Zilola Sh. Rabimova1, Dilzoda M. Azizova2, Feruza X. Inoyatova3
1Doctoral Student, Tashkent State Medical University, Tashkent, Uzbekistan
2DSc, Tashkent State Medical University, Tashkent, Uzbekistan
3DSc.prof., Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Zilola Sh. Rabimova, Doctoral Student, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
In metabolic syndrome, systemic inflammation is accompanied by increased activity of neutrophils and macrophages. This condition is manifested by: increased levels of anti-MPO, excessive production of reactive oxygen species, and endothelial and epithelial damage. A sharp increase in CHI3L1 levels reflects the activation of fibroblasts and increased extracellular matrix synthesis against this background of inflammation.
Keywords:
Metabolic syndrome, Anti-MRO, Alpha-1-anti-trypsin, Dyslipidemia, Insulin resistance
Cite this paper: Zilola Sh. Rabimova, Dilzoda M. Azizova, Feruza X. Inoyatova, Changes in Non-Respiratory and Respiratory Indicators of the Lungs and Their Interrelationship, International Journal of Virology and Molecular Biology, Vol. 15 No. 1, 2026, pp. 11-14. doi: 10.5923/j.ijvmb.20261501.03.
1. Introduction
Pulmonary fibrosis is a progressive and irreversible disease driven by chronic inflammation, oxidative stress, protease-antiprotease imbalance, and microvascular injury, leading to excessive matrix deposition and alveolar destruction [1].Elevated anti-myeloperoxidase (anti-MPO) autoantibodies are a key marker of neutrophil-mediated pulmonary capillaritis and vasculitis, directly contributing to alveolar hemorrhage and fibrotic remodeling in ANCA-associated vasculitis [2].Alpha-1-antitrypsin (AAT) is the primary inhibitor of neutrophil elastase, protecting lung parenchyma. In addition to its antiprotease function, AAT exerts strong anti-inflammatory effects: it inhibits NF-κB, reduces pro-inflammatory cytokines (TNF-α, IL-1β, IL-6, IL-8), suppresses neutrophil migration, attenuates oxidative stress, and prevents apoptosis of alveolar cells [4].Serum levels of surfactant proteins SP-A and SP-D, as well as YKL-40 (CHI3L1), are well-validated biomarkers of alveolar epithelial injury and active fibrogenesis [5].The aim of this study was to measure serum concentrations of SP-A, SP-D, YKL-40, anti-MPO, and AAT in experimental animals using ELISA.
2. Materials and Methods
Determination of lung fibrosis markers (surfactant proteins SP-A and SP-D, YKL-40 (CHI3L1)) in blood serum by ELISA.Lung fibrosis markers surfactant proteins SP-A and SP-D, as well as YKL-40 (CHI3L1) were measured in the blood serum of experimental animals using the enzyme-linked immunosorbent assay (ELISA) method.For this purpose, the animals were fasted for 10–12 hours. Blood samples were collected into anticoagulant-free tubes and allowed to clot at room temperature for 20–30 minutes. The samples were then centrifuged at 3000 rpm for 10–15 minutes. Serum was carefully separated. The markers were determined using the following reagents and materials: PBS (pH 7.4), PBS-Tween 0.05% (washing buffer), 1% BSA (blocking buffer), primary antibodies, secondary antibodies, and microplates.The results are presented as mean ± standard deviation (M ± SD). Differences were considered statistically significant at P < 0.05.
3. Results
Anti-MPO is an autoantibody directed against the enzyme myeloperoxidase (MPO) on neutrophils. High levels of anti-MPO are the most important marker of lung damage associated with vasculitis and capillaritis in lung tissue.Alpha-1-antitrypsin (AAT / A1AT) is a protease inhibitor of the serpin family synthesized in the liver. Normal values (by immunoturbidimetry method): 0.9–2.0 g/l (in some methods 2–4 g/l).Its main functions are to inhibit neutrophil elastase from respiratory functions, protecting the alveolar tissue of the lungs. Deficiency leads to early emphysema, chronic pulmonary insufficiency, and the development of bronchiectasis. Nonrespiratory functions: Blocks NF-κB → reduces cytokines such as TNF-α, IL-1β, IL-6, IL-8, reduces neutrophil migration and degranulation, reduces TLR2/4 expression, enhances protection against LPS (bacterial toxins), blocks apoptosis (protects alveolar epithelial and endothelial cells), reduces oxidative stress.Table 1. Changes in lung function in metabolic syndrome
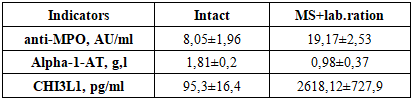 |
| |
|
Analysis based on tabular data showed that in the group with metabolic syndrome, an increase in anti-MPO levels was observed simultaneously with a decrease in α1-antitrypsin levels. In the intact group, anti-MPO levels were low (8.05 AU/ml) and α1-AT levels were relatively high (1.81 g/l), while in the metabolic syndrome group, anti-MPO levels were significantly increased (19.17 AU/ml) and α1-antitrypsin levels were decreased (0.98 g/l).The anti-MPO index is important in assessing immune-oxidative imbalance in the setting of metabolic syndrome. The level of anti-MPO is low in the intact group, indicating that neutrophil activity and oxidative stress are at physiological levels.The significant increase in anti-MPO levels in the MS + laboratory diet group indicates increased neutrophil activation, myeloperoxidase release, and autoantibody formation against it. It has been reported in the literature that hypochlorous acid produced by MPO enhances lipid peroxidation and endothelial damage, which stimulates an autoimmune response.α1-antitrypsin is the main protease inhibitor that protects tissues from neutrophil elastase and other proteolytic enzymes. While the α1-AT level was normal in the intact group, its sharp decrease was noted in the MS + laboratory diet group.This condition indicates that the protease-antiprotease balance is disturbed and the risk of tissue damage is increased. According to scientific data, α1-AT deficiency accelerates the development of inflammation, fibrosis and destructive processes in lung tissue.According to the table, the level of CHI3L1 (YKL-40) in intact rats was 95.3±16.4 pg/ml, while in rats with metabolic syndrome (MS) this indicator increased sharply to 2618.12±727.9 pg/ml.CHI3L1 is a glycoprotein involved in inflammation, tissue remodeling and fibrosis, and is synthesized mainly by macrophages, neutrophils, fibroblasts and epithelial cells. Its high level indicates the activation of chronic inflammation and fibrosis processes in the tissue.In conditions of metabolic syndrome, systemic inflammation occurs with increased activity of neutrophils and macrophages. This is manifested by: increased levels of anti-MPO, excessive production of reactive oxygen species, endothelial and epithelial damage. A sharp increase in CHI3L1 levels reflects the activation of fibroblasts and increased extracellular matrix synthesis against this background of inflammation.Anti-MPO ↑ → oxidative stress ↑ → CHI3L1 expression ↑ This chain forms the molecular basis of fibrotic processes in lung tissue.Pathogenetic relationship between CHI3L1 and α1-antitrypsinA decrease in the level of α1-antitrypsin leads to a violation of the protease-antiprotease balance. As a result: tissue damage deepens, normal regeneration mechanisms are lost, and fibrosis-type regeneration prevails.Under these conditions, CHI3L1 promotes fibroblast migration and collagen synthesis, thereby enhancing fibrosis in lung tissue. Thus, the combination of α1-AT ↓ + CHI3L1 ↑ is a high-risk biomarker profile for pulmonary fibrosis.Correlation between CHI3L1 and lipid metabolism parametersDyslipidemia and lipotoxic effects observed in metabolic syndrome: shift macrophages to a proinflammatory phenotype, increase CHI3L1 synthesis, and activate the action of cytokines (TGF-β) that enhance the fibrosis process. In lung tissue, these processes disrupt surfactant metabolism and lead to remodeling of alveolar structures. As a result, lipid metabolism disorders appear as a factor accelerating lung fibrosis through CHI3L1.CHI3L1 and hemodynamic changesChronic hypoxia, which develops as a result of arterial hypertension and microcirculatory disorders, leads to HIF-1α activation, increased expression of CHI3L1 by macrophages and fibroblasts, and collagen deposition. This indicates that CHI3L1 is an important link between hemodynamic disorders and pulmonary fibrosis.Metabolic syndrome → dyslipidemia + insulin resistance → systemic inflammation → anti-MRO ↑ → α1-antitrypsin ↓ → oxidative stress + protease dominance → CHI3L1 ↑ → fibroblast activity and collagen synthesis → endothelial/alveolar remodeling → pulmonary fibrosis.These changes indicate a negative correlation between anti-MPO and α1-antitrypsin. From a biological point of view, this phenomenon is explained by a violation of the protease-antiprotease balance in the lungs against the background of increased neutrophil activity and increased inflammatory processes. An increase in the level of anti-MPO indicates an increase in the release of myeloperoxidase from neutrophils, and a decrease in α1-antitrypsin indicates a weakening of protective mechanisms against the effects of proteolytic enzymes.The results show a clear tendency for α1-antitrypsin levels to decrease as the anti-MRO index increases. This condition confirms the weakening of the protective mechanism against the background of increased inflammatory processes in the lungs. According to scientific sources, myeloperoxidase is an important marker of neutrophil activity, and its increase has been shown to be associated with oxidative stress and tissue damage (Klebanoff; Hansson). At the same time, α1-antitrypsin is recognized as the main antiprotease that protects lung tissue from the effects of neutrophil elastase and other proteases [8]. In metabolic syndrome, systemic inflammation and insulin resistance lead to increased neutrophil activity and increased myeloperoxidase production, which leads to rapid depletion of α1-antitrypsin reserves and compromises the nonrespiratory functions of the lung detoxification, immunological defense, and metabolism of biologically active substances [7].The results obtained are consistent with the data presented in the literature and confirm that in metabolic syndrome the lung is not only an organ of respiration, but also an organ actively involved in systemic metabolic and inflammatory processes.LEVELS OF SURFACTANT PROTEINS (SP-A AND SP-D) IN SERUM OF RATS WITH METABOLIC SYNDROMEIn addition to its central role in maintaining respiratory mechanics, the pulmonary surfactant system (PS) has been recognized over the past 20–25 years as having other important functions. These include barrier function, innate and adaptive immunity, and regulation of inflammation. In particular, surfactant proteins (SPs) SP-A and SP-D serve as the first line of defense against microorganisms, viruses, allergens, and toxic substances. They also play a role in maintaining normal bronchial patency, and therefore, PS disorders play an important role in the pathogenesis of bronchial asthma, interstitial lung diseases, and pulmonary fibrosis.The main functions of surfactant components: DPPH is the main lipid that provides the biophysical properties of the ST, anionic phospholipids (phosphatidylglycerol - FG) - increase the stability of the monolayer, improve adsorption, SP-B and SP-C - facilitate the phase transition of lipids, maintain the uniformity of the alveolar surface, protect the ST from inactivation. SP-A and SP-D - have immunomodulatory and antimicrobial functions: bind LPS (lipopolysaccharide), agglutinate bacteria and viruses, regulate the activity of alveolar macrophages (AM), dendritic cells, lymphocytes and mast cells, SP-D is more potent in clearing cells that have undergone apoptosis, while SP-A has both pro- and anti-inflammatory effects. Deletion of SP-A and SP-D genes in transgenic mice has been shown to accelerate the development of emphysema, acute lung injury, and fibrosis. Serum levels of SP-A and SP-D have become one of the most important biomarkers of lung inflammation, fibrosis, and infection. Metabolic disorders (obesity, insulin resistance) strengthen the link between ST impairment and the development of pulmonary fibrosis.SP-A (Surfactant Protein A) ng/ml and SP-D (Surfactant Protein A) pg/ml proteins are important components of the pulmonary alveolar surfactant system and participate in the immune and inflammatory defense functions of the lungs.Table 2. Serum levels of surfactant proteins SP-A and SP-D in intact (healthy) rats and rats with metabolic syndrome receiving a laboratory ration
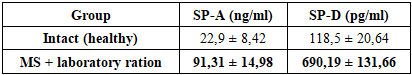 |
| |
|
Normally, SP-A plays a role in lung immunity, phagocytosis of microbes, and inhibition of inflammation, but the level of antibodies to this protein was 4 times higher in rats with MS than in intact rats. This indicates the onset of inflammation, damage, and fibrosis in the lungs.SP-D in normal amounts performs antimicrobial protection, regulation of apoptosis, anti-inflammatory, and anti-fibrotic functions. The level of antibodies to the SP-D protein was ~5.8 times higher than in intact animals, and this result indicates the development of very severe inflammation and damage in the alveoli.
4. Conclusions
The present study demonstrated that experimental metabolic syndrome in rats is accompanied by significant elevation of key lung fibrosis biomarkers in blood serum. Serum levels of surfactant proteins SP-A and SP-D increased 4-fold and 5.8-fold, respectively, compared with intact animals. These elevations, together with increased anti-MPO autoantibodies and reduced functional activity of alpha-1-antitrypsin, indicate pronounced alveolar epithelial injury, neutrophil-mediated inflammation, protease–antiprotease imbalance, and active fibrotic remodeling in the lungs.SP-A and SP-D, which normally perform protective immunomodulatory and anti-inflammatory functions, become sensitive markers of pulmonary damage when released into the systemic circulation. Their marked increase in the MS group reflects severe disruption of the pulmonary surfactant system and progression toward interstitial fibrosis.Thus, the combined determination of SP-A, SP-D, YKL-40, anti-MPO, and AAT in serum provides a reliable non-invasive panel for early detection and monitoring of lung fibrosis in conditions associated with metabolic syndrome. These findings support the concept that metabolic disorders accelerate pulmonary fibrogenesis through surfactant dysfunction and chronic low-grade inflammation, opening new perspectives for targeted preventive and therapeutic strategies.
References
| [1] | Wynn TA, Ramalingam TR. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat Med. 2012; 18(7): 1028-1040. |
| [2] | Alba MA, Jennette JC, Falk RJ. Pathogenesis of ANCA-Associated Pulmonary Vasculitis. Semin Respir Crit Care Med. 2018; 39(4): 413-424. |
| [3] | Salvati L, et al. Presentation and progression of MPO-ANCA interstitial lung disease. J Transl Autoimmun. 2024; 8: 100235. |
| [4] | Wang T, et al. α-1 Antitrypsin is a potential target of inflammation and immunomodulation (Review). Mol Med Rep. 2025; 31(4): 107. |
| [5] | Greene KE, et al. Serum surfactant proteins-A and -D as biomarkers in idiopathic pulmonary fibrosis. Eur Respir J. 2002; 19(3): 439-446. |
| [6] | van Moorsel CHM, et al. Serum and BALF YKL-40 levels are predictors of survival in idiopathic pulmonary fibrosis. Respir Med. 2011; 105(1): 106-113. Amjad A Khan 1,*, Mohammed A Alsahli 1, Arshad H Rahmani “Myeloperoxidase as an Active Disease Biomarker: Recent Biochemical and Pathological Perspectives” National institutes of health. |
| [7] | Hotamisligil; Barnes “Imbalance between Neutrophil Elastase and its Inhibitor α1-Antitrypsin in Obesity Alters Insulin Sensitivity, Inflammation, and Energy Expenditure”. |
| [8] | 1 Crystal et al “The multiple facets of alpha-1-antitrypsin” national institutes of health. |


 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML
