-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Journal of Laboratory Chemical Education
2015; 3(4): 59-66
doi:10.5923/j.jlce.20150304.01

An Active Learning-Based Integrated Project between High Schools and the University: Analysis of Bioethanol Fuel and Its Blends with Synthetic Gasoline
Luzia P. Novaki, Ana M. Chinelatto, Omar A. El Seoud
Institute of Chemistry, the University of São Paulo, São Paulo, Brazil
Correspondence to: Omar A. El Seoud, Institute of Chemistry, the University of São Paulo, São Paulo, Brazil.
| Email: |  |
Copyright © 2015 Scientific & Academic Publishing. All Rights Reserved.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In order to increase the interest of high-school students in chemistry, and initiate their contact with the Institute of Chemistry of the University of São Paulo (Chem-USP), we introduced a project of socio-economic relevance, analysis of bioethanol fuel (hereafter called “ethanol”) and its blends with synthetic gasoline. We used concepts that these students are familiar with, and techniques that can be employed in high schools. Due to historical reasons, Brazilian students are familiar with the use of ethanol as fuel, either hydrous, or as a blend with gasoline in “flex-fuel” cars and motorcycles. They may lack, however, knowledge about industrial production of ethanol and possible adulteration of fuels. We carried out this project in three stages; I and III at the schools and II at Chem-USP. In stage I, the students conducted a literature survey, answered a quiz that their teachers corrected and discussed in class, and then prepared ethanol-water mixtures. In stage II, they came to Chem-USP to determine the densities and refractive indices of their aqueous ethanol mixtures, and the refractive indices of ethanol-synthetic gasoline mixtures using digital density meter and refractometer. They recorded the Uv-Vis spectra of the natural dye β-carotene dissolved in ethanol-water mixtures. All students constructed calibration curves (physical property versus mixture composition) and determined the composition of commercial unknowns (household cleaning ethanol and hydrous ethanol fuel). In stage III, the students discussed with their teachers the results of their experiments, and the concepts involved (e.g., hydrogen-bonding) and then answered an evaluation sheet of the project. They liked the active learning approach; their working in groups with modern equipment. The experimental part of this project can be carried out using less sophisticated infrastructure, namely, volumetric glassware, analytical balance, Abbe refractometer, and a low-cost spectrophotometer.
Keywords: Active learning, Constructivist teaching, Bioethanol analysis, Gasoline, Density, Refractive index, Uv-Vis spectroscopy, β-carotene
Cite this paper: Luzia P. Novaki, Ana M. Chinelatto, Omar A. El Seoud, An Active Learning-Based Integrated Project between High Schools and the University: Analysis of Bioethanol Fuel and Its Blends with Synthetic Gasoline, Journal of Laboratory Chemical Education, Vol. 3 No. 4, 2015, pp. 59-66. doi: 10.5923/j.jlce.20150304.01.
Article Outline
1. Introduction
- The use of “gasohol” as fuel for cars is probably the best-known example of the large-scale use of renewable fuels. This product is sold in sixteen countries and ten states in the USA [1]. It is a blend of gasoline and bioethanol (hereafter designated as “ethanol”); the latter component is obtained by fermentation, e.g., of sugar-cane juice [2]. In addition to being from renewable sources, ethanol is an oxygenated fuel and has a higher octane number than those of hydrocarbons. For example, the RONs (Research Octane Numbers) are 108.6 and 88 to 100, for ethanol and commercial grades of gasoline, respectively [3].Brazil has a long history of using ethanol as fuel for internal combustion engines. Since 1931 the gasohol sold locally contains 25±1 v% of anhydrous ethanol. Mass-produced passenger cars that run on hydrous ethanol (92.5 to 93.8 wt%, or 95.1 to 96.0 v% ethanol) have been on the market for the last 36 years [4-6]. Since 2003, the cars sold in Brazil run on “flex-fuel”, i.e., gasohol, hydrous ethanol, or any mixture of both fuels. Motorcycles running on the same flex-fuel were introduced commercially in 2009 [7].Fuel analysis is important for economic reasons because the price/liter of hydrous ethanol (charged in Brazil) is maintained at ca. 70% of the price of gasohol, due to the lower calorific value of the former fuel [6, 8]. Due to the miscibility of gasoline and hydrous ethanol, one principle gasohol adulteration practice is to add more ethanol (beyond the 25 v% legal limit). Additionally, hydrous ethanol can be easily falsified by adding water (see, however, Figure 2 and the corresponding discussion).Because of this relevance, we introduced several schemes for fuel analysis in the undergraduate laboratory of the Institute of Chemistry of the University of São Paulo (Chem-USP), all of them based on absorption of synthetic [9] or natural dyes, namely β-carotene or chlorophyll [10]. We present here a project for analysis of ethanol-water mixtures (hereafter called “E-W”), and ethanol-synthetic gasoline mixtures (hereafter called “E-G”). Our aim is to increase the interest of high school students in chemistry and initiate their contact with Chem-USP. Using the active-learning approach, we carried out this project in collaboration with high school teachers in three stages. First, the teachers asked the students to do literature survey on the subject; they answered a quiz, and then they prepared E-W. In the second stage, they visited our laboratory at Chem-USP, where they determined the densities and refractive indices of E-W, the refractive indices of E-G (that we prepared); and recorded the absorbance of β-carotene solubilized in E-W. They constructed the appropriate calibration curves from which they determined the compositions of commercial household cleaning ethanol; hydrous ethanol fuel samples, and E-G unknowns that we provided. In the third stage, they discussed their results in class, and evaluated the project. Their positive answers indicated that they liked the active-learning approach; working in groups, and appreciated learning new techniques and concepts. The experiment is safe, generates little, easily disposable residue, and can be done in a typical high-school laboratory.
2. Experimental Part
2.1. Solvents, Reagents, and Commercial Ethanol Samples
- We purchased the solvents and reagents from Fluka and Synth (São Paulo). We treated commercial anhydrous ethanol (ca. 99.6%) with sodium (6 g/L ethanol), distilled and then stored the alcohol over activated 3Å molecular sieves. Xylenes (isomer mixture, 98.5%), 1-heptane (99%), decalines (cis and trans isomers, 99%), and β-carotene were used as received. Figure 1 shows the molecular structure of β-carotene.
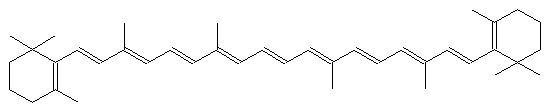 | Figure 1. The molecular structure of the green dye β-carotene |
2.2. Preparation of Synthetic Gasoline; Ethanol-Gasoline, and Ethanol-Water Mixtures
- Precision burettes with PTFE stopcocks, and 25 mL volumetric flasks with polypropylene stoppers (e.g., Schott Duran GLS114.202.05A, and GLS130.202.03, respectively) were used throughout. We prepared synthetic gasoline by volume, by mixing the following hydrocarbons: xylenes (50 v%), n-heptane (45 v%) and decalines (5 v%). In principle, the ethanol-containing mixtures employed in the present project are not ideal, vide infra; mixing of components is not thermo-neutral [11]. Therefore, we (at Chem-USP) and the students in their schools proceeded as follows: (i) E-G: We carried out this preparation by pipetting the required volumes of anhydrous ethanol in 25 mL volumetric flasks. Next, we added enough gasoline so that the total volume is ca. 0.5 mL below the mark of the flask. The latter was thermostated in a water-bath at 25°C for 10 min before completing the volume with gasoline. The mixtures employed for construction of the calibration curves contained 10 to 90 v% ethanol.(ii) E-W: Under supervision in their respective schools, the students prepared ethanol-water mixtures by using the anhydrous ethanol that we supplied. The procedure is similar to that employed in (i), except that they first pipetted ethanol, and then diluted with water. The mixtures employed for construction of the calibration curves contained 10 to 98 v% ethanol.
2.3. Equipment
- The students used the following equipment at Chem-USP: DMA 4500M digital, resonating density meter (Anton-Paar; d ± 0.00001 g. cm-3), equipped with a closed reservoir for collecting the spent sample after measurement, and a diaphragm pump for drying the measuring U-shaped quartz tube. The students determined the refractive index (n) with J357 digital refractometer, operating at 488 nm (Rudolph Research; n ± 0.00001). Both equipment are provided with Peltier thermoelectric heat pumps for temperature control, within 0.01°C. The students recorded the Uv-Vis spectra with UV-2500 double-beam spectrophotometer (Shimadzu) equipped with a cuvette holder whose temperature is controlled within 0.05°C by water circulation.Additionally, we used the following equipment: manual, precision Abbe 60 refractometer (Bellingham & Stanley; λ = 589nm; n ± 0.0005) whose temperature is maintained with water circulation; Laborette 17 ultrasonic bath (Fritsch; 140 W, 35 KHz), and MicroMax MP4R centrifuge (IEC).
2.4. Methods
- Note: We explained the equipment and carried out demonstration measurements before allowing the students to measure their samples, under our supervision.
2.4.1. Density
- The sample measuring quartz tube was cleaned with acetone and dried by air suction. The liquid (E-W) was injected into the inlet with 2 mL polypropylene syringe, e.g., CG-3080-02 (Chemglass). The students recorded the digital display (density) when the reading stayed constant (± 0.00001 g. cm-3).
2.4.2. Refractive Index
- The students cleaned the small, circular measuring surface (1 cm diameter) with a soft tissue paper. They added two drops of the solution (E-W or E-G), closed the measuring cell cover, and then recorded the digital display (refractive index) when the average of three consecutive readings was within ± 0.00001 unit.
2.4.3. Uv-Vis Spectra of β-Carotene Solubilized in Ethanol-Water Mixtures
- This experiment is time consuming because it involves weighing of β-carotene; its dissolution (by sonication) in the different aqueous ethanol mixtures, and separation of the undissolved natural dye (by centrifugation). Therefore, we used the E-W mixtures prepared by the students (and sent to us in advance) to prepare the β-carotene solutions, whose spectra were recorded in the Uv-Vis region. The procedure was as follows: In 2 mL volumetric tubes with polypropylene stoppers (e.g., Pyrex 5642) we weighted ca. 3 mg β-carotene/tube, and then added 2 mL of the E-W (40 to 98 v% ethanol). We sonicated the dye suspension for 30 minutes, and then centrifuged the tubes at 400 G for 30 minutes. The students received the tubes after centrifugation and then carefully transferred the supernatants into 0.2 cm path-length glass cuvettes with PTFE covers (e.g., G102; Azzota), placed the cuvettes into the thermostated cell compartment of the spectrophotometer, and then recorded the spectra from 350 to 550 nm. The students used the dye absorbance at 450 nm for constructing the calibration curve, vide infra.
3. Results and Discussion
3.1. Scheme of Activities
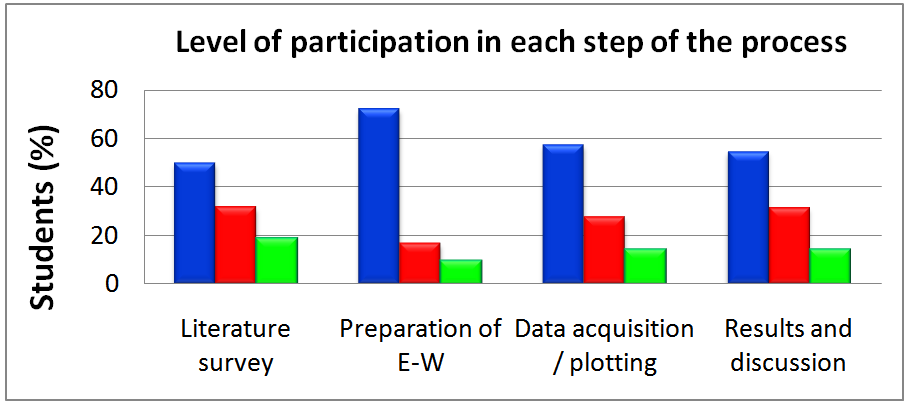
- Seventy-five students in their last year of high school, from three schools, participated in this project; its stages included: (a) Literature survey about the production and commercial uses of ethanol, and the physical properties of E-W; (b) Preparation (in high schools) of E-W with 10 to 98 v% ethanol; (c) Measuring (at Chem-USP) of the following properties as a function of mixture composition: densities and refractive indices of E-W; refractive indices of E-G; absorbance of β-carotene solubilized in E-W. The students worked in groups of eight or nine each; there were three groups from each high school.
3.1.1. Literature Survey and Answering the Quiz about Ethanol
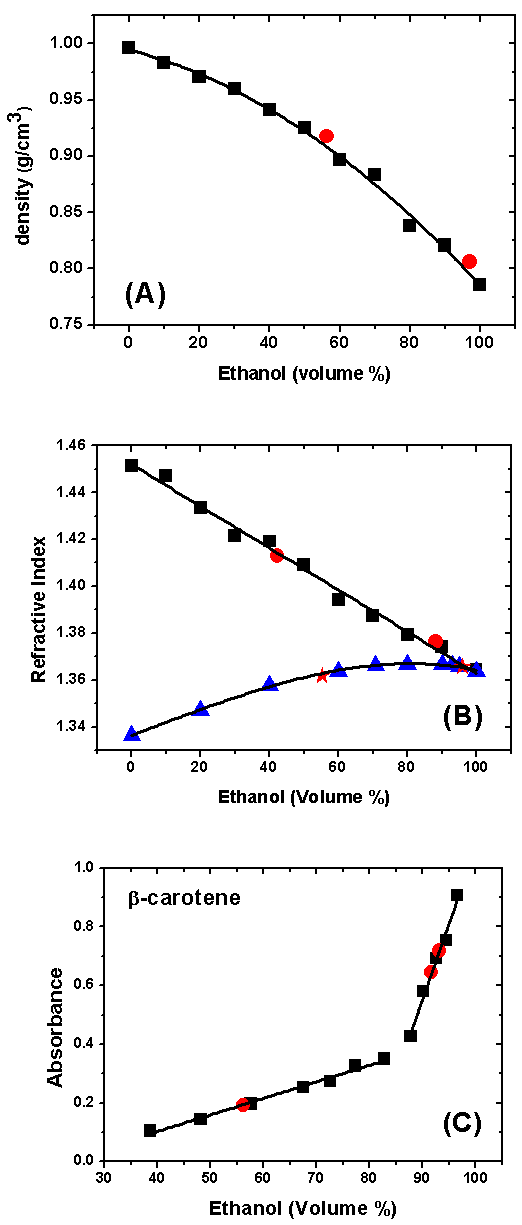
- Before visiting Chem-USP, the participating students searched the internet for the industrial production of ethanol; formation of ethanol-water azeotrope; commercial methods to remove water from hydrous ethanol, and methods to determine the water content in hydrous ethanol. The teachers assessed the results of their search by distributing a quiz that we supplied. The students answered the latter in class; later their teachers commented on their answers. We asked them about the advantages, if any, of using gasohol as fuel instead of gasoline. Most students (68%) cited the concomitant increases of the mixture octane number; 31% and 33% cited reduction of air pollution, and the fact that ethanol is a renewable fuel, respectively. Their answers point into the right direction as discussed in Introduction about the octane number. Additionally, sugar cane is a fast-growing crop; it is harvested every 12 or 18 months [12].With regard to commercial production of anhydrous ethanol (ca. 99.6 v % pure) from the E-W azeotrope (composition 95.5% ethanol by mass), the students gave the following answers: dehydrate the alcohol by using CaO (47%), or activated molecular sieves (37%), or add a third component and remove the water present as a 3-component azeotrope (16%). Although the use of CaO seems advantageous because of its low cost (ca. 4 cents to dehydrate one liter of ethanol), one should consider the additional energy cost of regenerating CaO from the corresponding Ca(OH)2 by calcination, and the safety aspect of heating large amounts of an ethanol-containing Ca(OH)2 “cake”. Hydrous ethanol forms ternary isotropic mixtures with relatively benign hydrocarbons, e.g., n-hexane and n-heptane [13], this is the basis of the so called “extractive distillation” method [14]. Therefore, the latter, along with treatment with activated molecular sieves, e.g., 3Å, are appropriate for industrial removal of water from hydrous ethanol. Finally, density was the method of choice for the determination of the water content of W-E (53%). The recognition of density as an appropriate property is certainly linked to the fact that (in Brazil) each pump that sells hydrous alcohol is provided with an alcoholmeter and a visible sticker, as shown in Figure 2. Therefore, the consumer is assured of the quality while the fuel flows into the car/motorcycle tank. The Gay-Lussac alcoholmeter is provided with an inner tube containing a bright-red liquid. The sticker fixed on its side shows three possibilities, based on ethanol content, i.e., on E-W density. The ethanol fuel is appropriate if the level of the red liquid is below, or at the surface of flow of hydrous ethanol, but not above it. The reason is that ethanol adulteration by adding water increases the density of the E-W, pushing the alcoholmeter “up”. The activities of this stage, namely, answering the quiz and the teacher’s comments (after correcting the answers) took one hour.
3.1.2. Preparation of Ethanol-Water Mixtures
- The students went to the school´s laboratory to prepare their E-W for analysis by the different experimental techniques. Under supervision, they prepared in one hour nine E-W, 25 mL each.
3.1.3. Activities in the Laboratory of Chem-USP
- The objective of the visit to Chem-USP was to determine the v% of ethanol in: (i) commercial household cleaning aqueous ethanol, (ii) commercial samples of hydrous ethanol fuel, (iii) samples of E-G. They determined the compositions of these samples by measuring their densities and refractive indices, as well as the Uv-Vis absorption of β-carotene solubilized by E-W. We chose these techniques because they are simple, and connected with concepts that the students at this level are familiar with, namely, solutions, mixtures, density, and refractive index. A comment on the safety aspect of the experiment is in order. Ethanol is commonly employed as solvent in any laboratory [15]. Rather than working with commercial local gasoline whose b.p. range is >35 to 220°C [16], we employed a synthetic mixture whose lowest b.p. component is 98.4°C (n-heptane). The composition of the synthetic gasoline that we employed (v%) is in the range of typical commercial gasolines, namely: 4-8% alkanes; 2-5%, alkenes; 25-40% isoalkanes; 3-7% cycloalkanes; 1-4% cycloalkenes; and 20-50% aromatics [17, 18]. The student groups rotated between the following three experiments: Determination of the densities of E-W; determination of the refractive indices either of E-W or E-G; determination of the absorbance (A) of β-carotene dissolved in E-W. Because determination of the composition of unknown samples is based on calibration curves, e.g., between (n) and v% of ethanol, each group measured the properties of three reference solutions, and two unknowns (furnished by us), see Figure 3. While doing the experiment, each group registered their experimental data in a table, using the Excel software. This table was completed when the last group finished its measurement at the equipment (d, n, or A).
 | Figure 3. The students working at the laboratory of Chem-USP. The photos (from left to right) show them using the density meter, the refractive index meter, and the Uv-Vis spectrophotometer |
3.1.4. Discussion of the Results and Students´ Evaluation of the Project
- The last stage of the project included discussion of the results in class, followed by evaluation of the project. Based on their previous literature survey, the students attributed the non-linear dependence of the density and refractive index of E-W (parts A and B of Figure 4) to the formation of hydrogen bonds between both components of the mixture, as discussed elsewhere [19]. In order to stress this non-ideality and to show that it is a general phenomenon when the components of a mixture interact strongly, their teachers showed them Figure 5, for 50 v% mixtures of organic solvents (acetone and ethanol) with water. We prepared these mixtures at 25°C and added a few mg of methyl orange for a better visualization of the (transparent) solvent level. As shown in Figure 5, the final volumes are less than the sum of the volumes of (water + organic solvent; 5 mL each) that we added to each flask.
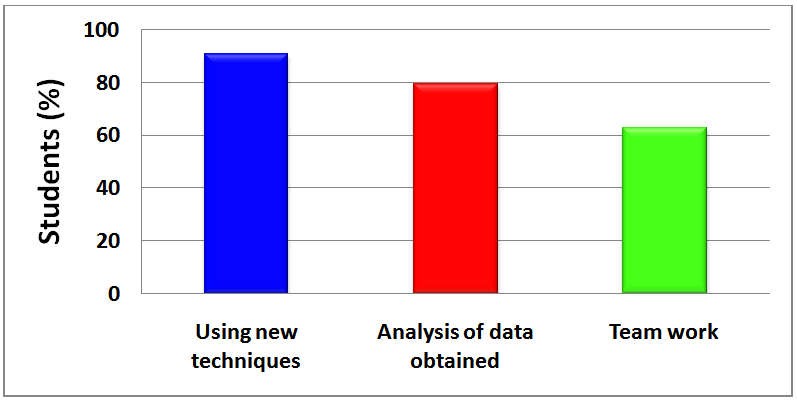 | Figure 7. Summary of the students´ answers to a question about the positive aspects of the project |
4. Conclusions
- We employed the active learning approach to introduce a project of socio-economic importance to high school students and promote their contact with Chem-USP. The students analyzed aqueous ethanol employed for household cleaning; hydrous alcohol fuel; E-W that they prepared in their schools, and E-G that we provided. The experiment is safe, can be done with simple equipment, generates little residue that is easy to dispose (e.g., by incineration), and serves to highlight important concepts (density, refractive index, light absorption) and link them to an everyday situation, buying a fuel at the gas station.
ACKNOWLEDGEMENTS
- We are grateful to the students of the following high schools for their participation in this project and their enthusiasm: Colégio Waldorf-Micael de São Paulo; Escola de Química., and Colégio Bandeirantes. We are indebted to their teachers that conducted the parts of the project in the schools: Paula Brotero (Colégio Waldorf-Micael), Gabriel B. Polho and André Franco (Escola de Química and School of Medicine-USP), Lilian Siqueira and Fábio Siqueira (Colégio Bandeirantes). O. A. El Seoud thanks FAPESP and CNPq research foundations for financial support and for research productivity fellowship, respectively.
References
| [1] | http://www.carsdirect.com/green-cars/gasohol-facts-figures-and-common-blends; accessed October 28th, 2015. |
| [2] | Basso, L.C.; Basso, T.O.; Rocha, N.S., “Ethanol production in Brazil: The industrial process and its impact on yeast fermentation”. www.intechopen.com/download/pdf/20058; accessed October 28th, 2015. |
| [3] | http://www.eesi.org/files/Robert_McCormick_061215.pdf, accessed October 21st, 2015; Celik, M. B.; “Experimental determination of suitable ethanol-gasoline blend rate at high compression ratio for gasoline engine”; Applied Thermal Engineering, 2008, 28 396 – 404. |
| [4] | http://nxt.anp.gov.br/nxt/gateway.dll/leg/resolucoes_anp/2013/fevereiro/ranp%207%20-%202013.xml; accessed October 28th, 2015. |
| [5] | http://www.mundoeducacao.com/geografia/alcool-combustivel.htm; accessed October 28th, 2015. |
| [6] | http://en.wikipedia.org/wiki/Ethanol_fuel_in_Brazil; accessed October 26th, 2015. |
| [7] | http://www.unica.com.br/download.php?idSecao=17&id=17629673; accessed October 28th, 2015. |
| [8] | http://www.afdc.energy.gov/fuels/fuel_comparison_chart.pdf, accessed October 26th, 2015; http://prh.mecanica.ufrj.br/index_arquivos/palestras/20121207-1.pdf, accessed October 28th, 2015. |
| [9] | Galgano, P. D.; Loffredo, C.; Sato, B. M.; Reichardt, C.; El Seoud, O. A.; “Introducing education for sustainable development in the undergraduate laboratory: quantitative analysis of bioethanol fuel and its blends with gasoline by using solvatochromic dyes”; Chem. Educ. Res. Pract., 2012, 13, 147. |
| [10] | Novaki, L. P.; Costa, R. T.; El Seoud, O. A., “Green chemistry in action: An undergraduate experimental project on the quantitative analysis of bioethanol blends with synthetic gasoline using natural dyes”, J. Lab. Chem. Educ., 2015, 3, 22-28. |
| [11] | Hsu, K.-Y.; Clever, H. L., “Enthalpy of mixing of some hydrocarbon-alcohol and hydrocarbon-nitroalkane mixtures at 25deg”, J. Chem. Eng. Data, 1975, 20, 268-271; Boyne, J. A.; Williamson, A.G., “Enthalpies of mixture of ethanol and water at 25 degree C”, J. Chem. Eng. Data, 1967, 12, 318–318. |
| [12] | http://www.agencia.cnptia.embrapa.br/gestor/cana-de-acucar/arvore/CONTAG01_33_711200516717.html; accessed October 28th, 2015. |
| [13] | Skrzecza, A.; Shaw, D.; Maczynski, A., “IUPAC-NIST Solubility Data Series 69. Ternary Alcohol– Hydrocarbon–Water Systems”, J. Phys. Chem. Ref. Data, 1999, 28(4), 983. |
| [14] | Lei, Z.; Li, C.; Chen, B., “Extractive Destillation: A Review”, Separation & Purification Reviews, 2003, 32(2), 121-213. |
| [15] | Vogel, A.I.; Tatchell, A. R.; Furnis, B. S.; Hannaford, A. J.; Smith, P. W. G.; in Vogel’s Textbook of Practical Organic Chemistry, 5th Edition, 1996. |
| [16] | http://www.br.com.br/wps/wcm/connect/52cb170043a79d87baa2bfecc2d0136c/fispq-auto-gasolina-comum.pdf?MOD=AJPERES; accessed October 28th, 2015. |
| [17] | International agency for research on cancer- summaries & evaluations. Gasoline, 45, 1989, 159. |
| [18] | http://www.atsdr.cdc.gov/toxprofiles/tp72-c3.pdf; accessed October 28th, 2015. |
| [19] | Kuchuk, V.I.; Shirokova, I. Y.; Golikova, E.V.; “Physicochemical properties of water-alcohol mixture of a homological series of lower aliphatic alcohols”; Glass Physics and Chemistry, 2012, 38 (5), 460-465.; Abdulagatov, I.M.; Akmedova-Azizova, L. A.; Azizov, N.D.; “Experimental study of the density and derived (excess, apparent, and partial molar volumes) properties of binary water + ethanol and ternary water + ethanol + lithium nitrate mixtures at temperatures from 298 K to 448 K up to 40 MPa”; Fluid Phase Equilibria, 2014, 376, 1-21. |
| [20] | Craft, N. E., Soares Jr, J.H., “Relative Solubility, Stability, and Absorptivity of Lutein and β-Carotene in Organic Solvents”, J. Agric. Food Chem., 1992, 40, 431-434. |
| [21] | Sujak, A.; Okulski, W.; Gruszecki, W.I.; “Organisation of xanthophyll pigments lutein and zeaxanthin in lipid membranes formed with dipalmitoylphosphatidylcholine”, Biochimica et Biophysica Acta, 2000, 1509, 255-263; Ruban, A.V.; Horton, P.; Young, A.J.; “Aggregation of higher plant xanthophylls: differences in absorption spectra and in the dependency on solvent polarity”, J. Photochem. Photobiol. B: Biol., 1993, 21, 229-234. |
| [22] | http://www.eventoscientificos.com/imoc2009/papers/pdf/59090.pdf; accessed October 28th, 2015; Possetti, G.R.C.; Muller, M.; Fabris, J.L.; “Refractometric optical fiber sensor for measurement of ethanol concentration in ethanol-gasoline blend”, 2009 SBMO/IEEE MTT-S International Microwave & Optoelectronics Conference, IMOC 2009, 616-620. |
| [23] | Kwak, H.-Y.; Oh, J.-H.; Park, S.-J.; Paek, K.-Y.; “Isothermal vapor–liquid equilibrium at 333.15K and excess volumes and molar refractivity deviation at 298.15K for the ternary system di-butylether (1) + ethanol (2) + toluene (3) and its binary subsystems”, Fluid Phase Equilibria, 2007, 262, 161. |
| [24] | alcool_etilico_hidratado_46_inpm_54gl_3jul15_1435929002.pdf; accessed October 28th, 2015. |
| [25] | http://nxt.anp.gov.br/nxt/gateway.dll/leg/resolucoes_anp/2013/fevereiro/ranp%207%20-%202013.xml; accessed October 28th, 2015. |
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML