-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Virology and Molecular Biology
p-ISSN: 2163-2219 e-ISSN: 2163-2227
2025; 14(5): 86-92
doi:10.5923/j.ijvmb.20251405.05
Received: Aug. 1, 2025; Accepted: Aug. 20, 2025; Published: Aug. 22, 2025

Immunostimulatory Properties of Common Chlorella (Chlorella vulgaris) Solutions
Bolta Kaхorov1, Makhliyo Teshajonova2, Gulchekhra Kadirova3
1Fergana State University, Uzbekistan
2National University of Uzbekistan named after Mirzo Ulugbek, Uzbekistan
3Institute of Microbiology, Academy of Sciences of Uzbekistan
Correspondence to: Bolta Kaхorov, Fergana State University, Uzbekistan.
| Email: |  |
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This study reports the isolation and characterization of various strains of Chlorella vulgaris obtained from plant-based sources. Based on the outcomes of preliminary analyses, a comprehensive experimental framework was developed to evaluate the therapeutic potential of natural biologically active solutions derived from Chlorella vulgaris. The investigation focused on elucidating the mechanisms underlying the quantitative biological activity of these phytogenic solutions. Special emphasis was placed on their immunomodulatory properties and their effectiveness in mitigating immunodeficiency conditions in animals. The experimental results suggest that bioactive compounds present in Chlorella vulgaris exhibit promising immunotherapeutic effects, thereby offering a natural and sustainable alternative for enhancing immune responses in veterinary contexts. The findings contribute to the growing body of research on algae-based medicinal agents and their role in the development of plant-derived immunostimulants.
Keywords: Biostimulant, Chlorella, Beneficial properties, Composition, Caloric content, Antigen, Immunostimulant, Immunocorrector, Serum, Algae, Blood cells, Monocytes, Lymphocytes
Cite this paper: Bolta Kaхorov, Makhliyo Teshajonova, Gulchekhra Kadirova, Immunostimulatory Properties of Common Chlorella (Chlorella vulgaris) Solutions, International Journal of Virology and Molecular Biology, Vol. 14 No. 5, 2025, pp. 86-92. doi: 10.5923/j.ijvmb.20251405.05.
1. Relevance
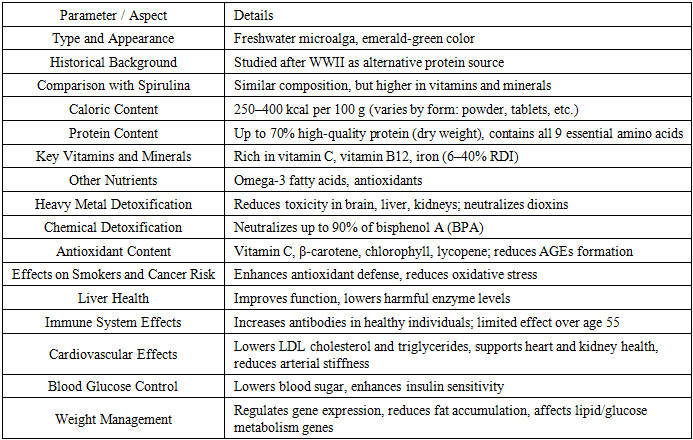
- Chlorella is a freshwater microalga with an emerald-green color. It was first studied after World War II as an alternative protein source for the population. Chlorella shares many similarities in composition and properties with Spirulina, but contains higher amounts of vitamins and minerals. Its caloric content ranges from 250 to 400 kcal per 100 g, depending on the form in which it is consumed (e.g., powder, tablets, etc.). The plant contains up to 70% high-quality protein (by dry weight), comprising all nine essential amino acids. Moreover, Chlorella is a rich source of vitamin C, vitamin B12, and iron—covering 6% to 40% of the recommended daily intake of this mineral. Like other algae, it also provides omega-3 fatty acids and antioxidants that support disease prevention.The rich composition of Chlorella has been shown to reduce the toxicity of heavy metals in the brain, liver, and kidneys. Its components neutralize many highly toxic substances found in food, including dioxins, which are known to disrupt hormonal function. In one study, Chlorella fusca was shown to neutralize up to 90% of bisphenol A (BPA), a chemical commonly leached from plastic food containers. Chlorella contains a wide range of antioxidants, including vitamin C, β-carotene, chlorophyll, and lycopene, which reduce the formation of advanced glycation end products (AGEs)—compounds that contribute to inflammation, chronic diseases, and diabetic complications.Algal supplements have been found to enhance antioxidant defense in chronic smokers and individuals at high risk for cancer. Supplementation also improves liver function in patients with various hepatic disorders, leading to a reduction in enzyme levels that are harmful to the liver. However, the benefits for healthy individuals remain unclear. In healthy subjects, Chlorella intake increases the production of antibodies that help combat foreign agents, enhancing immune defense. Nevertheless, one study indicated that the supplement stimulated immune responses in men and women aged 50–55, but showed no effect in individuals older than 55.Regardless of the form of intake, Chlorella has been shown to reduce levels of low-density lipoprotein (LDL) cholesterol and triglycerides in individuals with hypertension. Scientists suggest that this improvement is due to a combination of beneficial compounds, including dietary fiber, antioxidants, and vitamin B3. Chlorella supplements support cardiovascular and renal health, both of which are essential for maintaining normal blood pressure. They also contribute to reduced arterial stiffness. Researchers hypothesize that nutrients such as omega-3 fatty acids, potassium, calcium, and the amino acid arginine may play a role in preventing arterial hardening.Furthermore, Chlorella intake lowers blood glucose concentrations in individuals at high risk of diabetes and enhances insulin sensitivity, particularly in those with liver conditions [16,17,41,44].Studies conducted in Kyoto, Japan, demonstrated that Chlorella contributes to weight reduction by regulating gene expression and facilitating the conversion of genetic information from DNA nucleotide sequences into functional products such as RNA or proteins. This process leads to a reduction in fat accumulation. Researchers also identified that the genes influenced by Chlorella are involved in signaling pathways, lipid transport, glucose and insulin uptake. Among them are genes associated with metabolism, signaling, receptors, transporters, and cytokines, particularly those involved in fat and insulin metabolic pathways. Given the distinct gene expression profiles related to glucose uptake, it is likely that Chlorella activates insulin signaling pathways, which may explain its blood sugar-lowering effects [22,23,24,42].
 | Figure 1 |
|
2. Research Methods
- The study employed the identification of antibody-forming cells in the spleen. These cells were detected using the local hemolysis in agarose method, as described by Nordin and Jerne (1963). A model of secondary immunodeficiency was established using toxic hepatitis. Rats were administered a 20% solution of carbon tetrachloride (CCl₄) at a dose of 0.2 mL for three consecutive days. On the final day, immunization with sheep red blood cells (SRBCs) was performed, followed by a repeated intraperitoneal injection of CCl₄ to verify model consistency. The model effectively induced secondary immunodeficiency.Sheep erythrocytes (SRBCs) were used as antigens for immunization. The study hypothesized that the number of antibody-forming cells (AFCs) in the spleen and the mean count of hematopoietic cells could be statistically analyzed to assess the effects of treatment. The reliability of the results was set at a 95% confidence level. Red and white blood cells were counted using standard hematological methods and evaluated microscopically with a Goryaev chamber.
3. Results and Discussion
- The activity of natural medicinal bioactive compounds derived from Chlorella vulgaris was found to play a significant role in restoring immune function. Therefore, the effects of Chlorella vulgaris -based preparations on the immune system were investigated experimentally. Additionally, the study included Timalin, a thymus-derived peptide medication, as a comparison.The experimental groups were organized as follows:• Group I – Intact (healthy control);• Group II – CCl₄ control (immunodeficient model);• Group III – CCl₄ + Timalin;• Group IV – CCl₄ + Chlorella vulgaris (0,1%);• Group V – CCl₄ + Chlorella vulgaris (0,5%);• Group VI – CCl₄ + Chlorella vulgaris (1,0%).All animals were housed under identical conditions with temperatures maintained at 20–28°C and humidity at 50%, in separate cages under constant monitoring. No significant external changes were observed, although rats in the CCl₄-treated groups showed reduced appetite and skin changes. The animals treated with Chlorella vulgaris received therapeutic doses for five consecutive days. Following treatment, the number of AFCs in the spleen was quantified, and the immunostimulatory effects of the preparations were evaluated.Experimental Results:• In the intact control group, the average number of AFCs in the spleen was (7378 ± 503).• In the immunodeficient (CCl₄-only) control group, the number dropped significantly to (1032 ± 203), a 6.1-fold decrease (p < 0.05), confirming the severity of the induced immunodeficiency.• In the group treated with Timalin, the AFC count was (5241 ± 637), reflecting a 5.0-fold increase.• In the group receiving 0.1% Chlorella extract, the AFC count was (1303 ± 385), a 1.1-fold increase.• The 0.5% Chlorella group showed an AFC count of (6150 ± 687), representing a 4.5-fold increase (p < 0.05).• The 1.0% Chlorella group demonstrated the highest immune response, with an AFC count of (8052 ± 673), a 6.1-fold increase (see Table 2).
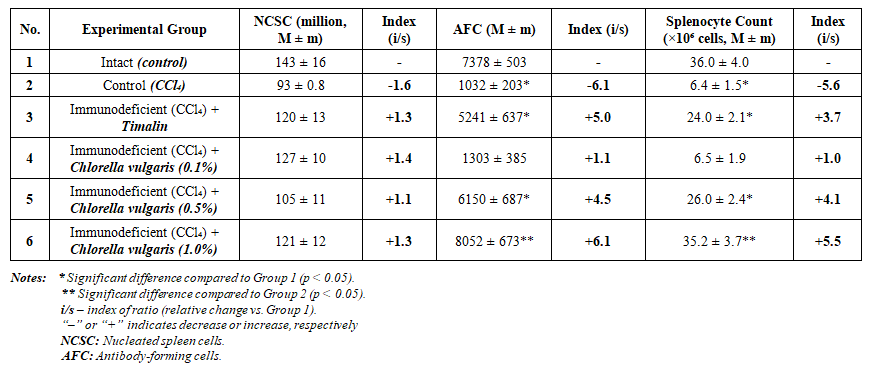 | Table 2. Effects of Chlorella-Based Preparations on Immune and Hematopoietic Parameters |
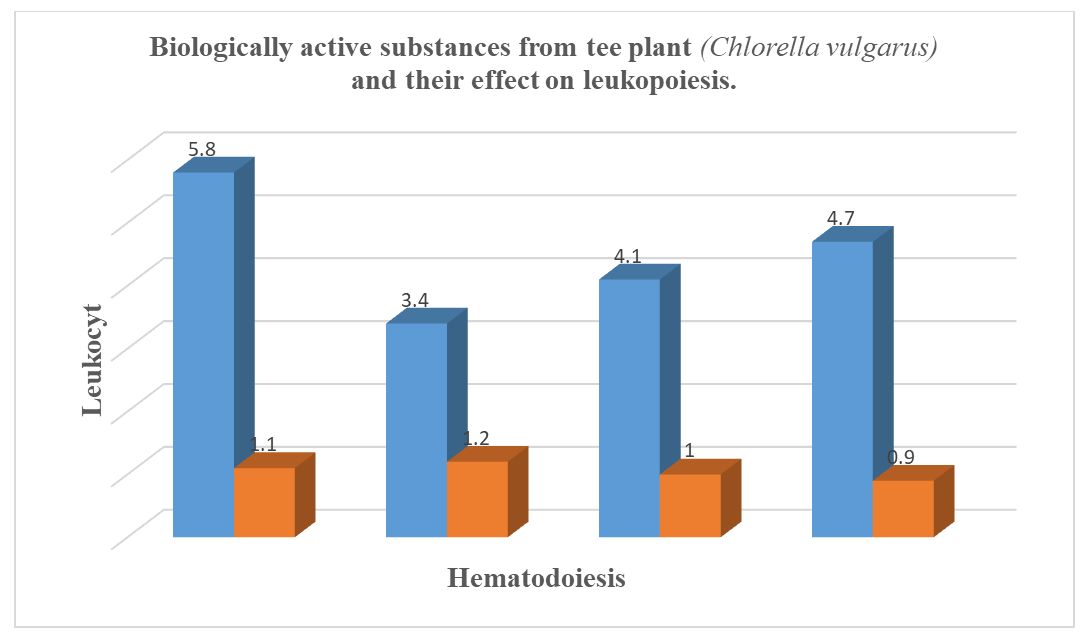 | Figure 2. Further Investigation of the Effects of Chlorella-Based Preparations on Erythropoiesis and Leukopoiesis |
 | Figure 3 |
4. Conclusions
- The results of this study demonstrate that natural medicinal bioactive substances derived from Chlorella vulgaris exhibit immunostimulatory properties and can contribute to the restoration of leukocyte counts in immunodeficient states. The evidence suggests that these compounds support immune system recovery and hematopoiesis, making them a promising candidate for therapeutic applications in veterinary medicine and possibly beyond.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML