-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Metallurgical Engineering
p-ISSN: 2167-700X e-ISSN: 2167-7018
2012; 1(6): 126-129
doi: 10.5923/j.ijmee.20120106.06
Ageing Characteristics of Sand Cast Al-Mg-Si (6063) Alloy
Aramide Fatai Olufemi
Metallurgical and Materials Engineering Department, Federal University of Technology, Akure, Ondo State, Nigeria
Correspondence to: Aramide Fatai Olufemi, Metallurgical and Materials Engineering Department, Federal University of Technology, Akure, Ondo State, Nigeria.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
The effect of variation of ageing temperature and soaking time at the ageing temperature mechanical properties of aluminium 6063 alloy was investigated. Various samples of the alloy in the as-cast condition were produced, homogenized at 560℃ held at the temperature for 2hrs, and cooled in the furnace. They were solution treated at 440℃ for 10minutes and quench in water, aged at/soaked for 125℃/3Hrs, 125℃/4Hrs, 125℃/5Hrs, 175℃/3Hrs, 175℃/4Hrs, 175℃/5Hrs, 200℃/3Hrs, 200℃/4Hrs, 200℃/5Hrs, and 225℃/3Hrs, 225℃/4Hrs and 225℃/5Hrs. Tensile and impact test were carried out on the alloy standard samples. It was observed that the Optimum tensile strength is observed in the sample aged at 175℃ held for 5hrs, optimum ductility is observed in sample aged at 125℃, held for 4hrs, Optimum toughness is observed in sample aged at 225℃, held 5hrs, closely followed by that of the sample aged at the same temperature, held for 3hrs. From the economic point of view the optimum parameter for obtaining a structural material of the optimum combination of mechanical properties from the sample is ageing temperature of 225℃ and soaking time of 3hrs.
Keywords: Ageing Temperature, Soaking Time, Intermetallic Phases, Solution Treatment
Cite this paper: Aramide Fatai Olufemi, "Ageing Characteristics of Sand Cast Al-Mg-Si (6063) Alloy", International Journal of Metallurgical Engineering, Vol. 1 No. 6, 2012, pp. 126-129. doi: 10.5923/j.ijmee.20120106.06.
Article Outline
1. Introduction
- Aluminium (6063) alloy is an alloy belonging to the 6xxx series. This alloy group contains silicon and magnesium in appropriate proportions to form magnesium silicide, thus making them heat-treatable. They possess good formability and corrosion resistance, with medium strength. Though the process of age hardening of aluminium alloys that contain Mg and Si was first discovered by Alfred Wilm in 1911,[1], this discovery led to searches for other aluminium alloy systems that would age harden[2]. These Al-Mg-Si alloys are the most widely used for the production of extruded sections. More than 80 percent of the aluminum alloys employed worldwide in the manufacturing of extruded sections belong to the 6xxx series[3]. In these alloys, Mg and Si combine to form the chemical compound Mg2Si (magnesium-silicide), the primary hardening phase[4]. Most of the added alloying elements to aluminum alloys are less soluble in the solid phase than in liquid, and then a gap between the liquidus and solidus will exist leading to the segregation of these alloying elements. This segregation is reduced by heating the ingots for an extended period close to the solidus temperature to homogenize the structure[8]. As the size of the cast product increases the chemical composition throughout the thickness will considerably vary, (macrosegregation). This type of segregation is not much affected by heating but by controlling the manufacturing parameters of the ingot. For example, in the Direct-Chill casting process decreasing the ingot thickness, lowering the casting speed and maximizing the superheating temperature will greatly reduce this sort of segregation.
2. Materials and Methods
- The materials used for this work are aluminium (6063) alloy obtained from ‘Aluminium Rolling Mill’, Otta, Ogun State, Nigeria. The chemical composition of the aluminium alloy is given in Table 1. in the as-cast condition.
|
2.1. Tensile Testing
- The tensile tests were performed on various samples using Monsanto tensometer. The fracture load for each sample was noted as well as the diameter at the point of fracture and the final gauge length. The initial diameter and initial gauge length for each sample was noted before uniaxial load. From the generated data the ultimate tensile strength and percentage elongation of each sample was calculated.
2.2. Impact Test
- The impact tests were performed on various sample determine the impact strengths by the “V-notch method using the Honsfield Balance Impact Testing Machine. Prior to mounting on the machine, the test sample is notched to a depth of 2mm with v-shaped hand file. The notched test sample was then mounted on the impact-testing machine, which is the operated to apply a (constant) impact force on the test sample. The impact strength (the amount of impact energy the specimen absorbed before yielding) was then read off the calibrated scale on the impact testing machine.
3. Results and Discussion
- Figure 1. depicts the effect of soaking time on the ultimate tensile strength of the samples; Figure 2. shows the effect of ageing temperature on the ultimate tensile strength of the samples; Figure 3. reveals the effect of ageing temperature on the ductility of the samples; Figure 4. depicts the effect of ageing temperature on the impact strength of the samples while in Figure 5. the effect of Soaking time on the Impact strength of the samples is clearly seen.It could be seen from Figure 1. that the ultimate tensile strength (UTS) of the samples aged at 125℃, increased slightly as the soaking time increases from 3 to 4 hours, thereafter further increase in the soaking time results in sharp reduction in the UTS. For those samples aged at 175℃ and 200℃ the UTS remains constant and reduced slightly respectively with increment in the soaking time from 3 to 4 hour, further increase in the soaking time results in increase in the UTS. The UTS of those samples aged at 225℃ decreased abruptly as the soaking time increased from 3 to 4 hours and thereafter increases sharply. This could be attributed to the fact that in silicon-containing aluminum alloys the most common phases are the α-Al8Fe2Si and β-Al5FeSi phases. Depending upon the alloy composition and solidification rate, one or both phases can be present in the microstructure. The β-Al5FeSi phase has needle-/ plate-like morphology and is considered detrimental to the ductility of the alloy. The α-Al8Fe2Si phase is believed to be less harmful than the β-phase form as it has more desirable compacted Chinese script morphology. It is well known that at higher cooling rates, neutralization by the addition of trace elements such as Mn, Be, Sr can change the crystallization of the iron intermetallic to the Chinese script α-phase form which improves the mechanical properties. Also, the presence of a small amount of beryllium produces a new Be-Fe phase (Al8Fe2SiBe) having a Chinese script morphology or polygon shapes, which is different from the β-phase in the alloy samples[10, 11]. From Figure 1., sample aged at 125℃ contain some α-Al8Fe2Si phase in the matrix of the alloy when it was soaked at the ageing temperature for between 3 and 4 hours, that explained the slight increase in the UTS within this period. With further increase in the soaking time, all the α-Al8Fe2Si phase in the matrix of the alloy were transformed into a mixture of β-Al5FeSi phase and Mg2Si precipitate, which accounts for the sharp reduction in UTS with further increase in the soaking time[10]. For those samples aged at 175℃ and 200℃ whose UTS remains constant and reduced slightly respectively with increment in the soaking time from 3 to 4 hour, this is due to the presence of a mixture of β-Al5FeSi phase and Mg2Si precipitate, within the soaking period at those temperatures. Increase in the soaking time to 5 hours resulted into transformation of the β-Al5FeSi phase and Mg2Si precipitate to a mixture of a new Be-Fe phase (Al8Fe2SiBe) and Mg2Si precipitate; this accounts for the increase observed in the UTS at this condition. The UTS of those samples aged at 225℃ which decreased abruptly as the soaking time increased from 3 to 4 hours and thereafter increases sharply, can be explained by the explanation given for the behaviour of the samples aged at 200℃, but because of higher ageing temperature the amount of β-Al5FeSi phase is more which later transformed to Be-Fe phase as the soaking time is increased to 5 hours [12].
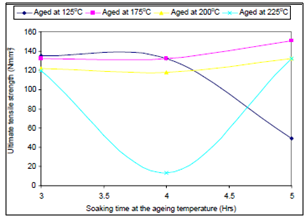 | Figure 1. Effect of soaking time on the ultimate tensile strength of the samples |
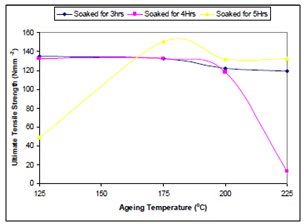 | Figure 2. Effect of ageing temperature on the ultimate tensile strength of the samples |
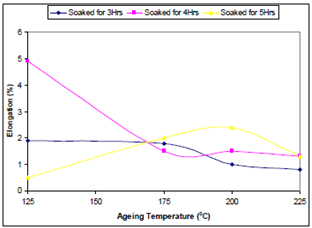 | Figure 3. Effect of ageing temperature on the ductility of the samples |
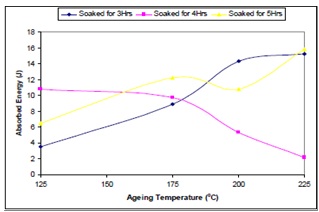 | Figure 4. Effect of ageing temperature on the impact strength of the samples |
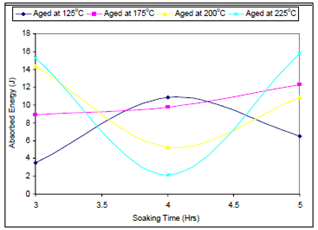 | Figure 5. Effect of Soaking time on the Impact strength of the samples |
4.Conclusions
- From the discussion thus far, it can be concluded that:● Optimum tensile strength is observed in the sample aged at 175℃ held for 5hrs.● Optimum ductility is observed in sample aged at 125℃, held for 4hrs.● Optimum toughness is observed in sample aged at 225℃, held 5hrs, closely followed by that of the sample aged at the same temperature, held for 3hrs.● From the economic point of view the optimum parameter for obtaining a structural material of the optimum combination of mechanical properties from the sample is ageing temperature of 225℃ and soaking time of 3hrs.
ACKNOWLEDGEMENTS
- The authors would like to acknowledge the assistance rendered by Mr Alo F.I., Mr Aminu, Mr Omolayo, Mr Olaoye and Mr Solanke, (all of the Department of Materials Science and Engineering, O.A.U., Ile-Ife) during the bench work of this research.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-Text HTML
Full-Text HTML