study (like General Chemistry, Analytical Chemistry, Organic Chemistry, and Instrumental Analysis) as determined by faculty.
ACKNOWLEDGEMENTS
- Portions of this work were developed in conjunction with some of the outstanding students in the Laboratory Science Technology program at Rochester Institute of Technology’s National Technical Institute for the Deaf (Rochester, NY USA). These students include Travis Dickson, Troy Hohlweck, Ahmed Ibrahim, Abiodun Odunlami, Shabnam Razmpour, Julia Renda, and Jacquelyn Wilson. The authors would like to thank Morgan Bida, Lori Poole, Charmaine Merchant, and Amie Sankoh for their assistance with some technical laboratory aspects of this project and Dr. Carmen Pagano for early discussions about the historical narrative portion of this project.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML