-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Virology and Molecular Biology
p-ISSN: 2163-2219 e-ISSN: 2163-2227
2026; 15(2): 34-42
doi:10.5923/j.ijvmb.20261502.03
Received: Feb. 17, 2026; Accepted: Mar. 10, 2026; Published: Apr. 25, 2026

Microbial Fibrinolytic Enzymes as Promising Thrombolytic Agents: Molecular Mechanisms, Functional Diversity and Therapeutic Perspectives
Khushnut Vokhidov, Akhmadjan Makhsumkhanov, Askar Kholikov, Barno Alimova, Ozodakhon Pulatova
Institute of Microbiology, Academy of Sciences of the Republic of Uzbekistan, Tashkent, Uzbekistan
Correspondence to: Khushnut Vokhidov, Institute of Microbiology, Academy of Sciences of the Republic of Uzbekistan, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Thrombotic cardiovascular and cerebrovascular diseases remain among the leading causes of global morbidity and mortality, highlighting the persistent need for safer and more effective thrombolytic therapies. Although clinically approved plasminogen activators, such as streptokinase and tissue-type plasminogen activator (tPA), have substantially improved acute thrombolytic management, their clinical utility is limited by systemic fibrinolysis, hemorrhagic complications, immunogenicity, and narrow therapeutic windows. These limitations underscore a critical gap in current thrombolytic strategies that has not been fully addressed by existing therapeutic agents. In recent years, microbial fibrinolytic enzymes have emerged as promising alternatives due to their structural diversity, unique mechanisms of action, and potential for improved fibrin specificity. However, previous reviews have largely focused on descriptive enzyme catalogs, with limited emphasis on mechanistic classification, translational barriers, and engineering-driven optimization strategies that are essential for clinical advancement. This review provides a comprehensive and critical synthesis of microbial-derived fibrinolytic and thrombolytic enzymes, classifying them into direct fibrinolytic proteases, indirect plasminogen activators, and mixed-mechanism enzymes. We integrate biochemical properties, microbial sources, and structure–function relationships with advances in recombinant production and protein engineering, highlighting how these approaches can address key translational challenges. Importantly, immunogenicity, pharmacokinetic limitations, safety concerns, and regulatory constraints are critically evaluated to delineate the major barriers preventing clinical implementation. By bridging microbial biodiversity with modern bioengineering and translational pharmacology, this review identifies priority directions for the development of next-generation thrombolytic agents. We propose that rationally engineered, fibrin-targeted microbial enzymes—particularly those with mixed mechanisms of action—represent the most promising candidates to overcome the efficacy–safety trade-off that has historically constrained thrombolytic therapy.
Keywords: Microbial fibrinolytic enzymes, Thrombolysis, Plasminogen activation, Protein engineering, Translational pharmacology, Cardiovascular diseases
Cite this paper: Khushnut Vokhidov, Akhmadjan Makhsumkhanov, Askar Kholikov, Barno Alimova, Ozodakhon Pulatova, Microbial Fibrinolytic Enzymes as Promising Thrombolytic Agents: Molecular Mechanisms, Functional Diversity and Therapeutic Perspectives, International Journal of Virology and Molecular Biology, Vol. 15 No. 2, 2026, pp. 34-42. doi: 10.5923/j.ijvmb.20261502.03.
Article Outline
1. Introduction
- Thrombotic cardiovascular and cerebrovascular diseases remain among the leading causes of mortality and long-term disability worldwide. Acute thrombotic events, including ischemic stroke, myocardial infarction, venous thromboembolism, and pulmonary embolism, share a common pathological basis— the formation of fibrin-rich intravascular clots that obstruct blood flow and precipitate ischemic tissue injury [23,28]. Consequently, rapid and effective thrombolysis remains a cornerstone of acute management aimed at restoring perfusion and limiting irreversible damage.Pharmacological thrombolysis using plasminogen activators has substantially improved clinical outcomes in selected patients. Tissue-type plasminogen activator (tPA) and its derivatives are widely used in acute ischemic stroke and other thrombotic emergencies; however, their clinical utility is constrained by a narrow therapeutic window, high risk of hemorrhagic complications, and strict patient selection criteria [1,11]. In parallel, bacterial-derived thrombolytics such as streptokinase remain in use in many regions due to cost-effectiveness and accessibility, yet their lack of fibrin specificity and pronounced immunogenicity significantly limit safety and repeated administration [3,16].These limitations highlight a fundamental and unresolved problem in thrombolytic therapy: the long-standing trade-off between clot-dissolving efficacy and systemic safety. Despite decades of clinical experience and incremental improvements, currently available thrombolytics continue to induce non-selective fibrinolysis, exposing patients to bleeding risk and restricting broader clinical applicability. Notably, most therapeutic advances have focused on modifying existing plasminogen activators rather than exploring fundamentally different mechanistic paradigms.In this context, microbial fibrinolytic enzymes have gained increasing attention as alternative thrombolytic candidates. Diverse bacteria, fungi, and microalgae produce enzymes capable of dissolving fibrin either directly, by proteolytic degradation of the fibrin network, or indirectly, by activating endogenous plasminogen pathways [12,16]. Importantly, several microbial enzymes demonstrate enhanced fibrin selectivity, reduced systemic fibrinolysis, and compatibility with scalable microbial production, suggesting potential advantages over conventional thrombolytics.However, despite extensive experimental characterization, the clinical translation of microbial fibrinolytic enzymes remains limited. Previous reviews have predominantly catalogued enzyme sources and biochemical properties, with insufficient integration of mechanistic classification, translational barriers, and bioengineering strategies required for clinical development. Moreover, critical issues such as immunogenicity, pharmacokinetics, delivery, and regulatory feasibility are often discussed in isolation rather than as interconnected determinants of therapeutic success.Therefore, this review aims to address these gaps by providing a critical and integrative analysis of microbial thrombolytic and fibrinolytic enzymes. Specifically, we (i) classify microbial enzymes based on their mechanisms of action into direct fibrinolytic, indirect plasminogen-activating, and mixed-mechanism categories; (ii) examine microbial sources and biochemical diversity in relation to therapeutic relevance; (iii) evaluate advances in recombinant production and protein engineering; and (iv) critically discuss translational challenges and future directions. By linking microbial biodiversity with modern bioengineering and translational pharmacology, this review seeks to identify realistic pathways toward the development of safer and more effective next-generation thrombolytic therapies.
2. Literature Search Strategy and Selection Criteria
- To ensure a comprehensive, transparent, and reproducible synthesis of existing knowledge, a structured literature search strategy was employed following best practices for narrative and critical review articles in biomedical sciences [2,12]. The literature search was conducted using multiple internationally recognized databases, including PubMed, Web of Science Core Collection, Scopus, ScienceDirect, and Google Scholar, which collectively provide broad coverage of pharmacological, microbiological, enzymological, and translational research.Publications from January 2000 to March 2025 were considered, corresponding to the period of rapid advancement in fibrinolytic enzyme discovery, recombinant protein technology, and thrombolytic drug development [12,16]. The search strategy employed combinations of keywords and Boolean operators such as “microbial fibrinolytic enzymes”, “thrombolytic proteases”, “nattokinase”, “streptokinase”, “staphylokinase”, “plasminogen activator”, “direct fibrinolysis”, “bifunctional thrombolytic enzyme”, “recombinant thrombolytics”, and “protein engineering”, as recommended in prior thrombolytic-focused reviews [12,23]. Where applicable, Medical Subject Headings (MeSH) terms were used to refine PubMed searches.Only peer-reviewed articles published in English were included to maintain scientific rigor and consistency. Eligible publications comprised original research articles, systematic and narrative reviews, and translational studies reporting experimental evidence on microbial fibrinolytic or thrombolytic activity. Studies lacking experimental validation, focusing exclusively on non-microbial proteases, or addressing coagulation without relevance to fibrinolysis were excluded, in line with established review methodologies [2,28].The selection process followed a two-stage screening approach. In the initial stage, titles and abstracts were screened to remove duplicates and clearly irrelevant records. Subsequently, full-text articles were assessed for eligibility based on predefined inclusion criteria: (i) confirmed microbial origin of the enzyme, (ii) demonstrated fibrinolytic or thrombolytic activity through in vitro, ex vivo, or in vivo assays, and (iii) relevance to therapeutic or translational applications [9,16]. Particular emphasis was placed on studies reporting enzyme purification and biochemical characterization, mechanistic insights into fibrin or plasminogen interactions, and comparative analyses with conventional thrombolytics [8,22].To enhance translational relevance, additional priority was given to publications addressing safety considerations, immunogenicity, pharmacokinetics, and delivery challenges, which are recognized as critical bottlenecks in thrombolytic drug development [1,12]. Studies describing recombinant expression systems, codon optimization, and protein engineering strategies were preferentially included due to their importance in clinical-scale production and molecular optimization [21,24].Data extracted from eligible studies included microbial source, enzyme classification (direct fibrinolytic, indirect plasminogen activator, or mixed-mechanism enzyme), molecular weight, catalytic properties, fibrin specificity, production and purification methods, recombinant expression host, and reported therapeutic outcomes. The selected literature was then synthesized and organized according to mechanistic categories to enable structured comparison and critical discussion, consistent with contemporary approaches to thrombolytic enzyme reviews [12,16].
3. Classification of Microbial Thrombolytic Enzymes
- Microbial thrombolytic enzymes constitute a structurally and mechanistically diverse group of biocatalysts capable of dissolving fibrin-rich thrombi through distinct biochemical pathways. Based on their primary mode of action within the fibrinolytic system, these enzymes are conventionally classified into three major categories: (i) direct fibrinolytic enzymes, (ii) indirect plasminogen activators, and (iii) enzymes exhibiting mixed or bifunctional mechanisms of action [12,18]. This mechanistic classification provides a functional framework for evaluating their therapeutic potential, safety profile, and translational applicability.
3.1. Direct Fibrinolytic Enzymes
- Direct fibrinolytic enzymes are proteases that hydrolyze fibrin independently of plasminogen activation. These enzymes act directly on the fibrin network constituting the structural backbone of thrombi, thereby minimizing systemic activation of the fibrinolytic cascade. As a result, direct fibrinolytic proteases are generally associated with enhanced fibrin specificity and a reduced risk of hemorrhagic complications compared with classical plasminogen activators [16,23].
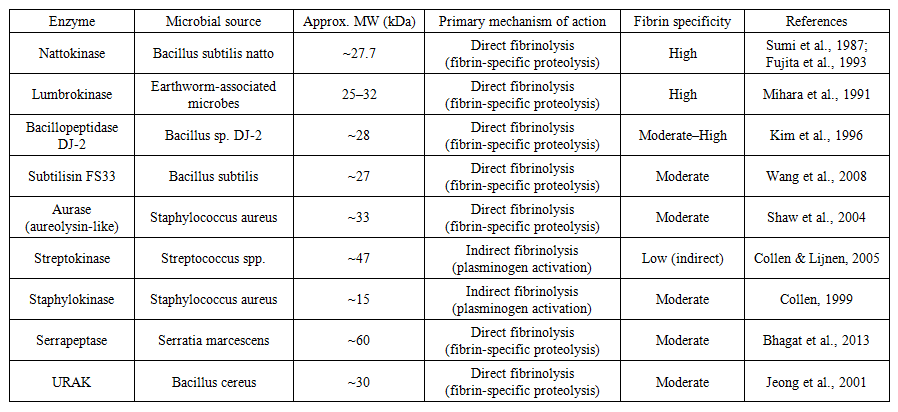 | Table 1. Microbial producers of thrombolytic and fibrinolytic enzymes and their key characteristics |
3.2. Indirect Plasminogen Activators
- Indirect plasminogen activators constitute a second major class of microbial thrombolytic enzymes. Unlike direct proteases, these agents do not possess intrinsic fibrinolytic activity but instead promote the conversion of plasminogen into active plasmin, which subsequently degrades fibrin. The prototypical example of this class is streptokinase, a bacterial protein produced by Streptococcus species [11,16].Streptokinase forms a stoichiometric complex with plasminogen, inducing a conformational change that exposes the active site of plasminogen and enables its proteolytic activity. This indirect mechanism results in potent thrombolysis and has led to extensive clinical use of streptokinase in the treatment of acute myocardial infarction and other thrombotic disorders, particularly in resource-limited settings [3,11]. However, because streptokinase activates circulating plasminogen systemically, it is associated with widespread fibrinolysis and an elevated risk of bleeding complications [1].Another clinically relevant indirect activator is staphylokinase, produced by Staphylococcus aureus. Staphylokinase exhibits greater fibrin specificity than streptokinase due to preferential activation of plasminogen bound to fibrin, which partially mitigates systemic effects [9]. Nevertheless, both streptokinase and staphylokinase are immunogenic bacterial proteins, and pre-existing or treatment-induced antibodies can significantly reduce their efficacy and preclude repeated administration [4,9].Despite these limitations, indirect plasminogen activators remain of high scientific and translational interest. Advances in recombinant DNA technology, protein engineering, and immunoinformatics-guided epitope modification have enabled the development of engineered variants with reduced immunogenicity and improved pharmacological profiles [13,20]. Thus, indirect activators continue to serve both as therapeutic agents and as valuable templates for next-generation thrombolytic design.
3.3. Mixed-Mechanism (Bifunctional) Thrombolytic Enzymes
- A third and increasingly important category comprises microbial enzymes that exhibit mixed or bifunctional mechanisms of action. These enzymes combine moderate direct fibrin degradation with partial activation of plasminogen, thereby integrating features of both direct proteases and indirect activators. This dual functionality is hypothesized to enhance clot dissolution while limiting excessive systemic fibrinolysis [9,12].Nattokinase is often cited as a representative example of a mixed-mechanism enzyme. In addition to its direct fibrinolytic activity, nattokinase has been reported to modulate endogenous fibrinolytic regulators, including the enhancement of tissue-type plasminogen activator (tPA) release and the inactivation of plasminogen activator inhibitor-1 (PAI-1), thereby indirectly promoting plasmin generation [26]. This multifaceted mode of action may contribute to its reported antithrombotic efficacy and relatively favorable safety profile.Fungal and marine-derived metalloproteases also represent promising candidates within this category. Certain zinc-dependent metalloproteases isolated from fungi and marine microorganisms demonstrate the ability to hydrolyze fibrin while simultaneously facilitating plasminogen activation, although the precise molecular determinants of this dual activity remain under investigation [12,26]. Importantly, these bifunctional enzymes challenge the traditional binary classification of thrombolytics and suggest that a continuum of fibrinolytic mechanisms exists.From a therapeutic perspective, mixed-mechanism enzymes are particularly attractive because they may achieve an optimal balance between efficacy and safety. By combining localized fibrin degradation with controlled amplification of endogenous fibrinolysis, these enzymes have the potential to overcome the major clinical limitations associated with conventional thrombolytics. However, rigorous mechanistic studies, in vivo validation, and pharmacokinetic profiling are required to substantiate these advantages.
4. Microbial Sources and Biochemical Diversity
- Microbial fibrinolytic enzymes originate from a wide spectrum of microorganisms inhabiting diverse ecological niches, including terrestrial soils, fermented foods, marine environments, and extreme ecosystems. This ecological diversity is directly reflected in the remarkable biochemical heterogeneity of the enzymes produced, encompassing differences in molecular weight, catalytic mechanisms, substrate specificity, stability profiles, and regulatory properties [12,16].Microbial thrombolytic enzymes can be classified into direct fibrinolytic enzymes, indirect plasminogen activators, and mixed-mechanism enzymes based on their primary mode of action within the fibrinolytic system. While this mechanistic classification provides a useful conceptual framework, its translational relevance becomes evident only when the relative advantages and limitations of each enzyme class are critically compared in the context of therapeutic efficacy, safety, and clinical applicability.
4.1. Bacterial Sources
- Bacteria represent the most extensively studied and prolific producers of fibrinolytic and thrombolytic enzymes. Among them, members of the genus Bacillus dominate the literature due to their ability to secrete high levels of extracellular proteases with strong fibrinolytic activity. These enzymes are typically subtilisin-like serine proteases, characterized by robust catalytic efficiency, thermostability, and activity over a broad pH range [8,17].Fermented foods have proven to be particularly rich reservoirs of fibrinolytic Bacillus strains. Nattokinase, isolated from Bacillus subtilis var. natto, remains the most widely investigated example and exhibits both direct fibrin degradation and indirect modulation of endogenous fibrinolytic regulators [22]. Similar fibrinolytic enzymes have been identified in strains derived from other fermented substrates, including soybean- and seafood-based products, underscoring the role of traditional fermentation practices in shaping enzyme diversity [19].Beyond food-associated strains, soil-derived Bacillus species produce fibrinolytic enzymes with distinct biochemical traits. For example, enzymes from Bacillus cereus and Bacillus pseudomycoides demonstrate high fibrin specificity and compatibility with scalable biotechnological production, making them attractive for pharmaceutical development [5,8]. These bacterial enzymes commonly exhibit molecular weights ranging from 25 to 40 kDa and retain activity under moderately alkaline conditions, which may be advantageous for stability during formulation and storage.Marine bacteria constitute another important bacterial source of fibrinolytic enzymes. Proteases isolated from marine Bacillus spp., including strains recovered from the Black Sea and South China Sea, show enhanced tolerance to salinity and alkaline pH, as well as elevated thrombolytic activity [18,25]. Such adaptations are likely a consequence of selective pressures in marine environments and may confer superior stability under physiological and industrial conditions.
4.2. Fungal and Microalgal Sources
- In addition to bacteria, fungi and microalgae have emerged as alternative sources of fibrinolytic enzymes with distinct biochemical properties. Fungal fibrinolytic enzymes are often metalloproteases or serine proteases that differ structurally and mechanistically from bacterial subtilisins. Certain filamentous fungi produce enzymes capable of degrading fibrin and fibrinogen while also exhibiting anticoagulant or antiplatelet effects, suggesting multifunctional therapeutic potential [12,26].Microalgae represent a comparatively underexplored but promising source of thrombolytic enzymes. A notable example is the fibrinolytic enzyme isolated from Chlorella vulgaris, which displays direct clot-lysing activity with minimal hemolytic effects [7]. The unique metabolic pathways and cellular organization of microalgae may give rise to enzymes with novel structures and modes of action, expanding the biochemical repertoire available for thrombolytic drug discovery.
4.3. Biochemical Diversity and Structure–Function Relationships
- The biochemical diversity of microbial fibrinolytic enzymes is reflected in variations in catalytic residues, domain architecture, metal-ion dependence, and substrate recognition sites. Serine proteases typically rely on the classical catalytic triad (Ser–His–Asp) and preferentially cleave fibrin α- and β-chains, whereas metalloproteases require divalent metal ions such as Zn²⁺ for activity and may exhibit broader substrate specificity [17,26].Environmental origin strongly influences enzyme properties. Enzymes derived from extreme environments—such as high-salinity marine habitats—often display enhanced thermostability, alkaline tolerance, and resistance to denaturation, traits that are advantageous for pharmaceutical formulation and industrial-scale production [18,25]. Conversely, enzymes from food-associated microbes may be better suited for oral or nutraceutical applications due to their stability under gastrointestinal conditions [22,26].
4.4. Implications for Therapeutic Development
- A comparative evaluation reveals that no single class of microbial thrombolytic enzymes is universally superior, as each exhibits a distinct balance between fibrinolytic potency, systemic safety, and translational feasibility.Direct fibrinolytic enzymes, predominantly subtilisin-like proteases from Bacillus species, demonstrate strong and localized fibrin-degrading activity with limited reliance on plasminogen activation. This property confers a theoretical safety advantage by reducing systemic fibrinolysis and hemorrhagic risk [12]. However, their proteolytic nature raises concerns regarding off-target substrate degradation, and their pharmacokinetic profiles remain insufficiently characterized for clinical translation.In contrast, indirect plasminogen activators, such as streptokinase and staphylokinase, exhibit robust thrombolytic efficacy supported by extensive clinical experience. Their principal limitation lies in non-selective plasminogen activation, which frequently leads to systemic fibrinolysis and bleeding complications [1,3]. Furthermore, pronounced immunogenicity significantly restricts repeated administration, limiting long-term or recurrent therapeutic use.Mixed-mechanism enzymes represent an intermediate and increasingly attractive category. By combining moderate direct fibrin degradation with controlled plasminogen activation or modulation of endogenous fibrinolytic regulators, these enzymes may partially overcome the classical efficacy–safety trade-off [9,12]. Although experimental evidence supporting this balanced mechanism is accumulating, most mixed-mechanism enzymes remain at a preclinical stage, and their translational advantages require validation in vivo.From a clinical perspective, direct fibrinolytic enzymes may be better suited for localized or adjunctive thrombolysis, whereas indirect activators retain relevance in acute, high-burden thrombotic events where rapid clot dissolution is critical. Mixed-mechanism enzymes, particularly those amenable to protein engineering and targeted delivery, appear to offer the greatest long-term potential for next-generation thrombolytic therapy.Overall, this comparative analysis underscores that mechanistic classification alone is insufficient for therapeutic prioritization. Instead, translational suitability emerges from the interplay between enzyme mechanism, safety profile, manufacturability, and capacity for molecular optimization, highlighting the need for integrative evaluation strategies in future thrombolytic research.
5. Recombinant Production and Protein Engineering (Final, with References)
- Recombinant DNA technology and protein engineering have become indispensable tools for advancing microbial fibrinolytic enzymes toward therapeutic application. Native microbial enzymes often exhibit low production yields, batch-to-batch variability, and unfavorable pharmacological properties, which limit their clinical utility. Recombinant expression systems and rational protein engineering strategies have therefore been widely applied to improve yield, stability, and catalytic performance [2,21].
5.1. Achievements of Recombinant Production
- Heterologous expression of microbial thrombolytic enzymes in Escherichia coli, Bacillus subtilis, and yeast systems has enabled high-level production and facilitated molecular and functional characterization. Streptokinase represents the most established example of a microbial thrombolytic produced recombinantly, with long-standing clinical use demonstrating the feasibility of large-scale microbial manufacturing [3,4].Similarly, recombinant production of subtilisin-like fibrinolytic enzymes from Bacillus species has yielded catalytically active proteins with improved purity and reproducibility compared with native isolates [17,19]. Codon optimization and process optimization have further enhanced expression efficiency in bacterial hosts, particularly for streptokinase and nattokinase variants [10,19].Protein engineering approaches, including site-directed mutagenesis and domain optimization, have been successfully applied to improve enzyme stability, thermal tolerance, and resistance to proteolytic degradation [20,25]. Collectively, these advances provide a robust technological platform for the production of engineered microbial thrombolytics.
5.2. The Translational Gap: Limitations Beyond Engineering Success
- Despite substantial progress in recombinant production and molecular optimization, the majority of engineered microbial fibrinolytic enzymes have not progressed beyond the preclinical stage, highlighting a critical translational gap [12,16].One major limitation is the lack of favorable pharmacokinetic properties in vivo. Many engineered enzymes demonstrate rapid plasma clearance and short half-lives, which significantly reduce therapeutic exposure and necessitate high or repeated dosing [28]. Unlike endogenous fibrinolytic proteins, microbial enzymes generally lack intrinsic regulatory domains or targeting mechanisms that confine activity to thrombus sites, increasing the risk of systemic effects.Immunogenicity remains another unresolved barrier, particularly for enzymes of bacterial origin. Although immunoinformatics-guided epitope modification and rational redesign have shown promise in reducing predicted antigenicity, convincing in vivo and clinical evidence demonstrating sustained efficacy after repeated administration is still limited [4,12]. Regulatory agencies therefore require extensive immunogenicity and safety evaluation, which has slowed clinical progression.In addition, regulatory considerations favor incremental innovation based on well-characterized thrombolytics, such as tPA derivatives, over mechanistically novel microbial enzymes. As a result, direct and mixed-mechanism microbial fibrinolytic enzymes face higher evidentiary thresholds, including comprehensive toxicological profiling and long-term safety assessment [1,12].
5.3. Aligning Protein Engineering with Clinical Translation
- To bridge the gap between recombinant engineering and clinical application, future strategies must prioritize clinically relevant design endpoints rather than enzymatic activity alone. These include extending plasma half-life, minimizing immunogenicity, enhancing fibrin specificity, and ensuring predictable pharmacokinetics [9,28].Emerging approaches such as fusion to carrier domains, structure-guided redesign of substrate-binding regions, and integration with targeted delivery systems may offer more realistic translational pathways than activity-focused mutagenesis alone [12,20]. Importantly, aligning recombinant design strategies with pharmacological and regulatory requirements from early development stages is essential for successful clinical translation.
6. Translational Challenges and Limitations
- Despite the substantial experimental progress achieved in the identification and characterization of microbial fibrinolytic enzymes, their translation from laboratory research to clinically approved thrombolytic agents remains challenging. Multiple biological, pharmacological, technological, and regulatory barriers must be addressed to ensure that these enzymes achieve acceptable safety, efficacy, and reproducibility profiles in human patients [2,12].
6.1. Immunogenicity and Host Immune Responses
- One of the most critical limitations of microbial-derived thrombolytic enzymes is their potential immunogenicity. Proteins of bacterial or fungal origin may elicit humoral immune responses, leading to the formation of neutralizing antibodies that reduce therapeutic efficacy and preclude repeated administration. Streptokinase is a well-documented example, as pre-existing anti-streptokinase antibodies are prevalent in human populations due to prior streptococcal exposure [3,4]. It is important to emphasize that immunogenicity is not exclusive to indirect plasminogen activators. Any protein of microbial origin—whether a direct fibrinolytic protease or an indirect activator—can elicit immune responses depending on sequence homology, structural motifs, and exposure frequency. While streptokinase represents a well-known example, direct fibrinolytic enzymes are not inherently non-immunogenic. The degree of immunogenicity is better viewed as a continuum influenced by molecular design and host factors rather than by mechanistic class alone.Even for direct fibrinolytic enzymes with lower systemic activity, immunogenicity remains a concern, particularly during chronic or prophylactic use. Immune-mediated adverse reactions, including allergic responses and hypotension, represent significant safety risks that must be carefully evaluated during preclinical development [1]. Although immuneinformatics-guided epitope modification has shown promise in reducing immunogenic potential, these strategies require extensive validation in relevant in vivo models [12].
6.2. Pharmacokinetics and Biodistribution
- Incomplete pharmacokinetic characterization constitutes another major translational bottleneck. Many microbial fibrinolytic enzymes have been evaluated primarily in vitro or in ex vivo clot-lysis models, with limited data available on absorption, distribution, metabolism, and elimination in vivo. Rapid clearance from circulation and short plasma half-life can significantly limit therapeutic effectiveness, necessitating high or repeated dosing [12,28].Furthermore, achieving sufficient enzyme concentration at the thrombus site without inducing systemic fibrinolysis remains a complex challenge. Unlike endogenous fibrinolytic proteins, microbial enzymes often lack natural targeting mechanisms, which may result in off-target activity and increased bleeding risk [23]. Advanced delivery strategies, including nanoparticle encapsulation and fibrin-targeted carriers, have been proposed but remain largely experimental.
6.3. Safety and Off-Target Proteolysis
- The proteolytic nature of fibrinolytic enzymes raises inherent safety concerns related to off-target substrate degradation. Broad-spectrum protease activity may affect non-fibrin substrates, potentially disrupting extracellular matrix components, plasma proteins, or coagulation regulators [12,26]. Such off-target effects can exacerbate bleeding risk and complicate dose optimization.Direct fibrinolytic enzymes, while generally associated with reduced systemic fibrinolysis, are not exempt from these risks. Careful biochemical profiling, including substrate specificity assays and proteomic analyses, is required to identify and mitigate unintended proteolytic interactions prior to clinical translation [12].
6.4. Formulation, Stability, and Route of Administration
- Formulation challenges represent another significant obstacle in the development of enzyme-based thrombolytics. Proteins are inherently sensitive to denaturation, aggregation, and proteolytic degradation during storage and administration. Maintaining enzyme stability under physiological conditions and during long-term storage requires optimized formulation strategies, including stabilizing excipients and controlled storage environments [24].The route of administration further complicates translational development. While intravenous delivery remains the standard for acute thrombolysis, interest in oral or minimally invasive administration has increased, particularly for enzymes such as natto kinase. However, proteolytic degradation in the gastrointestinal tract and limited bioavailability poses substantial barriers to oral delivery of active enzymes [22,26,27].Oral delivery of fibrinolytic enzymes remains one of the most debated and scientifically challenging aspects of enzyme-based thrombolytic therapy. Following oral intake, proteins are exposed to gastric acidity, intestinal proteases, and harsh physicochemical conditions that markedly reduce structural integrity and enzymatic activity. Even if partial survival occurs, trans-epithelial absorption of intact proteases is highly inefficient. Furthermore, enzymes entering portal circulation undergo hepatic first-pass metabolism, which may substantially lower bioactive concentrations before reaching systemic thrombi. Therefore, claims regarding oral thrombolytic efficacy should be interpreted cautiously unless supported by pharmacokinetic and bioavailability data. Encapsulation, enteric coatings, and carrier-mediated delivery systems may partially mitigate these barriers, but their clinical effectiveness remains to be convincingly demonstrated.
6.5. Regulatory and Manufacturing Challenges
- From a regulatory perspective, microbial thrombolytic enzymes must meet stringent requirements for biologic therapeutics, including batch-to-batch consistency, purity, potency, and validated safety profiles. Variability inherent to biological production systems can complicate compliance with regulatory standards set by agencies such as the FDA and EMA [2,12].Manufacturing scalability and cost-effectiveness are additional considerations, particularly for low- and middle-income settings where thrombolytic therapy is most urgently needed. Although microbial fermentation offers advantages in scalability, downstream purification and quality control remain technically demanding and cost-intensive [21].
6.6. Limitations of Current Preclinical Models
- Finally, the predictive value of existing preclinical models remains limited. Many animal thrombosis models fail to fully recapitulate the complexity of human thrombotic disease, including comorbidities, clot composition, and vascular heterogeneity. Consequently, promising preclinical results may not always translate into clinical success [28].To overcome these limitations, the adoption of more physiologically relevant models, including advanced imaging-guided thrombosis models and humanized systems, is increasingly advocated. Such approaches may improve translational predictability and facilitate the rational selection of candidate enzymes for clinical development.
7. Future Perspectives
- The future development of microbial fibrinolytic enzymes as clinically viable thrombolytic agents lies at the intersection of molecular engineering, systems biology, and translational pharmacology. While early-generation thrombolytics primarily focused on maximizing clot lysis, next-generation strategies increasingly emphasize precision, safety, and targeted efficacy, reflecting a paradigm shift in thrombolytic drug design [12,28].
7.1. Engineering Next-Generation Thrombolytics
- Advances in protein engineering are expected to play a central role in optimizing microbial fibrinolytic enzymes. Structure-guided mutagenesis, domain swapping, and rational redesign of substrate recognition sites offer powerful tools to enhance fibrin specificity while minimizing off-target proteolysis [9,20]. In particular, the development of hybrid enzymes that combine direct fibrin degradation with regulated plasminogen activation represents a promising strategy to balance thrombolytic efficacy and hemorrhagic risk.Emerging computational approaches, including machine learning–assisted protein modeling and in silico screening, are likely to accelerate the identification of optimized enzyme variants with tailored pharmacokinetic and pharmacodynamic profiles. Such data-driven strategies may significantly reduce experimental trial-and-error and shorten development timelines [12].
7.2. Targeted Delivery and Formulation Innovations
- Targeted delivery systems constitute another critical frontier in thrombolytic therapy. Nanoparticle-based carriers, fibrin-targeting peptides, and antibody-guided delivery platforms have the potential to localize thrombolytic activity at the clot site, thereby enhancing efficacy while reducing systemic exposure [23,28]. For microbial enzymes, encapsulation within biodegradable nanocarriers may also protect against premature degradation and immunogenic recognition.In parallel, formulation science will be essential for improving enzyme stability, shelf-life, and administration routes. Advances in protein stabilization, lyophilization, and controlled-release systems may enable broader clinical use, including potential prophylactic or adjunctive applications [24].
7.3. Translational and Clinical Research Directions
- From a translational standpoint, future research must prioritize robust in vivo validation using clinically relevant thrombosis models that better reflect human disease complexity. Integration of imaging-guided assessment, biomarker-based monitoring, and comparative studies against existing thrombolytics will be critical for establishing clinical value [28].Moreover, early engagement with regulatory frameworks is likely to streamline clinical translation. Incorporating regulatory science considerations—such as safety margins, immunogenicity profiling, and manufacturing reproducibility—at early development stages can reduce attrition in later-phase trials [2,12].
7.4. Expanding the Microbial Resource Space
- The continued exploration of underrepresented microbial niches, including extreme environments and uncultured microorganisms, holds significant promise for uncovering novel fibrinolytic enzymes with unique biochemical properties. Advances in metagenomics and synthetic biology now enable access to microbial genetic diversity previously inaccessible through conventional cultivation methods [16].Coupled with recombinant expression and modular protein design, these approaches may yield entirely new classes of thrombolytic enzymes optimized for specific clinical contexts, ranging from acute ischemic stroke to chronic thrombotic disorders.
8. Conclusions
- Microbial fibrinolytic enzymes constitute a heterogeneous but increasingly well-defined group of biocatalysts that offer mechanistic alternatives to conventional plasminogen activators. Accumulated evidence indicates that enzymes derived from Bacillus spp., selected fungi, and microalgae are capable of degrading fibrin through distinct direct, indirect, or mixed mechanisms, with several candidates demonstrating superior fibrin selectivity and reduced systemic fibrinolytic activation compared with clinically used thrombolytics.Among the identified enzyme classes, direct fibrinolytic proteases, particularly subtilisin-like enzymes from Bacillus species, emerge as the most promising candidates for reducing hemorrhagic risk due to their localized fibrin-degrading activity. In parallel, mixed-mechanism enzymes, which combine moderate direct fibrinolysis with controlled modulation of endogenous plasminogen pathways, represent a particularly attractive translational strategy, as they directly address the long-standing efficacy–safety trade-off that limits current thrombolytic therapy. In contrast, indirect plasminogen activators remain clinically effective but are unlikely to overcome immunogenicity and systemic fibrinolysis without substantial molecular redesign.Despite clear biochemical and mechanistic advantages, the clinical translation of microbial fibrinolytic enzymes remains constrained by a limited number of prioritized bottlenecks, most notably immunogenicity, short plasma half-life, insufficient thrombus targeting, and regulatory barriers associated with novel biologics. Importantly, advances in recombinant production and protein engineering have demonstrated that enzymatic activity alone is insufficient for clinical success; future development must explicitly integrate pharmacokinetics, safety profiling, and regulatory feasibility into early-stage enzyme design.Taken together, microbial fibrinolytic enzymes should be regarded not as direct replacements for existing thrombolytics, but as a flexible development platform for next-generation, fibrin-targeted thrombolytic agents. Strategic prioritization of mixed-mechanism enzymes, coupled with rational protein engineering and targeted delivery approaches, is most likely to yield clinically translatable candidates capable of addressing unmet needs in the treatment of thrombotic cardiovascular and cerebrovascular diseases.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML