-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Virology and Molecular Biology
p-ISSN: 2163-2219 e-ISSN: 2163-2227
2025; 14(6): 93-98
doi:10.5923/j.ijvmb.20251406.01
Received: Aug. 3, 2025; Accepted: Aug. 22, 2025; Published: Aug. 30, 2025

Detection of Autoantibodies against Endothelin-1 and Apolipoprotein B Proteins in Hepatic Tissue and Circulation of Experimental Alzheimer's Disease Model Animals
Amanbayeva S. S.1, Bahodirov H. Q.2, Mustafakulov M. A.3
1Kokand University Andijan Branch, Andijan State University, Andijan, Uzbekistan
2Institute of Biophysics and Biochemistry, National University of Uzbekistan, Tashkent, Uzbekistan
3Department of Biochemistry, Andijan State Medical Institute, Andijan, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Non-alcoholic fatty liver disease associated with Alzheimer's disease (NAFLD-AD) represents a complex pathophysiological condition characterized by hepatic steatosis and disrupted lipid homeostasis. The relationship between neurodegeneration and hepatic metabolic dysfunction involves multiple biomarkers, including endothelin-1 (ET-1) and apolipoprotein B (ApoB). Objective: To investigate the presence and dynamics of autoantibodies against ET-1 and ApoB proteins in hepatic tissue and blood circulation of experimental Alzheimer's disease model animals, and to analyze associated biochemical alterations. Methods: Twenty-four male Wistar rats (324±16 g) were divided into five groups: control (n=11), Alzheimer's disease model (n=15), AD+Quercetin (n=15), AD+SMC (1:9) (n=14), and AD+SMC (1:5) (n=17). Alzheimer's disease was induced using intranasal streptozotocin (3 mg/kg). Blood samples were collected at 7, 14, and 21-day intervals for biochemical analysis. ET-1 and ApoB levels were determined using ELISA methodology. Results: Significant dyslipidemia was observed in AD model animals, including elevated total cholesterol (1.86-fold, p<0.01), triglycerides (2.67-fold, p<0.01), and LDL-cholesterol (2.71-fold, p<0.01), with concurrent 33% reduction in HDL-cholesterol levels. Inflammatory markers showed substantial increases: C-reactive protein (4-fold), fibrinogen (1.75-fold), and IL-6 (2.92-fold). ET-1 levels increased 1.66-fold in blood serum and 1.04-fold in liver tissue. ApoB concentrations rose 2.42-fold in serum and 2.18-fold in hepatic tissue compared to controls. Conclusions: The experimental NAFLD-AD model demonstrates significant cardiovascular risk markers through elevated ET-1 and ApoB levels, suggesting a strong correlation between neurodegeneration and hepatic metabolic dysfunction. These findings support the potential use of these biomarkers for early detection and monitoring of NAFLD-AD progression.
Keywords: Alzheimer's disease, Endothelin-1, Apolipoprotein B, Hepatic steatosis, Autoantibodies, Neurodegeneration, Cardiovascular risk
Cite this paper: Amanbayeva S. S., Bahodirov H. Q., Mustafakulov M. A., Detection of Autoantibodies against Endothelin-1 and Apolipoprotein B Proteins in Hepatic Tissue and Circulation of Experimental Alzheimer's Disease Model Animals, International Journal of Virology and Molecular Biology, Vol. 14 No. 6, 2025, pp. 93-98. doi: 10.5923/j.ijvmb.20251406.01.
Article Outline
1. Introduction
- The interconnection between Alzheimer's disease (AD) and peripheral metabolic dysfunction has gained considerable attention in recent neuroscience research [1,2]. Non-alcoholic fatty liver disease associated with Alzheimer's disease (NAFLD-AD) represents a complex pathophysiological state characterized by hepatic lipid accumulation concurrent with progressive neurodegeneration [3]. Recent evidence suggests that metabolic dysfunction-associated fatty liver disease may serve as a more appropriate nomenclature for describing hepatic pathologies linked to known metabolic disorders [4]. The prevalence of NAFLD-AD ranges from 1.5% to 6.5% globally, though absolute prevalence remains uncertain due to genetic and environmental variability [5]. This condition involves multiple pathophysiological mechanisms, including insulin resistance, chronic inflammation, oxidative stress, and disrupted lipid metabolism [6]. Insulin resistance plays a pivotal role in NAFLD-AD pathogenesis by inducing long-term hypertriglyceridemia, which subsequently impairs endothelium-dependent vasodilation and promotes oxidative stress development [7]. This cascade serves as a critical prognostic marker for atherosclerosis progression, characterized by specific biochemical alterations in blood serum [8]. Endothelin-1 (ET-1), a potent vasoconstrictor peptide, has emerged as a significant biomarker for cardiovascular system damage in metabolic disorders [9]. Elevated ET-1 concentrations in blood serum and plasma correlate with enhanced vascular tone, increased arterial pressure, impaired microcirculation, and cardiac muscle hypoxia [10]. Recent studies have demonstrated that ET-1 synthesis increases in endothelial cells under metabolic syndrome conditions, particularly in the presence of insulin resistance and chronic inflammation [11].Apolipoprotein B (ApoB) constitutes the structural component of major atherogenic lipoprotein particles and reflects their total concentration in circulation [12]. The ApoB protein family plays crucial roles in lipid transport and metabolism, with ApoB-100 being particularly important in low-density lipoprotein formation and clearance [13]. Increased ApoB levels indicate impaired receptor-mediated endocytosis and suggest enhanced cardiovascular risk [14]. The relationship between neurodegeneration and peripheral metabolic markers remains incompletely understood. Growing evidence suggests that autoimmune mechanisms may contribute to both neuronal damage and hepatic dysfunction in AD patients [15]. Autoantibodies against various proteins, including ET-1 and ApoB, may serve as novel biomarkers for disease progression and therapeutic monitoring. Current diagnostic approaches for NAFLD-AD rely primarily on imaging techniques and biochemical markers. However, the identification of specific autoantibodies could provide earlier detection capabilities and improved prognostic accuracy. Furthermore, understanding the temporal dynamics of these biomarkers may facilitate targeted therapeutic interventions.ObjectiveTo detect and quantify autoantibodies against endothelin-1 and apolipoprotein B proteins in hepatic tissue and blood circulation of experimental Alzheimer's disease model animals, and to investigate the temporal dynamics of associated biochemical parameter changes.
2. Materials and Methods
- Experimental Animals and EthicsThe research was conducted at the Metabolomics Laboratory of the Institute of Biophysics and Biochemistry, National University of Uzbekistan. Twenty-four male Wistar rats weighing 324±16 g (age 6-8 weeks) were utilized for the experimental procedures. All studies were performed in accordance with international guidelines for laboratory animal care and use, including the Helsinki Declaration (World Medical Association, Edinburgh, 2000), Council for International Organizations of Medical Sciences (CIOMS) standards, and the "Guide for the Care and Use of Laboratory Animals". Animals were housed in standard laboratory conditions with controlled temperature (22±2°C), humidity (55±10%), and 12-hour light/dark cycles. Food and water were provided ad libitum throughout the experimental period. All procedures were approved by the institutional animal care and use committee.Experimental Design and GroupsExperimental animals were randomly allocated into five groups:Group I: Control group (n=11) - received saline vehicleGroup II: Alzheimer's disease model (n=15) - streptozotocin-induced ADGroup III: AD + Quercetin treatment (n=15) - received quercetin supplementationGroup IV: AD + SMC (1:9) (n=14) - received supramolecular complex compound at 1:9 ratioGroup V: AD + SMC (1:5) (n=17) - received supramolecular complex compound at 1:5 ratioAlzheimer's Disease Model InductionAlzheimer's disease was induced using intranasal administration of streptozotocin (STZ) at a dose of 3 mg/kg body weight. STZ was dissolved in sterile phosphate-buffered saline (pH 7.4) immediately before administration. Animals were lightly anesthetized with isoflurane, and STZ solution was administered dropwise into both nostrils using a micropipette.Treatment ProtocolsFollowing a 14-day establishment period post-STZ administration, treatment groups received their respective interventions:Group III: Quercetin (50 mg/kg) administered orally once daily for 7 daysGroup IV: Supramolecular complex compound (1:9 ratio, 40 mg/kg) administered orally once daily for 7 daysGroup V: Supramolecular complex compound (1:5 ratio, 50 mg/kg) administered orally once daily for 7 daysThe supramolecular complex compound used in this study represents a novel therapeutic approach for treating hepatic alterations in experimental neurodegeneration models, demonstrating enhanced bioavailability and targeted delivery properties compared to conventional treatments.Blood Sample CollectionBlood samples (200-300 μL) were collected from the tail vein at predetermined intervals (days 7, 14, and 21) using 27-gauge needles. Prior to blood collection, animals were placed in warm water (38-40°C) for 2-3 minutes to facilitate vasodilation. Samples were collected in EDTA-coated microtubes and immediately processed or stored at -80°C until analysis.Biochemical Parameter AnalysisLipid Profile DeterminationTotal cholesterol: Enzymatic cholesterol oxidase methodTriglycerides: Glycerol-3-phosphate oxidase-phenol aminophenazone (GPO-PAP) methodHDL-cholesterol: Precipitation method with phosphotungstic acidLDL-cholesterol: Calculated using Friedewald equationAtherogenic coefficient: Calculated as (Total cholesterol - HDL)/HDLInflammatory Marker AssessmentC-reactive protein (CRP): High-sensitivity immunoturbidimetric assayFibrinogen: Clauss clotting time methodInterleukin-6 (IL-6): Enzyme-linked immunosorbent assay (ELISA)ET-1 and ApoB Detection MethodologyEndothelin-1 and apolipoprotein B concentrations were determined using commercial ELISA kits according to manufacturer protocols. Briefly, microplate wells pre-coated with specific capture antibodies were incubated with 100 μL of appropriately diluted serum samples and standards at 37°C for 90 minutes. Following primary incubation, wells were washed three times with phosphate-buffered saline containing 0.05% Tween-20 (PBST). Detection antibody (100 μL) was added and incubated at 37°C for 60 minutes. After washing cycles, horseradish peroxidase-conjugated secondary antibody was applied and incubated for 30 minutes at 37°C. Color development was achieved using tetramethylbenzidine (TMB) substrate solution (90 μL per well) with incubation at 37°C for 15 minutes. The reaction was terminated using 2M sulfuric acid (50 μL per well), and optical density was measured at 450 nm using a microplate reader.Statistical AnalysisStatistical analyses were performed using Microsoft Excel and OriginPro 8.6 software. Data are presented as mean ± standard error of the mean (SEM). Statistical significance was assessed using Student's t-test for pairwise comparisons and one-way analysis of variance (ANOVA) for multiple group comparisons. Post-hoc analyses were conducted using Tukey's honest significant difference test. Statistical significance was defined as p<0.05, with additional significance levels at p<0.01 and p<0.001.
3. Results
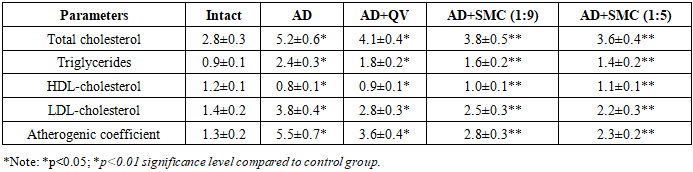
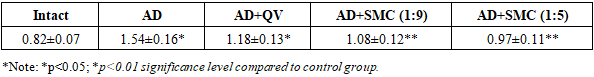
- Experimental Animal CharacteristicsAll experimental procedures were successfully completed using the designated animal groups. No significant adverse effects were observed during the experimental period, and animal welfare was maintained throughout the study duration.Blood Lipid Profile AlterationsSignificant dyslipidemia was observed in the Alzheimer's disease model group compared to control animals (Table 1). Total cholesterol levels increased 1.86-fold (from 2.8±0.3 to 5.2±0.6 mmol/L, p<0.01), while triglyceride concentrations showed a 2.67-fold elevation (from 0.9±0.1 to 2.4±0.3 mmol/L, p<0.01).
|
|
|
|
|
|
|
|
4. Discussion
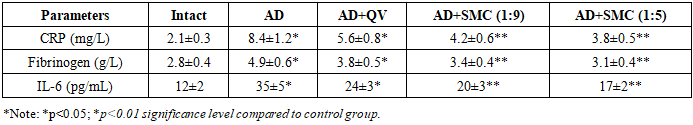
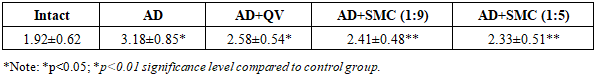
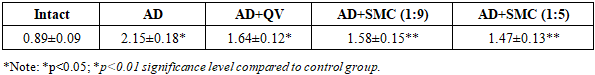
- The present study demonstrates significant alterations in autoantibody levels against ET-1 and ApoB proteins in an experimental NAFLD-AD model, providing novel insights into the interconnection between neurodegeneration and peripheral metabolic dysfunction. Our findings reveal a complex pathophysiological network involving lipid metabolism disruption, inflammatory activation, and cardiovascular risk factor elevation.Lipid Metabolism DysregulationThe observed dyslipidemia pattern in our AD model aligns with clinical observations in patients with metabolic syndrome and neurodegenerative disorders. The 1.86-fold increase in total cholesterol, coupled with the 2.71-fold elevation in LDL-cholesterol, indicates severe disruption of hepatic lipid homeostasis. These changes likely result from impaired insulin signaling, enhanced hepatic glucose production, and altered fatty acid metabolism [3]. The 33% reduction in HDL-cholesterol levels is particularly concerning, as HDL particles play crucial roles in reverse cholesterol transport and anti-inflammatory processes. This reduction may contribute to accelerated atherosclerosis development and increased cardiovascular mortality risk in AD patients. The dramatic increase in atherogenic coefficient (from 1.3 to 5.5) exceeds the threshold value of 3.0 typically associated with high cardiovascular risk. This finding suggests that even in experimental AD models, significant atherosclerotic processes may develop rapidly, highlighting the need for early intervention strategies.Inflammatory Cascade ActivationThe substantial elevations in CRP (4-fold), fibrinogen (1.75-fold), and IL-6 (2.92-fold) confirm the presence of systemic inflammatory processes in our NAFLD-AD model. These findings support the neuroinflammation hypothesis of AD pathogenesis and suggest that peripheral inflammatory markers may serve as accessible biomarkers for disease monitoring.C-reactive protein, an acute-phase reactant synthesized primarily in hepatocytes, reflects the intensity of systemic inflammation. The observed 4-fold increase suggests significant hepatic involvement in the inflammatory response, which may contribute to both neuronal damage and hepatic steatosis progression.Interleukin-6, a pleiotropic cytokine involved in both neuroinflammation and metabolic regulation, showed particularly pronounced elevation. This finding aligns with clinical studies demonstrating elevated IL-6 levels in AD patients and suggests potential therapeutic targets for anti-inflammatory interventions.Endothelin-1 as a Cardiovascular Risk MarkerThe differential increase in ET-1 levels between hepatic tissue (1.34-fold) and blood serum (1.66-fold) provides insights into the systemic nature of endothelial dysfunction in NAFLD-AD. ET-1, recognized as one of the most potent vasoconstrictors, plays critical roles in vascular tone regulation, blood pressure control, and microcirculatory function [9]. The preferential elevation of ET-1 in circulation suggests enhanced endothelial cell activation and potential autocrine/paracrine signaling disruption. This finding may explain the increased cardiovascular mortality observed in AD patients and supports the vascular hypothesis of AD pathogenesis. Recent studies have demonstrated that ET-1 can directly influence neuronal function and contribute to blood-brain barrier disruption. Our findings suggest that peripheral ET-1 elevation may serve as both a consequence of and contributor to neurodegeneration, creating a vicious cycle of vascular and neuronal damage.Apolipoprotein B and Atherogenic RiskThe consistent increase in ApoB levels across both hepatic tissue (2.18-fold) and blood serum (2.42-fold) indicates severe disruption of lipoprotein metabolism. ApoB-100, the primary apolipoprotein component of VLDL and LDL particles, serves as a marker for atherogenic lipoprotein particle number rather than cholesterol content alone [12]. The elevation in ApoB levels suggests impaired hepatic VLDL assembly, enhanced VLDL secretion, or reduced LDL clearance through hepatic receptors. These mechanisms are commonly observed in insulin-resistant states and may contribute to accelerated atherosclerosis development in AD patients.The LDL-cholesterol to ApoB ratio analysis reveals the presence of small, dense LDL particles, which possess enhanced atherogenic potential due to increased oxidative susceptibility and arterial wall penetration capacity. This finding supports the concept that qualitative lipoprotein analysis may provide superior cardiovascular risk assessment compared to traditional cholesterol measurements alone.Therapeutic ImplicationsThe differential responses to treatment interventions provide valuable insights into potential therapeutic strategies for NAFLD-AD management. The supramolecular complex compound, particularly at the 1:5 ratio, demonstrated superior efficacy in normalizing multiple biomarkers simultaneously, suggesting multi-target therapeutic potential.Quercetin treatment showed intermediate efficacy, consistent with its known antioxidant and anti-inflammatory properties. However, the limited improvement compared to the supramolecular complex suggests that single-target approaches may be insufficient for addressing the complex pathophysiology of NAFLD-AD.Clinical Relevance and Biomarker PotentialThe identification of elevated autoantibodies against ET-1 and ApoB in experimental NAFLD-AD models provides a foundation for clinical biomarker development. These findings suggest that routine monitoring of these parameters in AD patients may facilitate early detection of cardiovascular complications and guide therapeutic decision-making. The temporal dynamics of biomarker changes observed in our study indicate that significant alterations occur within relatively short timeframes, supporting the feasibility of these markers for disease monitoring and treatment response assessment.Study LimitationsSeveral limitations should be acknowledged in interpreting our findings. First, the experimental model may not fully recapitulate the complexity of human NAFLD-AD pathophysiology. Second, the relatively short observation period may not capture long-term disease progression patterns. Third, the specific mechanisms underlying autoantibody generation against ET-1 and ApoB require further investigation.
5. Conclusions
- This study demonstrates significant alterations in autoantibodies against endothelin-1 and apolipoprotein B proteins in experimental NAFLD-AD models, accompanied by substantial disruptions in lipid metabolism and inflammatory marker profiles. The key findings include:Severe dyslipidemia characterized by elevated total cholesterol (1.86-fold), triglycerides (2.67-fold), and LDL-cholesterol (2.71-fold), with concurrent HDL-cholesterol reduction (33%).Systemic inflammatory activation evidenced by increased C-reactive protein (4-fold), fibrinogen (1.75-fold), and interleukin-6 (2.92-fold) levels.Cardiovascular risk marker elevation including ET-1 increases in blood serum (1.66-fold) and liver tissue (1.34-fold), and ApoB elevation in both serum (2.42-fold) and hepatic tissue (2.18-fold).Atherogenic lipoprotein particle predominance indicated by altered LDL-cholesterol to ApoB ratios, suggesting enhanced cardiovascular risk.Therapeutic response variability with supramolecular complex compound (1:5 ratio) demonstrating superior multi-target efficacy compared to conventional antioxidant approaches.These findings support the concept that NAFLD-AD involves complex interactions between neurodegeneration, hepatic dysfunction, and cardiovascular risk factors. The elevated autoantibodies against ET-1 and ApoB may serve as valuable biomarkers for early detection, disease monitoring, and therapeutic response assessment in clinical settings.Future research should focus on validating these biomarkers in human populations, investigating the mechanistic basis of autoantibody generation, and developing targeted therapeutic interventions addressing the multi-system nature of NAFLD-AD pathophysiology.
Funding
- This research was supported by grants from the Ministry of Higher Education, Science and Innovation of the Republic of Uzbekistan and the Institute of Biophysics and Biochemistry, National University of Uzbekistan.
Ethical Approval
- All experimental procedures were conducted in accordance with international guidelines for laboratory animal care and use and were approved by the Institutional Animal Care and Use Committee (Protocol #2024-015).
Conflict of Interest
- The authors declare no conflicts of interest related to this research.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML