-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Virology and Molecular Biology
p-ISSN: 2163-2219 e-ISSN: 2163-2227
2026; 15(2): 21-26
doi:10.5923/j.ijvmb.20261502.01
Received: Feb. 9, 2026; Accepted: Feb. 22, 2026; Published: Mar. 7, 2026

Assessment of the Potential Distribution of the Rare Species Astragalus Knorringianus in the Nurata Nature Reserve Based on a Bioclimatic Model
Shakhnoza Doniyorova1, Natalya Beshko2, Husniddin Abulfayzov2
1Jizzakh State Pedagogical University, Sharof Rashidov street, Jizzakh, Uzbekistan
2Institute of Botany Academy of Sciences Republic of Uzbekistan, Tashkent, Uzbekistan
Correspondence to: Shakhnoza Doniyorova, Jizzakh State Pedagogical University, Sharof Rashidov street, Jizzakh, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Based on the BIOCLIM model using 17 ecological factors, the potential distribution area of the rare endemic of the Northwest Pamir-Alai, Astragalus knorringianus Boriss., was assessed within the Nurata State Nature Reserve. It was found that the most significant ecological factors influencing the species’ distribution are the mean temperature of the wettest quarter, daily and annual temperature variability, slope aspect, solar radiation, and the minimum vegetation index. The results of the bioclimatic modeling showed that the most favorable habitats for the species are located on the southern dry slopes of the Nurata Ridge, with a total area of 35.78 km² (9.4% of the total area of the Nurata Reserve and its buffer zone).
Keywords: Range, Bioclimatic modeling, Nature reserve, Climate change, Red Book, Rare species, Ecological niche, Ecological factor, Endemic, Uzbekistan
Cite this paper: Shakhnoza Doniyorova, Natalya Beshko, Husniddin Abulfayzov, Assessment of the Potential Distribution of the Rare Species Astragalus Knorringianus in the Nurata Nature Reserve Based on a Bioclimatic Model, International Journal of Virology and Molecular Biology, Vol. 15 No. 2, 2026, pp. 21-26. doi: 10.5923/j.ijvmb.20261502.01.
1. Introduction
- Plants are of key importance to life on Earth and are integral components of ecosystems. At the same time, anthropogenic pressure on the plant world continues to intensify globally, resulting in the process of biodiversity loss reaching catastrophic proportions. Another global factor threatening biodiversity is climate change. Currently, due to the cumulative impact of anthropogenic and climatic factors, many endemic and stenotopic species have become endangered [1,2,3]. In this regard, targeted research into the ecology and biology of narrowly distributed endemics and the phenomenon of endemism as a whole represents a pressing scientific issue [4,5,6]. One of the most modern methods is the identification of bioclimatic niches for rare and vulnerable species, the determination of ecological factors influencing the distribution of these species, and the assessment of potential species responses to climate change using computer modeling [7,8].The Nurata State Nature Reserve is located in the northwestern foothills of the Pamir-Alay mountain system, in the central part of the Nurata Range. The current area of the reserve is 22.18 km², and together with the buffer zone, the total area under study is 380.83 km². The central part of the Nurata Range, including the territory of the reserve and its buffer zone, is an internationally recognized key biodiversity area within the Central Asia Mountains Global Biodiversity Hotspot [9,10]. This region is rich in rare and endemic species, with a flora endemism rate of 2.7% [11]. In this regard, the Nurata Reserve is of great importance for the conservation of biological diversity in the Republic of Uzbekistan, including many endemic, rare, and endangered plant species.In this study, based on the BIOCLIM model, the potential distribution range of Astragalus knorringianus Boriss. a rare endemic of the North-Western Pamir-Alai, listed in the Red Book of the Republic of Uzbekistan with status 2 [12] was analyzed within the territory of the Nurata State Nature Reserve. The environmental factors favorable for this species have been identified.
2. Research Methods
- As the subject of the study, one of the rare, narrowly endemic species of the legume family (Fabaceae) was chosen Astragalus knorringianus Boriss. a perennial, nearly stemless, gray-green, closely pubescent herbaceous plant, 5–10 cm tall, hemicryptophyte. The leaves are 2–5 cm long, imparipinnate. The leaflets are arranged in 7–9 pairs, linear or linear-lanceolate, 5–11 mm long and 0.5–1.5 mm wide. Flower stalks are 2–5 cm long. There are 2–3 light yellow flowers. The standard is 32–37 mm long, and the calyx is 17–20 mm long. The pods are linear, closely covered with white and black hairs, up to 7 cm long and 4 mm wide, with a short beak. It blooms from March to April and bears fruit from May to June. It reproduces by seeds. It is distributed in the northwestern spurs of the Pamir-Alai (the Nurata, Turkestan, and Malguzar ranges). It grows on finely earthy, rocky, and gravelly soils, in the foothills, the lower and middle mountain belts, at elevations ranging from 497 to 1,728 meters above sea level.In this study, for the first time, the method of bioclimatic modeling using the BIOCLIM software package was applied to determine the ecological niche and potential distribution range of Astragalus knorringianus within the Nurata Reserve. The BIOCLIM program approximately defines the bioclimatic contour and summarizes the climatic conditions in the specified areas [13]. The indicators obtained from the GEE platform were processed to achieve a spatial resolution of 1 km. In the study, based on the SRTM digital elevation model, a number of topographic indicators were obtained that characterize the features of the terrain (elevation above sea level, slope, aspect, as well as the topographic wetness index) [14]. The Terra Climate dataset covers a range of variables reflecting environmental and climate parameters, based on regular measurements of relevant data. Annual solar radiation, maximum solar radiation, and sunshine hours were selected as such variables. Since most satellite data with long time series are characterized by low spatial resolution, inconsistent accuracy, and lack of comparability, the CHIRPS dataset [15] was used in this study. Atmospheric parameters such as cloud cover, wind speed, and surface temperature were taken from ERA5-Land [16]. Soil parameters were downloaded from the SoilGrids database [17]. From the WorldClim v2.1 database, among 19 bioclimatic variables, bio_2, bio_3, bio_8, and bio_19 were selected [18]. To identify the main ecological factors shaping species habitats, a Principal Component Analysis (PCA) was conducted. This analysis makes it possible to reduce the correlation between numerous ecological variables and determine their principal directions of influence [19].Selection of environmental variables and the BIOCLIM modelIn this study, 41 environmental variables and the occurrence points of Astragalus knorringianus were introduced into the BIOCLIM model. In the correlation analysis, the relationships among the 41 variables were assessed using the Pearson coefficient. As a result of this assessment, 24 variables with high correlation (r ≥ |0.7|) were excluded, leaving 17 factors with low intercorrelation for modeling. The remaining factors (bio_2, bio_3, bio_8, bio_19, wet_season_Rad, SOC, sand, silt, clay, twi, Slp, Asp, Cloud, Wnd_speed, avg_wet_Prcp, min_NDVI, max_NDVI) provide independent ecological information for the accurate and reliable assessment of the species' ecological niche through bioclimatic modeling (Figure 1).
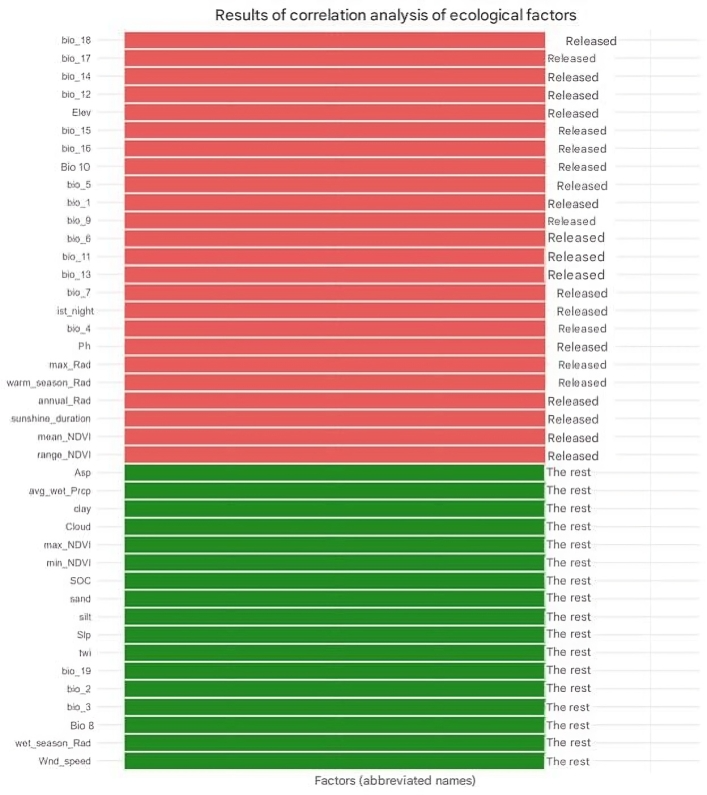 | Figure 1. Selected environmental variables for the BIOCLIM model |
3. Results
- Based on the results of the BIOCLIM model, the potential distribution range of Astragalus knorringianus was determined within the study area of the Nurata Reserve and its buffer zone (with a total area of 380.83 km²). According to the model analysis, the most favorable habitats for this plant species, which provide optimal ecological conditions, total 35.78 km² (9.40% of the study area). The area of unfavorable territories for the species, where the necessary ecological conditions are absent, amounts to 345.05 km² (90.60%) (Figure 2).
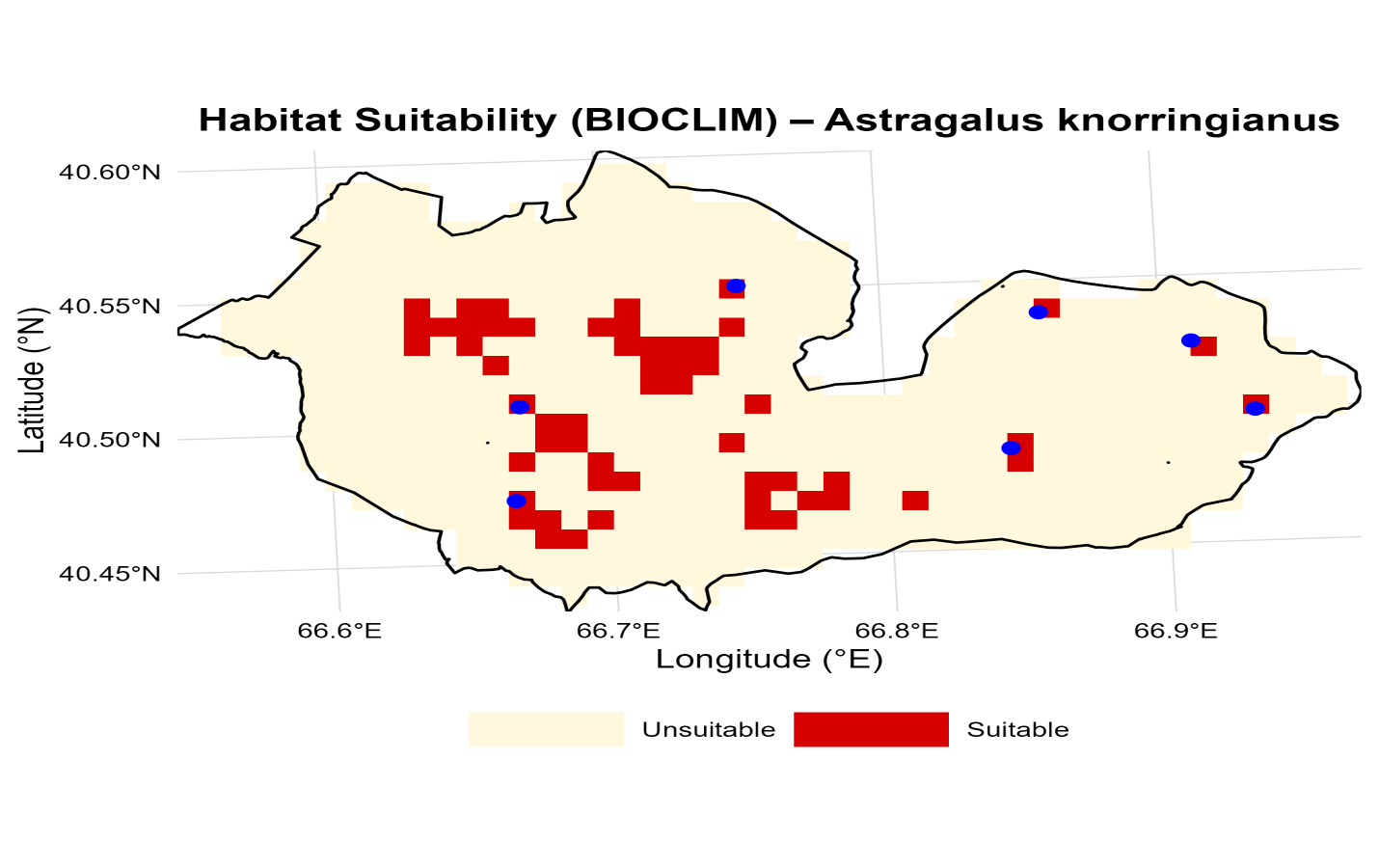 | Figure 2. Potential distribution of Astragalus knorringianus in the Nurata Reserve based on the BIOCLIM model |
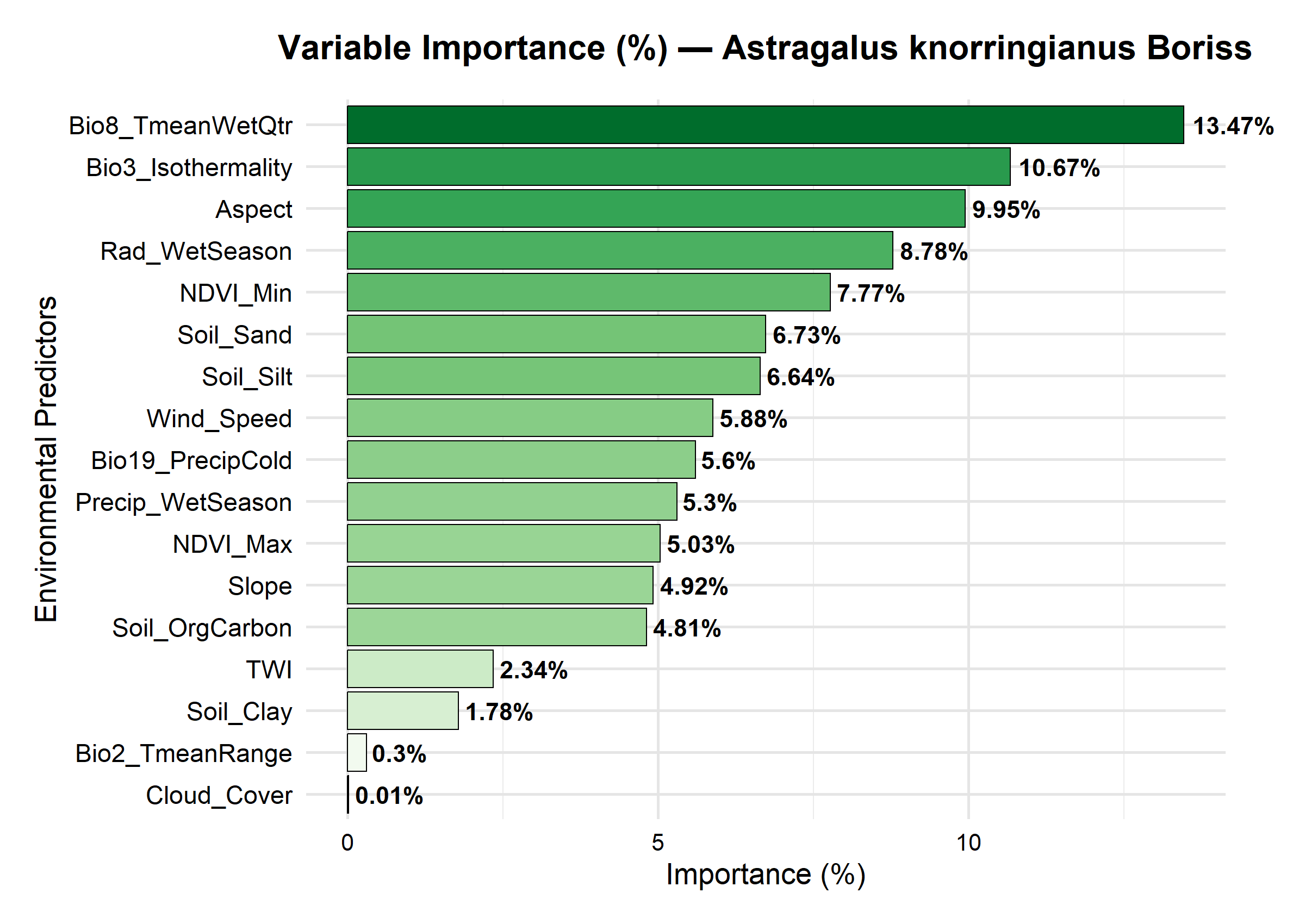 | Figure 3. The influence of environmental factors on the distribution of Astragalus knorringianus |
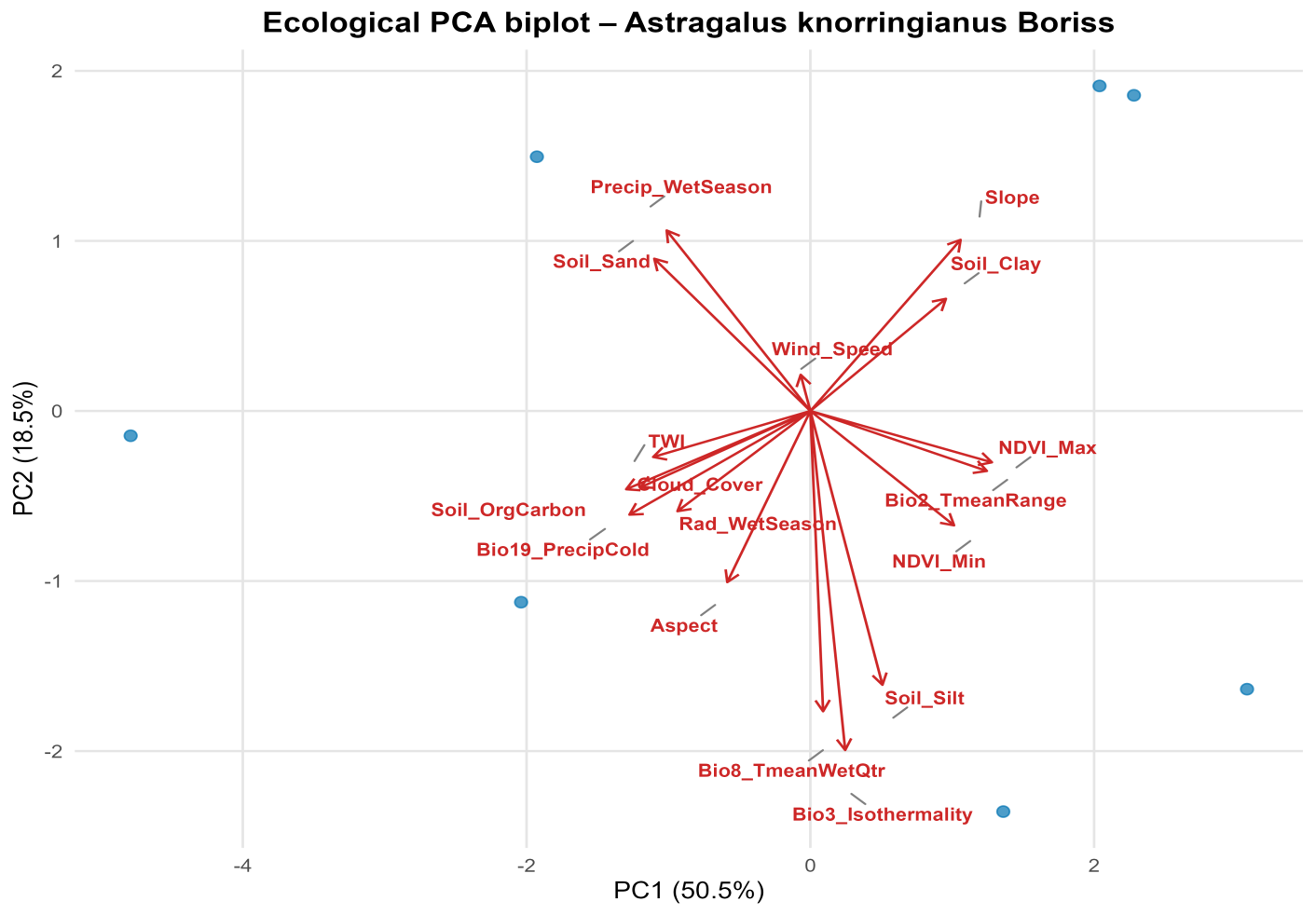 | Figure 4. PCA analysis of Astragalus knorringianus |
4. Conclusions
- The results of the study showed that the rare endemic species Astragalus knorringianus is a typical xerophyte, found within a limited ecological range. It was established that the main bioclimatic factors determining the species’ distribution are the average temperature of the wettest quarter, daily and annual temperature variability, terrain exposure, solar radiation, and the minimum vegetation index. The most favorable habitats for the species are located on open, dry southern slopes of the Nurata Ridge with light, well-drained soils. Because the species has a limited range and a narrow ecological niche, climate change and anthropogenic pressure can pose a significant threat to its populations. The ecological characteristics of the species must be taken into account when developing a targeted conservation strategy.
ACKNOWLEDGMENTS
- The research was conducted as part of the state program of the Institute of Botany of the Academy of Sciences of the Republic of Uzbekistan, “Digital Nature: Creation of a Digital Platform for the Flora of Central Uzbekistan” (2025–2029).
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML