R. Z. Saydaliyeva1, A. E. Zaynabiddinov2
1Assistant, Department of Medical Chemistry and Biological Sciences, Central Asian Medical University
2Professor, Doctor of Biological Sciences Andijan State University, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
This study investigates the antihypertensive potential of specific polyphenol compounds A51, A52, and A54 through in vivo experimental models. Hypertension remains a critical risk factor for cardiovascular diseases, necessitating the search for effective natural compounds with minimal side effects. The research focuses on the comparative analysis of these three polyphenols in regulating blood pressure. Our findings indicate varying degrees of efficacy, with A54 showing a significantly more potent hypotensive effect compared to A51 and A52. The results suggest that these polyphenols could serve as promising candidates for the development of new-generation antihypertensive phytopreparations.
Keywords:
Polyphenols, A51, A52, A54, Antihypertensive activity, In vivo analysis, Comparative study, Cardiovascular health
Cite this paper: R. Z. Saydaliyeva, A. E. Zaynabiddinov, In Vivo Analysis and Comparative Study of the Antihypertensive Activity of A51, A52, and A54 Polyphenols, International Journal of Virology and Molecular Biology, Vol. 15 No. 2, 2026, pp. 47-51. doi: 10.5923/j.ijvmb.20261502.05.
1. Introduction
Polyphenols are secondary metabolites found in plants, known for their antioxidant and vasoprotective properties. Recent pharmacological studies have highlighted their role in modulating nitric oxide (NO) bioavailability and inhibiting angiotensin-converting enzyme (ACE). This article presents a comparative evaluation of three newly isolated or synthesized polyphenol derivatives (A51, A52, A54) to determine their therapeutic index in managing high blood pressure.
2. Materials and Methods
Test Compounds: Polyphenol fractions A51, A52, and A54.Experimental Models: The study was conducted on white laboratory rats with induced arterial hypertension.Ethical Approval: All experimental protocols were reviewed and approved by the Institutional Animal Care and Use Committee (IACUC) of Andijan State University (Approval No: ASU-2024/05-BP). The study was conducted in strict accordance with the NIH Guide for the Care and Use of Laboratory Animals and the European Convention for the Protection of Vertebrate Animals used for Experimental and other Scientific Purposes. Hypertension Induction Method: Acute hypertension was induced using the adrenaline-induced model. To stimulate a rapid hypertensive response, epinephrine hydrochloride (0.1 mg/kg) was administered via the tail vein. Baseline blood pressure (BP) was measured prior to induction, and only rats exhibiting a stable systolic blood pressure (SBP) increase of at least 25–30% were included in the treatment groups.Randomization and Grouping: A total of 16–18 healthy male Wistar rats were used. To minimize selection bias, animals were assigned to four experimental groups (n=4 per group) using a simple randomization technique (computer-generated random numbers). The groups were defined as follows:• Group 1 (Control): Administered intravenous adrenaline (0.1 mg/kg) followed by saline (0.9% NaCl).• Group 2 (A51): Adrenaline + Compound A51 (50 mg/kg).• Group 3 (A52): Adrenaline + Compound A52 (25 mg/kg).• Group 4 (A54): Adrenaline + Compound A54 (50 mg/kg).Statistical Analysis: Data are expressed as mean ± Standard Error of the Mean (SEM). The normality of data distribution was assessed using the Shapiro-Wilk test. Comparisons between multiple groups were performed using One-way Analysis of Variance (ANOVA) followed by Tukey’s post-hoc test for pairwise comparisons. A p-value of < 0.05 was considered statistically significant. All statistical calculations were conducted using GraphPad Prism 9.0 or SPSS version 26.0 software.
3. Results
In the preceding sections, the compounds A51, A52, and A54 were individually evaluated under in vivo conditions, with their antihypertensive effects analyzed across specific dosage levels (25 mg/kg and 50 mg/kg). These studies were conducted using an adrenaline-induced hypertension model in rats, and the results were assessed based on the dynamic changes in systolic and diastolic blood pressure (SBP/DBP) parameters. The analysis further focused on the temporal progression of each compound's effect on blood pressure, including the duration of action, peak efficacy (maximal effect), and recovery trends[1]. In this section, the in vivo activity of all three compounds is comparatively evaluated, with a comprehensive discussion of their therapeutic potential, optimal dosages, and pharmacodynamic profiles. Through this comparative analysis, the most effective, stable, and safe candidate among A51, A52, and A54 is identified based on their antihypertensive efficacy. This evaluation serves as the primary selection criterion for subsequent pre-clinical studies [2,6]. In vivo evaluation of the antihypertensive activity of compound A51In order to further validate the in vitro and in silico results presented in the preceding sections and to evaluate the efficacy of the compound within a real biological system, in vivo experimental studies were conducted. This approach is essential for determining the actual physiological impact of the preparations and assessing their pharmacodynamic characteristics, which is particularly critical for compounds targeting the cardiovascular system [3,8]. In vivo experiments were conducted on laboratory rats (n=3), utilizing the non-invasive tail-cuff method for blood pressure measurement. Initially, the therapeutic dose levels of compound A51 were identified; subsequently, the antihypertensive efficacy of the substance was evaluated in animal models with adrenaline-induced hypertension. Within the experimental design, two dosage levels were selected: 25 mg/kg and 50 mg/kg. The preparation was administered to the animals intravenously via the tail vein.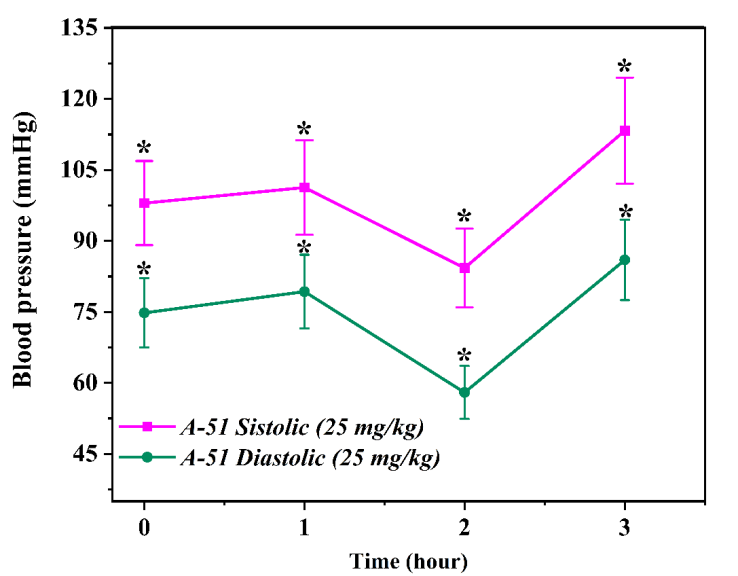 | Figure 1. Changes in systolic and diastolic blood pressure following administration of A-51 (25 mg/kg) over a 3-hour period. Effect of compound A-51 (mg/kg) on systolic and diastolic blood pressure. The graph illustrates the changes in blood pressure over time (hours). Each data point is expressed as mean ± SEM (n = 3). *p < 0.05 indicates a statistically significant difference compared to the control group |
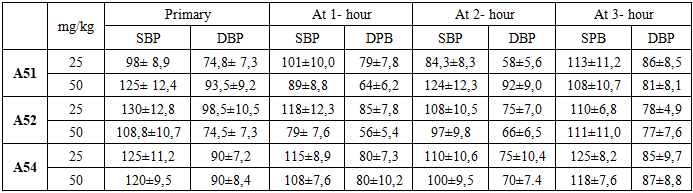
The effect of compound A51 at a dose of 25 mg/kgAt the beginning of the experiment, the rats' systolic blood pressure (SBP) was recorded at 98.0 ± 8.9 mmHg, while the diastolic blood pressure (DBP) stood at 74.8 ± 7.3 mmHg. One hour after the administration of the preparation, the SBP rose to 101.3 ± 10.0 mmHg and the DBP increased to 79.3 ± 7.8 mmHg. This observation indicates that the compound caused a slight, short-term increase in blood pressure during the initial phase of its action [4]. However, measurements recorded two hours post-administration revealed a significant reduction, with systolic blood pressure (SBP) decreasing to 84.3 ± 8.3 mmHg and diastolic blood pressure (DBP) to 58.0 ± 5.6 mmHg. This decline indicates that compound A51 exerts potent antihypertensive activity over time. By the third hour, a slight rebound in blood pressure was observed, with SBP reaching 113.3 ± 11.2 mmHg and DBP at 86.0 ± 8.5 mmHg. This trend suggests either the metabolic degradation of the compound or a limited duration of action [5,9]. 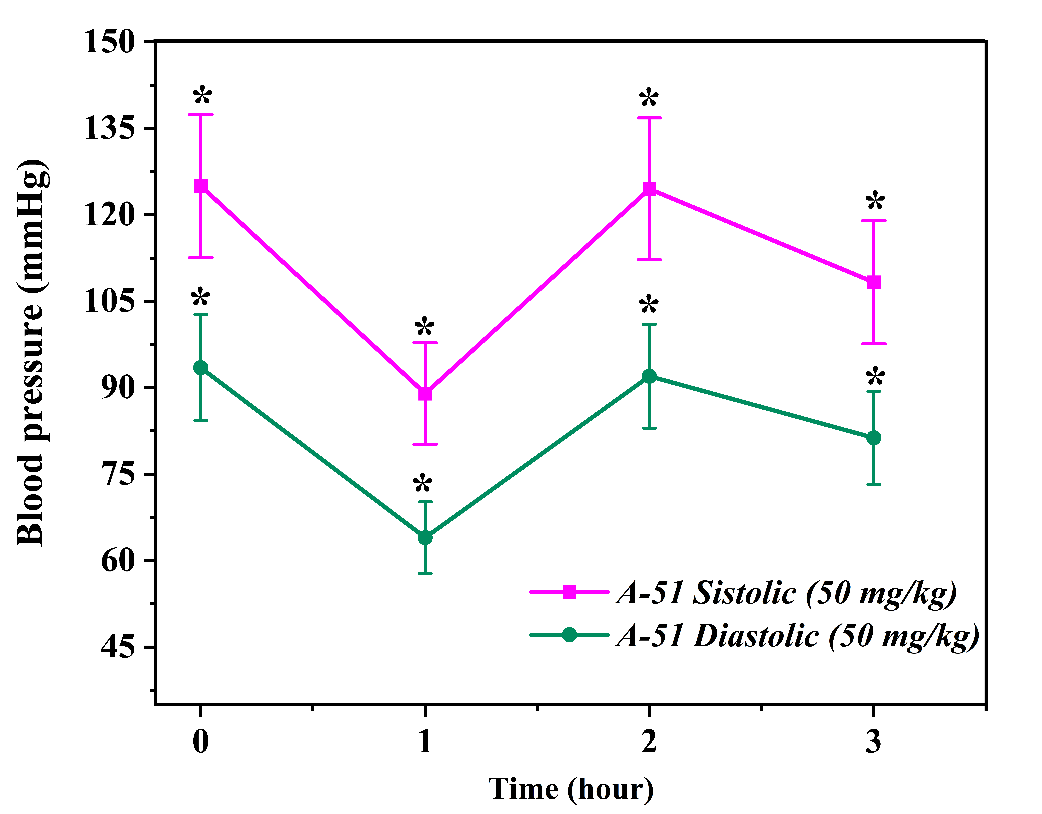 | Figure 2. Time-dependent changes in systolic and diastolic blood pressure following administration of A-51 (50 mg/kg) over a 3-hour period. Effect of compound A-51 at a dose of 50 mg/kg on systolic and diastolic blood pressure. The graph illustrates the changes in blood pressure over time (hours). Each data point is expressed as mean ± SEM (n = 3). *p < 0.05 indicates a statistically significant difference compared to the control group |
The effect of compound A51 at a dose of 50 mg/kgIn the next stage, the effect of compound A51 at a higher dosage (50 mg/kg) was evaluated. At baseline, under the conditions of the hypertensive model, the systolic blood pressure (SBP) was 125.0 ± 12.4 mmHg and the diastolic blood pressure (DBP) was 93.5 ± 9.2 mmHg. One hour after the administration of the preparation, the SBP decreased to 89.0 ± 8.8 mmHg and the DBP fell to 64.0 ± 6.2 mmHg. These values demonstrate the rapid-acting capability of compound A51.In the second hour, a rebound in blood pressure was observed, with systolic blood pressure (SBP) recorded at 124.5 ± 12.3 mmHg and diastolic blood pressure (DBP) at 92.0 ± 9.0 mmHg. This dynamic change suggests a transient effect of the preparation and the potential activation of compensatory mechanisms. After three hours, the blood pressure decreased slightly once more, with SBP reaching 108.3 ± 10.7 mmHg and DBP at 81.3 ± 8.1 mmHg, indicating a restoration of the compound's effect. Based on these results, the 50 mg/kg dose of compound A51 was selected for further in-depth experiments in the hypertension model, as it demonstrated a more stable and effective antihypertensive response. This finding serves as compelling evidence validating the antihypertensive potential of compound A51 within the cardiovascular system [10,12]. In vivo evaluation of the antihypertensive effect of compound A52In order to analyze the anticipated pharmacological activity of compound A52, validate the positive results obtained from prior in vitro and in silico testing, and demonstrate its efficacy within a biological system, comprehensive experiments were conducted under in vivo modeled hypertensive conditions. At this stage, the animal studies were focused on determining the direct impact of compound A52 on the cardiovascular system. Throughout the experiments, the therapeutic dosage range, onset of action, and duration of the compound's effect were evaluated. The experiments were conducted on healthy male Wistar rats (n=3). Prior to the study, all physiological conditions of the animals were subject to medical supervision. A non-invasive tail-cuff method was employed to determine blood pressure. Two dosage levels of compound A52—25 mg/kg and 50 mg/kg—were selected, and the preparations were administered to the rats intravenously (via the tail vein). The time-dependent dynamic effect of each dose was evaluated by analyzing blood pressure parameters at 1, 2, and 3-hour intervals post-administration [9,10]. The effect of compound A52 at a dose of 25 mg/kg on the cardiovascular system was initially evaluated. At baseline, the rats in this group exhibited a systolic blood pressure (SBP) of 130.0 ± 12.8 mmHg and a diastolic blood pressure (DBP) of 98.5 ± 10.5 mmHg. These values reflect a typical hypertensive state within the model. Following administration, a significant reduction in blood pressure was observed during the first hour; SBP decreased to 118.8 ± 12.3 mmHg, while DBP fell to 85.8 ± 7.8 mmHg. This dynamic decline reflects the rapid onset of action of the compound. 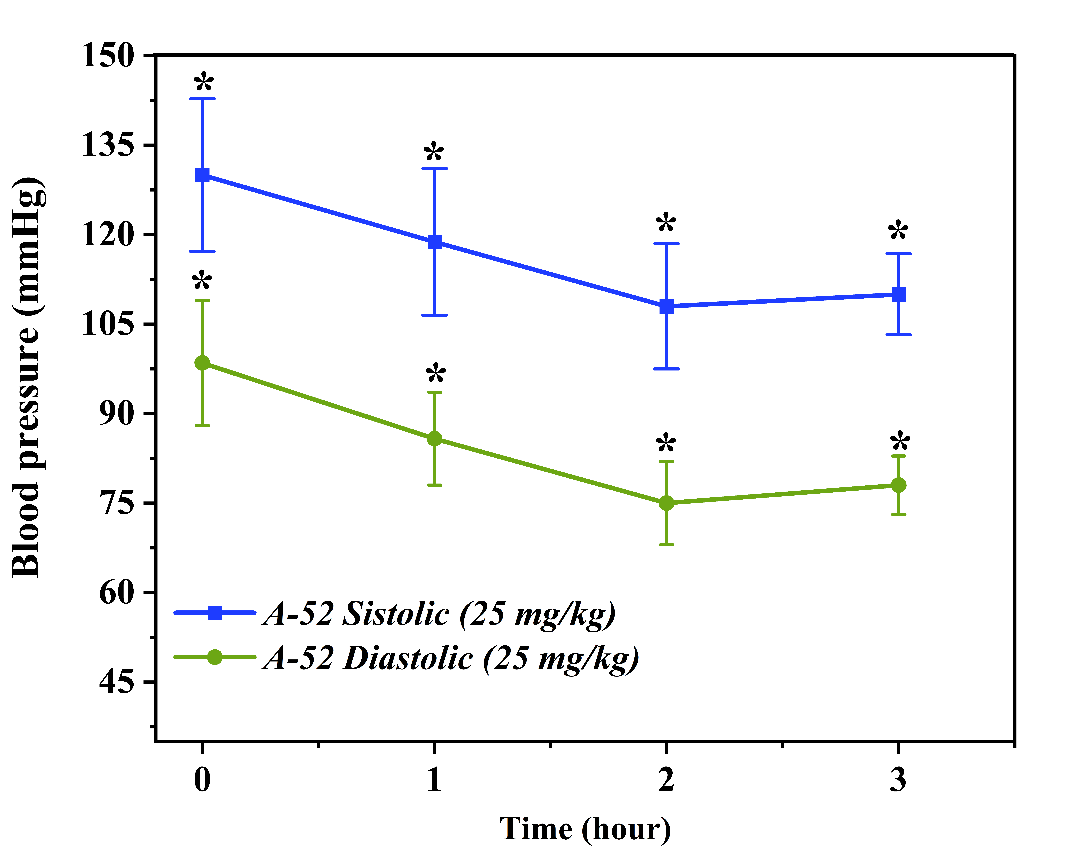 | Figure 3. Time-dependent changes in systolic and diastolic blood pressure following administration of A-52 (25 mg/kg) over a 3-hour period. Effect of compound A-52 at a dose of 25 mg/kg on systolic and diastolic blood pressure. The graph illustrates the changes in blood pressure over time (hours). Each data point is expressed as mean ± SEM (n = 3). *p < 0.05 indicates a statistically significant difference compared to the control group |
By the second hour, a further reduction in blood pressure was recorded, with SBP reaching 108.0 ± 10.5 mmHg and DBP at 75.0 ± 7.0 mmHg. This stage marked the peak antihypertensive effect of the compound. The sustained decline in blood pressure suggests that compound A52 may possess vasodilatory or calcium channel blocking properties. This, in turn, indicates the potential for A52 to exert either a direct or indirect positive influence on endothelial function. By the third hour, although a slight increase in blood pressure was observed, levels remained below the baseline: SBP was recorded at 110.0 ± 6.8 mmHg and DBP at 78.0 ± 4.9 mmHg. This suggests that the compound exhibits prolonged, albeit moderate, antihypertensive activity. Overall, this dosage demonstrated an optimal antihypertensive effect and was selected as the target dose for further in-depth research. In the subsequent stage, the effect of compound A52 at a higher dosage of 50 mg/kg was evaluated. This dose was pharmacologically anticipated to exert a more potent effect; however, the experimental results did not fully support this hypothesis. The baseline blood pressure in this group was relatively low, with SBP at 108.8 ± 10.7 mmHg and DBP at 74.5 ± 7.3 mmHg. This observation suggests that the hypertension model may not have developed uniformly across all experimental groups. 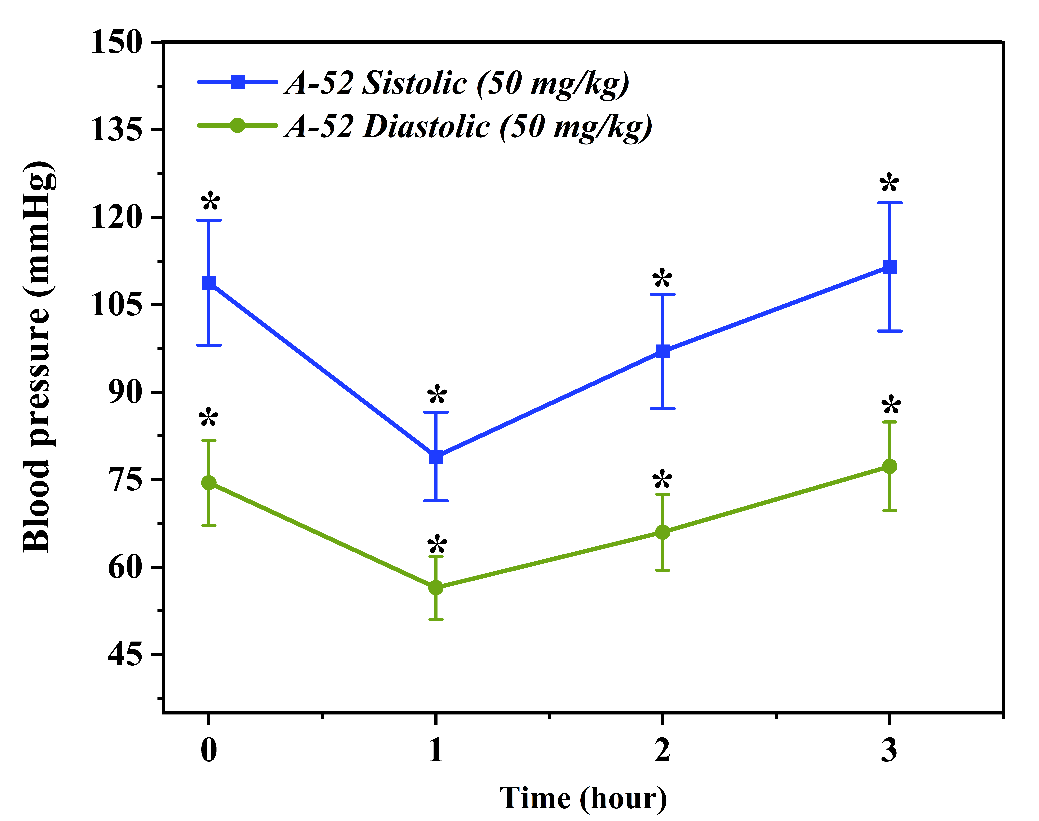 | Figure 4. Time-dependent changes in systolic and diastolic blood pressure following administration of A-52 (50 mg/kg) over a 3-hour period. Effect of compound A-52 at a dose of 50 mg/kg on systolic and diastolic blood pressure. The graph illustrates the changes in blood pressure over time (hours). Each data point is expressed as mean ± SEM (n = 3). *p < 0.05 indicates a statistically significant difference compared to the control group |
Following administration, a sharp decline in blood pressure was observed within the first hour: SBP fell to 79.0 ± 7.6 mmHg and DBP to 56.5 ± 5.4 mmHg. This reduction approached the threshold of hypotension, which may be pharmacologically hazardous. Such a profound decrease in blood pressure could be attributed to either a reduction in cardiac output or excessive peripheral vasodilation. By the second hour, pressure parameters began to partially recover, with SBP rising to 97.0 ± 9.8 mmHg and DBP to 66.0 ± 6.5 mmHg. By the third hour, the values reached 111.5 ± 11.0 mmHg and 77.3 ± 7.6 mmHg, respectively. This trend indicates a waning of the preparation's effect over time and the activation of the body's physiological compensatory mechanisms. However, the acute drop in blood pressure at the 50 mg/kg dose, coupled with the subsequent lack of stability, suggests that the compound may be potentially toxic or hazardous at this dosage. Consequently, this dose was excluded from further experiments. Furthermore, these results indicate the existence of a'therapeutic window' for compound A52 in the dose-response relationship, suggesting that excessive dosing may diminish the desired therapeutic effect. Evaluation of the antihypertensive activity of substance A54 in vivoIn vivo studies conducted to determine the effects of substance A54 on the cardiovascular system served to identify the pharmacodynamic properties of this compound and to experimentally validate previous in vitro and in silico results. At this stage, the dose-dependent antihypertensive activity of substance A54 was evaluated through animal experiments, and the duration and stability of its action were analyzed [7,9]. Experiments were conducted on healthy male Wistar rats (n=3). A non-invasive tail-cuff technique was employed to measure blood pressure. Two distinct doses of substance A54 were selected: 25 mg/kg and 50 mg/kg. The compound was administered intravenously via the tail vein. Blood pressure was recorded at 0 hours (baseline), 1 hour, 2 hours, and 3 hours for each dosage group, with systolic (SBP) and diastolic (DBP) parameters being evaluated separately. In the experimental phase using a 25 mg/kg dose, the rats' baseline systolic blood pressure (SBP) 125,0 ± 11,2 mmHg, and the diastolic blood pressure (DBP) was 90,7 ± 7,2 mmHg. These values are characteristic of hypertension. Following administration of the substance, SBP decreased to 115,8 ± 9,9 mmHg and DBP to 80,7 ± 3,3 mmHg within the first hour. This reduction indicates that the compound possesses rapid-acting properties. 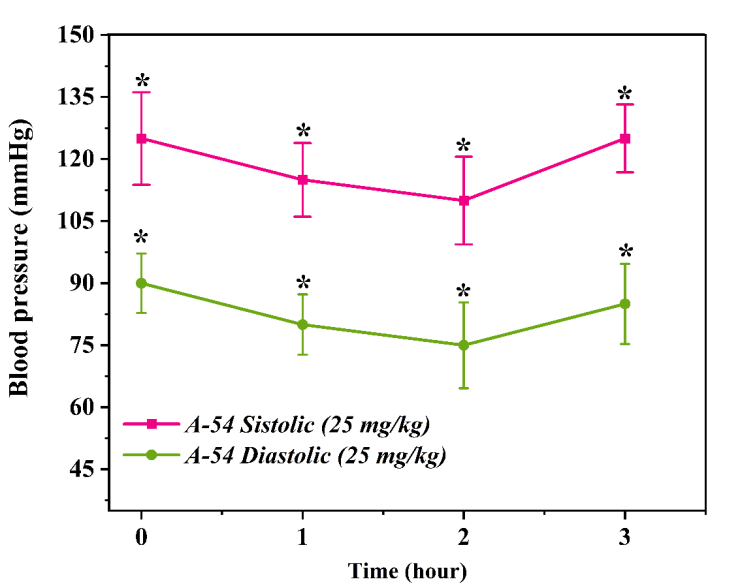 | Figure 5. Time-dependent changes in systolic and diastolic blood pressure following administration of A-54 (25 mg/kg) over a 3-hour period. Effect of A-54 substance at a dose of 25 mg/kg on systolic and diastolic blood pressure. The graph illustrates the changes in blood pressure over time (hours). Each point is expressed as mean ±SEM (n = 3). p < 0.05 indicates a statistically significant difference compared to the control group |
By the second hour, the reduction in blood pressure became even more pronounced, with SBP falling to 110.0 ± 10.6 mmHg and DBP to 75.0 ± 10.4 mmHg. This indicates that the dosage exerted a significant yet controlled antihypertensive effect. However, by the third hour, the blood pressure returned to its baseline levels, with SBP recorded at 125.0 ± 8.2 mmHg and DBP at 85.9 ± 7.7 mmHg. This trend suggests that the effect of the preparation was transient and began to wane after the second hour. The lack of a sustained effect implies that the therapeutic efficacy of compound A54 at a dose of 25 mg/kg may be limited. The effect of compound A54 at a higher dosage of 50 mg/kg was also analyzed. In this group, the baseline blood pressure was recorded at SBP 120.0 ± 9.5 mmHg and DBP 90.0 ± 8.4 mmHg, which is consistent with the hypertensive model. One hour after administration, the SBP decreased to 108.0 ± 7.6 mmHg, while the DBP fell to 80.0 ± 10.2 mmHg. This represents a moderate reduction in blood pressure, indicating that the compound exerts its effect safely even at a higher dosage. By the second hour, the blood pressure fell to SBP 100.0 ± 9.5 mmHg and DBP 70.0 ± 7.4 mmHg. At this stage, the peak antihypertensive effect was observed, demonstrating a dose-dependent pharmacological response. By the third hour, the blood pressure parameters had partially recovered, with SBP recorded at 118.0 ± 7.6 mmHg and DBP at 87.0 ± 8.8 mmHg. Unlike the 25 mg/kg dose, this observation indicates that the 50 mg/kg dose of the preparation exhibits a longer-lasting effect. Table 1. Effects of A-51, A-52, and A-54 at doses of 25 mg/kg and 50 mg/kg on systolic (SBP) and diastolic (DBP) blood pressure measured at baseline and over a 3-hour period
 |
| |
|
In this section, compounds A51, A52, and A54 were evaluated using an adrenaline-induced hypertension model. For this purpose, the rats were divided into four experimental groups. The first group served as the control group, receiving only adrenaline without any supplemental compounds. The remaining three groups were administered compounds A51 (50 mg/kg), A52 (25 mg/kg), and A54 (50 mg/kg), respectively. Each group consisted of 3–4 rats. A 3-hour observation period was maintained throughout the experiments, during which changes in systolic (SBP) and diastolic (DBP) blood pressure parameters were systematically recorded.
4. Conclusions
In this study, the in vivo antihypertensive effects of compounds A51 and A52 were evaluated across different dosage levels (25 mg/kg and 50 mg/kg) using a rat model.• For Compound A51: The results demonstrated that while the 25 mg/kg dose provided a temporary reduction in blood pressure, it lacked long-term stability. In contrast, the 50 mg/kg dose exhibited a more consistent and potent antihypertensive response, making it the optimal choice for further in-depth cardiovascular research.• For Compound A52: The 25 mg/kg dose showed a significant and stable reduction in blood pressure, reaching its peak effect in the second hour. This suggests potential vasodilatory or calcium channel blocking mechanisms that positively influence endothelial function. However, increasing the dose to 50 mg/kg led to an acute and potentially hazardous drop in blood pressure (approaching hypotension) followed by physiological instability.The comparative analysis indicates that the therapeutic efficacy of these compounds is strictly dose-dependent. For A51, the 50 mg/kg dose is most effective, whereas for A52, the 25 mg/kg dose represents the optimal therapeutic window. These findings provide compelling evidence for the antihypertensive potential of both compounds and establish a foundation for their further pharmacological development as cardiovascular agents.
References
| [1] | Wang Y, Thatcher SE, Cassis LA. Measuring Blood Pressure Using a Noninvasive Tail Cuff Method in Mice. Methods Mol Biol. 2017; 1614: 69-73. doi: 10.1007/978-1-4939-7030-8_6. PMID: 28500596. |
| [2] | Wilde E, Aubdool AA, Thakore P, Baldissera L Jr, Alawi KM, Keeble J, Nandi M, Brain SD. Tail-Cuff Technique and Its Influence on Central Blood Pressure in the Mouse. J Am Heart Assoc. 2017 Jun 27; 6(6): e005204. doi: 10.1161/JAHA.116.005204. PMID: 28655735; PMCID: PMC5669161. |
| [3] | Sia JG, Schwam E, Mann W, Sepinwall J. Tail-cuff method for measuring systolic bold pressure in squirrel monkeys. Physiol Behav. 1975 May; 14(5): 659-62. doi: 10.1016/0031-9384(75)90196-1. PMID: 806086. |
| [4] | Buñag RD. Validation in awake rats of a tail-cuff method for measuring systolic pressure. J Appl Physiol. 1973 Feb; 34(2): 279-82. doi: 10.1152/jappl.1973.34.2.279. PMID: 4686367. |
| [5] | Buñag RD, Mueting N, Riley E. Regional vascular influences on tail-cuff measurements of drug-induced changes in systolic pressure. J Appl Physiol. 1975 Nov; 39(5): 724-7. doi: 10.1152/jappl.1975.39.5.724. PMID: 1184511. |
| [6] | Buñag RD, Riley E. Simultaneous measurements in awake rats of drug-induced changes in carotid and tail-cuff systolic pressures. J Appl Physiol. 1974 May; 36(5): 621-4. doi: 10.1152/jappl.1974.36.5.621. PMID: 4363612 |
| [7] | Fritz M, Rinaldi G. Blood pressure measurement with the tail-cuff method in Wistar and spontaneously hypertensive rats: influence of adrenergic- and nitric oxide-mediated vasomotion. J Pharmacol Toxicol Methods. 2008 Nov-Dec; 58(3): 215-21. doi: 10.1016/j.vascn.2008.08.002. Epub 2008 Aug 13. PMID: 18765287. |
| [8] | Fritz M, Rinaldi G. Influence of nitric oxide-mediated vasodilation on the blood pressure measured with the tail-cuff method in the rat. J Biomed Sci. 2007 Nov; 14(6): 757-65. doi: 10.1007/s11373-007-9191-1. Epub 2007 Jul 18. PMID: 17634759. |
| [9] | Cimini CM, Zambraski EJ. Non-invasive blood pressure measurement in Yucatan miniature swine using tail cuff sphygmomanometry. Lab Anim Sci. 1985 Aug; 35(4): 412-6. PMID: 3900580. |
| [10] | Zatz R. A low cost tail-cuff method for the estimation of mean arterial pressure in conscious rats. Lab Anim Sci. 1990 Mar; 40(2): 198-201. PMID: 2157103. |
| [11] | Franklin SS. Ageing and hypertension: the assessment of blood pressure indices in predicting coronary heart disease. J Hypertens Suppl. 1999 Dec; 17(5): S29-36. PMID: 10706323. |
| [12] | Feng M, DiPetrillo K. Non-invasive blood pressure measurement in mice. Methods Mol Biol. 2009; 573: 45-55. doi: 10.1007/978-1-60761-247-6_3. PMID: 19763921. |


 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML




