-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Virology and Molecular Biology
p-ISSN: 2163-2219 e-ISSN: 2163-2227
2026; 15(1): 15-20
doi:10.5923/j.ijvmb.20261501.04
Received: Feb. 15, 2026; Accepted: Feb. 27, 2026; Published: Mar. 6, 2026

Inhibition of Pancreatic Lipase and α-amylase by Gingko Biloba Endophytes
Tashkhan Gulyamova1, Dilaram Ruziyeva1, Behruz Tojiyev1, Gulchekhra Rasulova1, Liliya Abdulmyanova1, Maftuna Yoldosheva2, Kseniya Kondrasheva1
1Institute of Microbiology, Academy of Sciences Republic of Uzbekistan, Tashkent, Uzbekistan
2University of Business and Science, Non-state Higher Education Institution, Tashkent, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

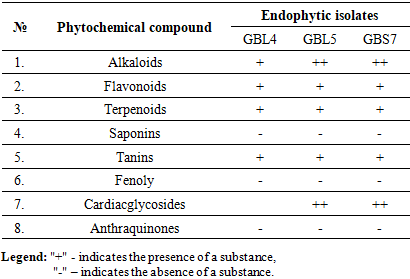
Pancreatic lipase and pancreatic α-amylase inhibitors are effective drugs for obesity and diabetes. Endophytes are a promising but little-studied source of inhibitors of therapeutically essential enzymes. The activity of α-amylase and lipase was determined spectrophotometrically. Studies of extracts of endophytic isolates GBL4, GBL5 from leaves, and GBS7 from the stem of Ginkgo biloba have shown their high inhibitory activity against pancreatic lipase and α-amylase. IC50 values of GBL4 and GBL5 isolates were 2.6 and 5.6 mcg/ml, respectively, while the IC50 of Xenical as a positive control was 20.0 mсg/ml, and the IC50 of GBS7 extract was close to Xenical (26.5 mcg/ml). IC50 values of amylase inhibition by GBL4 and GBL5 were 5.2 and 5.95 mcg/ml, and 1,5 mcg/ml by GBS7 extract, which is two times lower than acarbose (3.7 mcg/ml) as a positive control. Considering that high enzyme inhibition values with low IC50 values indicate the effectiveness of the inhibitory effect of the substance, it can be assumed that the endophytic isolates Gingko biloba GBL4, GBL5 from the leaves, and GBL5 from the item of the plant have significant potential as sources of effective inhibitors of two pancreatic enzymes - lipase and amylase.
Keywords: Obesity, Pancreatic lipase, Pancreatic α-amylase, Inhibitors, Endophytic fungi, Ginkgo biloba, Ethyl acetate extracts
Cite this paper: Tashkhan Gulyamova, Dilaram Ruziyeva, Behruz Tojiyev, Gulchekhra Rasulova, Liliya Abdulmyanova, Maftuna Yoldosheva, Kseniya Kondrasheva, Inhibition of Pancreatic Lipase and α-amylase by Gingko Biloba Endophytes, International Journal of Virology and Molecular Biology, Vol. 15 No. 1, 2026, pp. 15-20. doi: 10.5923/j.ijvmb.20261501.04.
Article Outline
1. Introduction
- Obesity and diabetes mellitus (DM) are essential medical and social problems that have reached the scale of a global epidemic [1,2]. According to the World Health Organization (WHO), the prevalence of obesity has increased significantly in recent decades, and this condition now affects millions of people around the world. According to the forecast of the International Diabetes Federation, the number of diabetics may increase to 552 million by 2030 [3], while WHO reports that more than 1 billion adults in the world are overweight, 300 million of them are obese [1]. Meanwhile, numerous studies indicate a close relationship between these two diseases. Obesity is one of the main risk factors for the development of metabolic disorders, including DM2 [4].It is known that in the treatment strategy for both diabetes and obesity, the most effective method is the use of inhibitors of key enzymes of lipid and carbohydrate metabolism. Thus, pancreatic lipase (PL) plays a key role in the hydrolysis of triglycerides in the intestine, and its inhibition leads to a decrease in fat absorption, which makes PL inhibitors effective drugs to combat obesity [5,6]. Pancreatic α-amylase (PA) is an important enzyme involved in the hydrolysis of starch to maltose and glucose, and its inhibitors prevent postprandial hyperglycemia, which is a significant factor in the treatment of diabetes [7,8]. Currently, several enzyme inhibitors are available on the market and successfully used in therapeutic practice [9–11]. Such practical medicine in treating diabetes and obesity includes pancreatic alpha-amylase and lipase inhibitors, such as acarbose and orlistat [12]. However, long-term use of these inhibitors is accompanied by severe side effects, including hepatotoxicity, gallstones, kidney stones, acute pancreatitis, abdominal discomfort, bloating, flatulence, and diarrhea [13], which necessitates the development of new safe and effective drugs for natural nature.Recent studies have convincingly proved that endophytes are a prosperous and valuable source of natural products with various chemical structures and biological activity, making them essential for discovering and developing new medicines [14,15]. In particular, it has been shown that endophytes of many medicinal plants produce compounds that can serve as inhibitors of several therapeutically essential enzymes, including pancreatic amylase and lipase [16–18].Ginkgo biloba is one of the most famous and popular medicinal plants. Due to the wide range of bioactive properties of the compounds contained in G. biloba, many different therapeutic phytopreparations have been developed and are commercially widely available [19]. However, given that the plant grows for a long time, it is interesting to study the endophytes associated with it. Thus, many endophytic fungi have been isolated from the leaves, fruits, bark, stem, and roots of G. biloba seedlings, classified in various genera and producing such phytocompounds as flavonoids, terpenoids, and other compounds [20,21]. It has been shown that the secondary metabolites of G. biloba endophytes exhibit various bioactive properties and are of interest for developing commercial prodrugs.Most importantly, from a therapeutic point of view, G. biloba has no side effects even after prolonged use. According to the available data on the endophytes inhabiting G. biloba, more than 30 different genera of microorganisms have been identified, among which three recently described species [22]. Over 60 secondary metabolites have been reported to have a wide range of activities, including antimicrobial, antioxidant, antitumor, and biocontrol. However, despite the variety of metabolites and their bioactive properties, there needs to be more information in the literature about G. biloba endophytes inhibiting amylase or lipase.The objective of our study was isolation of endophytic fungi associated with Ginkgo biloba and studying the inhibitory activity of their secondary metabolites on pancreatic lipase (PL) and pancreatic α-amylase (PA) to assess the prospects of using G. biloba endophytes as a basis for the development of new prodrugs against obesity and diabetes.
2. Material and Methods
- Objects of researchThe object of the study was endophytic fungi isolated from the leaves and stem of G. biloba, growing in the Tashkent region.Isolation of fungal endophytesThe isolation of endophytic fungi was carried out according to Hazalin et al. [23] from the roots, stems, leaves, and inflorescences of collected plants. After pretreatment each segment of the plant was aseptically crushed and placed on Petri dishes with agarized Chapek-Dox medium containing chlortetracycline at a concentration of 50 mg/ml and streptomycin sulfate at a concentration of 250 mg/ml to suppress the growth of bacterial microflora. The cups were incubated for 7–14 days at a temperature of 28°C. Individual colonies of endophytes grown after incubation were selected using a thin needle, transferred to tubes with agar and incubated at 28°C for seven days. A Chapek-Dox medium with an antibiotic was used as a control.Endophyte fungal cultures are stored by periodic re-sowing on Chapek-Dox agar. All isolates are stored in the refrigerator at +4°C.Cultivation of endophytesEndophytes were grown submerged in Chapek-Dox medium on an orbital shaker at 120 rpm for seven days at 28°C. After the cultivation, the biomass was separated from the fermentation liquid by centrifugation at 6000 rpm for 15 minutes.Extraction of secondary metabolitesAbout 5 g of biomass was homogenized and extracted twice with 25 ml ethyl acetate for 24 hours on an orbital shaker at room temperature. Then, the mixture was filtered through a paper filter (Watman paper No. 1), and Na₂SO₄ was added at a rate of 40 μg/ml to remove the water layer. The mixture was evaporated dry on a rotary evaporator, and 1 ml of dimethyl sulfoxide was added. The resulting extract was used as a stock and stored at +4°C.Determination of lipase inhibitory activityAbout 50 mg of bovine pancreatic lipase (“Sigma”, 100 U/ml) was suspended in 10 ml of tris-NaCl buffer, pH 7.4. The solution was shaken for 15 minutes, followed by centrifugation at 4000 rpm for 10 min. The supernatant was collected and used as an enzyme solution. Initial solutions of extracts and Xenical were prepared in DMSO with concentrations of 10 mg/ml. The final reaction mixture contained 875 µl of buffer, 100 µl of the enzyme, and 20 µl of extract, pre-incubated for 5 minutes at 37°C, followed by the addition of 10 µl of the substrate (4-nitrophenyl palmitate, 10 mM in acetonitrile). The optical density of the final mixture was measured after 5 min (SPEKOL–1300) at 405 nm [24]. The percentage of inhibition was calculated using the formula:% inhibition of PL = [(Ae − At) / Ae] × 100,where Ae is the optical density of the enzyme control (without an inhibitor), At is the difference between the optical density of the test sample with and without a substrate.Determination of α-amylase inhibitory activityThe activity of α-amylase was determined spectrophotometrically at 630 nm as described by Picot et al. [25].Determination of IC₅₀ valueThe semi-maximal concentration of active extracts, with an inhibitory activity of more than 70%, was carried out within a range from 0.1 to 0.5 mg/ml. The IC₅₀ value was calculated from the regression curve using the least squares method of a semi-logarithmic graph relative to the percentage inhibition curves using GraphPad Prism version 4.0 [26].All measurements were carried out in triple.
3. Results and Discussion
- Ginkgo Biloba is a scarce plant in Uzbekistan. There are at most five such trees in the Tashkent region. In our work, we used the leaves and stems of one of the plants growing on the territory of the Kibray sanatorium, Tashkent. As a result, only ten isolates of endophytic fungi were isolated, which, according to morphophysiological characteristics, were previously classified as Aspergillus, Penicillium and Fusarium (Fig. 1).
 | Figure 1. Isolates of Ginkgo biloba endophytic fungi on Chapek-Dox agar |
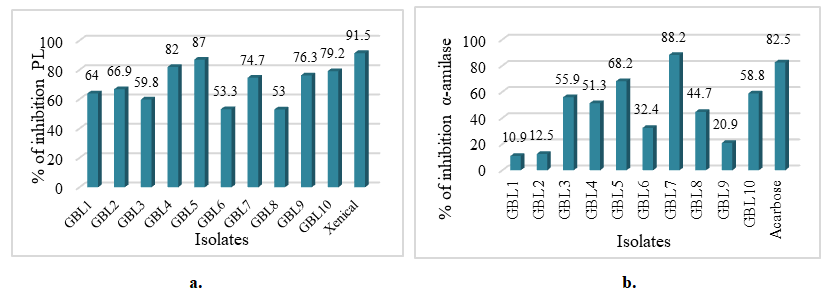 | Figure 2. PL-inhibiting activity (a) and PA - inhibiting activity (b) of Gingko biloba endophytes |
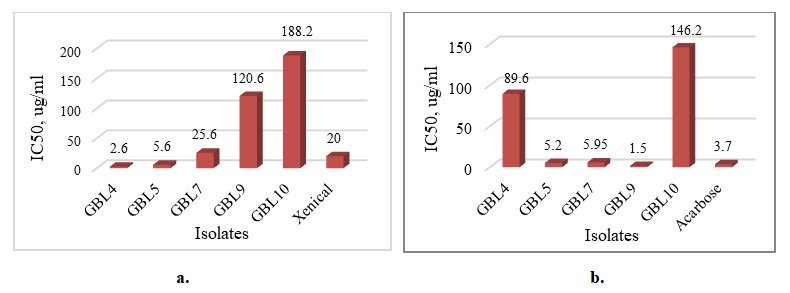 | Figure 3. IC50 lipase inhibition (a) and IC50 α-amilase inhibition (b) by Gingko biloba endophytes |
|
4. Conclusions
- Thus, it is shown for the first time that Ginkgo biloba endophytes represent a promising object for biotechnological and pharmaceutical research to create new effective inhibitors of pancreatic lipase and α-amylase. The high inhibitory activity of extracts of these endophytes indicates their potential effectiveness as natural inhibitors of these enzymes, which may contribute to developing new therapeutic agents for controlling lipid and carbohydrate metabolism.
ACKNOWLEDGEMENTS
- The study was carried out within the framework of basic funding.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML