-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Virology and Molecular Biology
p-ISSN: 2163-2219 e-ISSN: 2163-2227
2026; 15(2): 43-46
doi:10.5923/j.ijvmb.20261502.04
Received: Mar. 22, 2026; Accepted: Apr. 16, 2026; Published: Apr. 30, 2026

Taxonomic Inventory, Trophic Guild Structure and Alpha-Diversity of the Entomofauna of Tugay Forests in the Lower Amudarya Basin, Khorezm Region, Uzbekistan
Madina Allayarova1, Lola Gandjayeva2, Nazirakhon Allayarova3
1PhD., Department of Natural Sciences, Khorezm Ma’mun Academy, Khiva, Uzbekistan
2DSc., Professor, Department of Chemistry, Tashkent International University of Chemistry, Tashkent, Uzbekistan
3PhD., National Centre for Agricultural Knowledge and Innovation, Research Institute of Plant Genetic Resources, Tashkent, Uzbekistan
Correspondence to: Madina Allayarova, PhD., Department of Natural Sciences, Khorezm Ma’mun Academy, Khiva, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

(1) Background: The tugay riparian forests of the lower Amudarya in Khorezm Region, Uzbekistan, harbour a biogeographically significant but poorly inventoried entomofauna whose functional structure remains undocumented. (2) Methods: A three-year standardised survey (2023–2025) was conducted at three localities spanning the principal hydrological gradient of the region, employing sweep-net transects, bark-and-litter hand searching and UV light-trapping. Population density was computed as D = N S⁻¹ (ind. m⁻²); community diversity was characterised by the Shannon–Wiener entropy (H’), Simpson’s concentration index (D) and the Margalef richness estimator (Mg). (3) Results: The survey yielded 19 species across four orders (Heteroptera, Coleoptera, Orthoptera, Lepidoptera), ten families and 18 genera. Coleoptera was the most species-rich order (42.1%). Arboreal biotopes harboured the greatest assemblage richness (11 species, 57.9%); trophic guild analysis assigned 10 species to the phytophagous, 6 to the entomophagous and 3 to the saprophagous guild. Shannon entropy ranged from H’ = 2.48 to 2.71; Pearson correlation between vegetation density and species richness was r = 0.94 (χ² = 6.84, df = 4, p ≤ 0.05). The pentatomid Mustha gigantea (Fabricius, 1794) is recorded for the first time from Uzbekistan; 14 additional species are new for Khorezm Region. (4) Conclusions: These results establish a quantitative entomofaunal baseline and confirm the primacy of intact arboreal tugay as a conservation priority in the lower Amudarya basin.Keywords Lower Amudarya tugay, Insect species inventory, Trophic guild analysis, Mustha gigantea, First provincial record, Riparian alpha-diversity, Xero-halophytic shrubland entomofauna
Keywords:
Cite this paper: Madina Allayarova, Lola Gandjayeva, Nazirakhon Allayarova, Taxonomic Inventory, Trophic Guild Structure and Alpha-Diversity of the Entomofauna of Tugay Forests in the Lower Amudarya Basin, Khorezm Region, Uzbekistan, International Journal of Virology and Molecular Biology, Vol. 15 No. 2, 2026, pp. 43-46. doi: 10.5923/j.ijvmb.20261502.04.
Article Outline
1. Introduction
- Among the most biogeographically distinctive terrestrial ecosystems of the Palaearctic arid belt, the tugay riparian woodlands fringing the rivers of Central Asia have long attracted botanical and geobotanical attention disproportionate to the interest directed at their invertebrate faunas [1,2]. In Uzbekistan, the residual tugay stands concentrated along the lower Amudarya represent a floristically and structurally attenuated remnant of a formerly far more extensive riparian forest zone, reduced over the past five decades by irrigation-driven flow diversion, direct vegetation clearance and the progressive salinisation of floodplain soils [2,3]. The entomological consequences of this contraction — in terms of species loss, trophic restructuring and reduced functional redundancy — remain almost entirely undocumented [6,7].Insects are quantitatively and functionally the most significant animal component of tugay ecosystems. Phytophagous guilds — notably the xylophagous Cerambycidae and the sucking-feeding Pentatomidae and Pyrrhocoridae — regulate primary productivity through herbivory and cambium exploitation; entomophagous Carabidae and Coccinellidae impose top-down control on herbivore populations; and saprophagous taxa participate directly in the decomposition of woody and herbaceous litter [4]. The composition of these guilds integrates information on vegetation structure, thermal and moisture regime and disturbance history in ways that single-taxon surveys cannot replicate, thereby endowing comprehensive entomofaunal inventories with a diagnostic utility extending well beyond simple species accounting [5].The present study — the faunistic complement to a methodological companion paper [Allayarova et al., in press, IJGE] — addresses this deficit through: (i) a complete annotated taxonomic inventory of the 19 insect species recorded, with identification of new national and provincial records; (ii) the distribution of species across three operationally defined biotope classes; (iii) a trophic guild analysis of the assembled fauna; (iv) the characterisation of seasonal and inter-annual dynamics in population density; and (v) site-level alpha-diversity estimates generated from three complementary indices.
2. Materials and Methods
- Field collections were undertaken at three tugay localities in Khorezm Region, Uzbekistan: Gurlan district (41.84° N, 60.39° E), representative of near-natural arboreal tugay retaining close hydrological connectivity with the main Amudarya channel; Urgench district (41.55° N, 60.63° E), a moderately disturbed arboreal–shrub stand affected by urban expansion; and Shovot district (41.66° N, 60.29° E), a xero-halophytic shrub-dominated fragment with evidence of historical overgrazing and progressive salinisation. All sampling was restricted to the May–September growing season, with each locality receiving a minimum of five visits per season at approximately four-week intervals.Three complementary sampling methods were deployed concurrently at each visit: standardised sweep-net transects (10–20 m, 10–15 sweeps per transect) targeting mobile taxa in the herbaceous and lower shrub strata; systematic bark-and-litter hand searching for cryptic cerambycid and carabid beetles; and UV light-trapping (dusk to midnight, two non-consecutive nights per visit) for nocturnally active Lepidoptera. Specimens were fixed in 70% ethanol (v/v) or dry-pinned according to taxon, and identified to species under a stereobinocular microscope (10–40×) using the determinative keys of Bey-Bienko [8] and Kerzhner & Yachevskiy [9]. Nomenclature follows the Catalogue of Palaearctic Coleoptera (Löbl & Löbl, 2017) and Fauna Europaea (de Jong et al., v.3).Population density was computed as D = N S⁻¹ (ind. m⁻²), where N is the individual count per sampling event and S is the effective sampled area (estimated as 2 m² per 10 sweeps for the net method and as the measured search area for hand-searching). Encounter-frequency-based dominance categories followed Pesenko [10]: dominant (>15%), subdominant (5–15%) and rare (<5%). Alpha-diversity was characterised by the Shannon–Wiener entropy H’ = −Σ(pᵢ ln pᵢ), Simpson’s concentration D = 1 − Σpᵢ² and the Margalef richness estimator Mg = (S − 1)(ln N)⁻¹. The association between canopy cover density and species richness was tested by Pearson product-moment correlation and Pearson’s chi-squared in R v.4.3.0.
3. Result and Discussion
3.1. Taxonomic Composition and Faunistic Novelties
- The pooled three-year dataset comprises 19 species distributed across four orders, ten families and 18 genera (Table 1). Coleoptera was the most species-rich order (eight species, 42.1%), a result congruent with the well-documented tendency for beetles to dominate species counts in structurally complex temperate and sub-temperate riparian woodlands, where the diversity of woody substrates, bark micro-niches and soil–litter interfaces collectively supports an unusually broad spectrum of feeding specialisations [11]. Heteroptera followed with five species (26.3%), all assigned to two families — Pentatomidae and Pyrrhocoridae. Orthoptera and Lepidoptera each contributed three species (15.8% each).
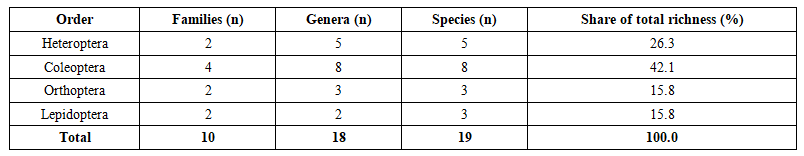 | Table 1. Taxonomic breakdown of insect species recorded in Khorezm Region tugay forests, 2023–2025 |
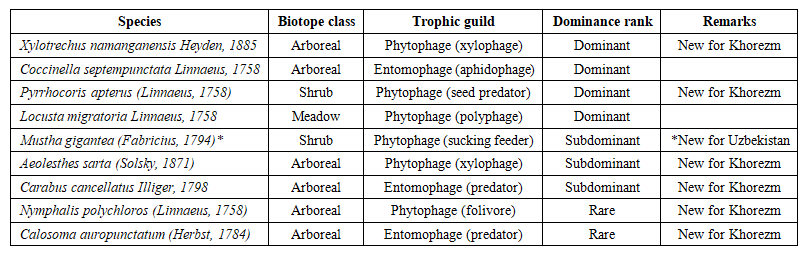 | Table 2. Selected species: biotope class, trophic guild, dominance category and distributional status (* first record for Uzbekistan; all other listed taxa new for Khorezm Region) |
3.2. Biotope-Level Distribution of Species
- Species richness exhibited a strongly biotope-dependent pattern, declining from 11 species in arboreal biotopes (57.9% of total) through five in shrub biotopes (26.3%) to three in meadow biotopes (15.8%). At locality level, Gurlan supported 15 species (78.9%), Urgench 14 (73.7%) and Shovot 12 (63.2%), a rank order mirroring the gradient in arboreal canopy cover from the river-connected to the hydrologically isolated end of the locality sequence. The Pearson correlation between vegetation canopy density and species richness attained r = 0.94 (χ² = 6.84, df = 4, p ≤ 0.05), confirming that plant community physiognomy is the dominant proximal predictor of entomofaunal richness across the study localities [12].The arboreal biotope owes its species-richness superiority to the simultaneous availability of woody substrates colonised by xylophagous Cerambycidae (X. namanganensis, A. sarta, C. faldermanni, T. scabrum), litter–soil interfaces exploited by predatory Carabidae (Carabus cancellatus Illiger, 1798; Calosoma auropunctatum (Herbst, 1784)), and foliar resources supporting pentatomid and lepidopteran folivores. Shrub biotopes supported an almost exclusively phytophagous hemipteran fauna, and meadow biotopes were dominated by three acridid grasshoppers — Locusta migratoria L., Calliptamus italicus (L.) and Gryllus desertus Pallas, 1771.
3.3. Trophic Guild Structure
- Assignment of species to trophic guilds yielded ten phytophagous taxa (52.6%), six entomophagous taxa (31.6%) and three saprophagous taxa (15.8%). The numerical preponderance of phytophages reflects both the diversity of plant resources available in structurally heterogeneous tugay and the general tendency of phytophagous guilds to attain higher species richness than carnivores in most terrestrial ecosystems. The entomophagous component, represented primarily by polyphagous carabid ground beetles and aphidophagous coccinellids, indicates the presence of a functional biocontrol assemblage capable of exerting top-down regulation on herbivore populations. The saprophagous guild, whilst numerically modest, performs an ecologically indispensable role in the mineralisation of the organic matter fraction of tugay soils.Dominance analysis placed eight species in the dominant category (encounter frequency >15%) — including all three acridid Orthoptera and the hemipterans Pyrrhocoris apterus (L.), Aelia acuminata (L.) and Dolycoris baccarum (L.) — with seven subdominant (5–15%) and four rare (<5%) taxa completing the assemblage. This distributional structure, in which a core of ecologically versatile, habitat-generalist species accounts for the majority of encounters whilst a peripheral set of microhabitat-specialist taxa contributes substantially to overall richness, is broadly consistent with the lognormal rank-abundance model typical of moderately diverse arthropod assemblages in continental climates.
3.4. Seasonal and Inter-Annual Population Dynamics
- Mean population density across all localities followed a unimodal seasonal trajectory: 12.4 ind. m⁻² in spring (May–June), rising to a summer peak of 21.6 ind. m⁻² (July–August) and subsiding to 15.3 ind. m⁻² in autumn (September). This pattern is a predictable consequence of the sharply continental thermal regime of Khorezm [13], in which the convergence of maximum ambient temperature, peak aboveground plant biomass and maximum food-plant leaf area index during mid-summer simultaneously accelerates development rates and drives peak reproductive activity across multiple taxonomic groups.Inter-annual trends in both density and richness were consistently positive: mean seasonal density increased from 15.8 ind. m⁻² in 2023 through 16.6 in 2024 to 17.2 ind. m⁻² in 2025, tracking a parallel increase in cumulative species richness from S = 13 (2023) through S = 16 (2024) to S = 19 (2025). These increments most plausibly reflect the combined effect of progressive accumulation of sampling effort — with rare species becoming detectable only as sample sizes increase — alongside year-on-year expansion of the faunistically sampled habitat volume.
3.5. Alpha-Diversity and Comparison with Regional Data
- Alpha-diversity metrics computed at locality level (Table 3) indicate moderate to high, broadly comparable diversity across all three research sites. Shannon entropy (H’ = 2.48–2.71), Simpson’s concentration (D = 0.84–0.89) and the Margalef richness index (Mg = 2.76–3.14) concur in ranking Gurlan highest and Shovot lowest, entirely consistent with the vegetation physiognomy gradient. The consistently elevated Simpson D values (≥0.84) across all sites indicate that no single taxon exerts overwhelming numerical dominance, implying a relatively equitable distribution of individuals — a characteristic associated with ecologically intact or only moderately disturbed arthropod assemblages.
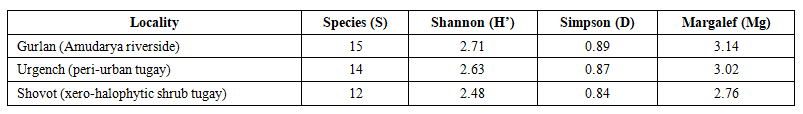 | Table 3. Alpha-diversity indices for insect assemblages recorded at three Khorezm Region tugay localities, 2023–2025 |
4. Conclusions
- The three-year entomological survey of lower Amudarya tugay forests in Khorezm Region yielded a 19-species inventory spanning four orders, with Coleoptera accounting for the greatest proportion of species richness. The assemblage is organised around a phytophage-dominated core, a functionally significant entomophage complement and a modest but ecologically essential saprophage contingent. The first record of Mustha gigantea from Uzbekistan and 14 additional new records for Khorezm Region represent concrete additions to knowledge of the regional fauna. The steep positive association between vegetation structural complexity and species richness (r = 0.94) underscores the primacy of intact arboreal tugay as a conservation priority and provides a quantitative basis for predicting the entomological costs of further canopy loss under scenarios of progressive aridification and land-use intensification.
ACKNOWLEDGEMENTS
- The authors are indebted to the Khorezm Ma’mun Academy for provision of laboratory facilities and logistical support throughout the field campaigns, and to the technical staff of the Research Institute of Plant Genetic Resources for assistance with specimen archiving. The authors declare no competing interests.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML