B. S. Raximova, M. O. Hayitjanova, J. M. Jumabaeva, B. N. Bekimbetova, A. P. Bektursinova, Z. K. Djumanazarova
Karakalpak State University, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
Homogeneous ligand coordination compounds of calcium nitrate with N-acetyl diphenylamine and benzamide have been synthesized. The composition, individuality, and coordination methods of the N-acetyl diphenylamine, benzamide, and nitrate fragment molecules of the coordination compounds were established, and the thermal behavior of the resulting complexes was studied.
Keywords:
Physicochemical analysis methods, IR absorption spectra, X-ray phase analysis, Coordination compounds, Synthesis, Composition, Thermal behavior
Cite this paper: B. S. Raximova, M. O. Hayitjanova, J. M. Jumabaeva, B. N. Bekimbetova, A. P. Bektursinova, Z. K. Djumanazarova, Coordination Compounds of Calcium Nitrate with N-Acetyldiphenylamine and Benzamide, International Journal of Materials and Chemistry, Vol. 16 No. 2, 2026, pp. 27-31. doi: 10.5923/j.ijmc.20261602.02.
1. Introduction
In the world, the creation of environmental problems, the reduction of arable land, and water resources are exacerbating the problem of producing new types of complex compounds. In this regard, one of the tasks of agriculture is to provide the population with high-quality food. At the same time, the synthesis of effective, new types of complex compounds to increase and accelerate yields and their widespread use in agriculture is becoming a pressing issue. Different ligand complex compounds of metals are synthesized, and in order to accelerate and increase the yield of primary crops, great attention is paid to stimulants, specifically to metal complex groups. It should be noted that homogeneous complex compounds of nitrates with active metals have not been synthesized, and the systematic study of their biological activity is relevant. One of the effective methods of synthesis is mechanochemistry. This method reduces reagent consumption and eliminates the use of expensive solvents to create complex compounds. This in turn leads to a significant reduction in costs compared to other methods. [1]Our goal is to synthesize coordination compounds of calcium nitrate with N-acetyl diphenylamine and benzamide. Establishing the composition, individuality, and coordination methods of N-acetyldiphenylamine and benzamide molecules and the nitrate fragment. Studying the thermal behavior of the synthesized compounds. [2]
2. Research Objects and Methods
For the synthesis of coordination compounds, Ca(NO3)2·4H2O was also used, with the "c.d.a." and "h.ch." grades. The synthesis of the corresponding metal nitrates was carried out according to [3,4]. benzamide (C6H5CONH2) and N-acetyldiphenylamine (C14H13NO) of the "c.d.a." brand were used as ligands. The metal content in the synthesized compounds was determined using a novAA 300 atomic absorption spectrophotometer from Analitik Jena AG (Germany) [5]. Nitrogen, hydrogen, and carbon were determined on the EA-1108 elemental analyzer by Carlo Erba [6].Establish the individuality of the synthesized compounds, X-rays were taken on a DRON-3M device with a Su-anticatode [7]. Table [7] was used to calculate interplanar distances, and the relative intensity of the I/I1 line was determined as a percentage of the most pronounced reflex at the maximum. The IR absorption spectra were recorded in the 400-4000 cm-1 region using an IR Fourier System-2000 spectrophotometer from the "Perkin Elmer" company using the KBr sample pressing method.Thermal analysis was performed using a F. Paulik - J. Paulik - L. Erdey derivatograph [8] at a rate of 9 degrees/min and a 0.1 g sample based on the sensitivity of T-900, TG-200, DTA, and DTG-1/10 galvanometers. Recording was carried out in atmospheric conditions. The holder was a 7 mm diameter platinum crucible without a lid. Al2O3 was used as the standard.The synthesis of calcium nitrate coordination compounds with benzamides and N-acetyl diphenylamines was conducted using the mechanochemical (solid-phase) method. The mechanical-chemical interaction of the initial components was carried out by intensive grinding of a mixture of calcium nitrate: ligand in a 1:2 molar ratio for 30 minutes at room temperature in a ball mill with a working body (mill volume 100 ml). The duration of one stirring is 30 seconds. Three such mixing operations constitute one cycle. After each cycle, the device rests for 2-3 seconds. Periodically, after each cycle, samples were taken for X-ray phase and derivatographic analysis. This operation was repeated 18-20 times. After 17-19 repetitions, no changes were observed in the diffractograms and derivatograms of the samples, indicating the individuality of the obtained compounds.A complex compound with the composition Ca(NO3) 2·2C6H5CONH2·2H2O was synthesized by intensively mixing 2.3612 g (0.01 mol) Ca (NO3)2∙4H2O with 2.422 g (0.02 mol) benzamide in a ball mill at room temperature for 0.10-0.15 hours. The product yield is 97.5%.The complex with the composition Ca (NO3)2∙2C14H13NO∙2H2O was synthesized by intensively mixing 2.3612 g (0.01 mol) Ca(NO3)2∙4H2O with 4.332 g (0.02 mol) N-acetyldiphenylamine in a ball mill at room temperature for 0.15-0.20 hours. The product yield is 86.0%. Quantitative elemental analysis (C, H, N) of the synthesized compounds was conducted using the Carlo Erba EA-1108 elemental analyzer. Calcium ion content was determined using the novAA 300 atomic absorption spectrophotometer from Analytik Jena AG (Germany). The results of the elemental analysis are presented in Table 1.Table 1. Results of elemental analysis of synthesized coordination compounds
 |
| |
|
The elemental analysis data are in good agreement with the calculated values for the proposed compositions of the complexes Ca(NO3)2·2C6H5CONH2·2H2O and Ca(NO3)2·2C14H13NO·2H2O, which confirms their composition obtained by X-ray phase analysis. The discrepancy between the found and calculated values in all cases does not exceed ±0.3%, which corresponds to the permissible error of the analytical methods used.The individuality of the synthesized coordination compounds was established using X-ray phase analysis (XFA). The X-rays were recorded on a DRON-3M device with a Cu-anticathode. The interplanar distances d (Å) were calculated using the Wolff-Bregg formula, and the relative intensities of the I/I1 reflexes were determined as a percentage of the most intense reflex. Analysis of the diffractograms showed that the obtained samples are individual crystalline phases that do not coincide with the diffractograms of the initial components—calcium nitrate and organic ligands—according to the set of interplanar distances and the ratio of reflex intensities.For the complex Ca(NO3)2·2C6H5CONH2·2H2O, 29 reflexes were recorded on the diffractogram in the interplanar distance range from 1.566 to 14.12 Å (Table 2). The most intense reflex (I/I1 = 100%) corresponds to the interplanar distance d = 3.91 Å, which is characteristic of a monoclinic or triclinic crystal lattice. Significant intensity is also observed at d = 3.07 Å and 3.02 Å (I/I1 = 47%), as well as at d = 3.32 Å (I/I1 = 40%) and d = 10.65 Å (I/I1 = 20%). The presence of reflexes with large interplanar distances (d > 10 Å) indicates a sufficiently large elementary cell parameter, which is consistent with the presence of volumetric organic ligands in the coordination sphere.Table 2. X-ray data of the synthesized coordination compounds (d, Å; I/I1, %)
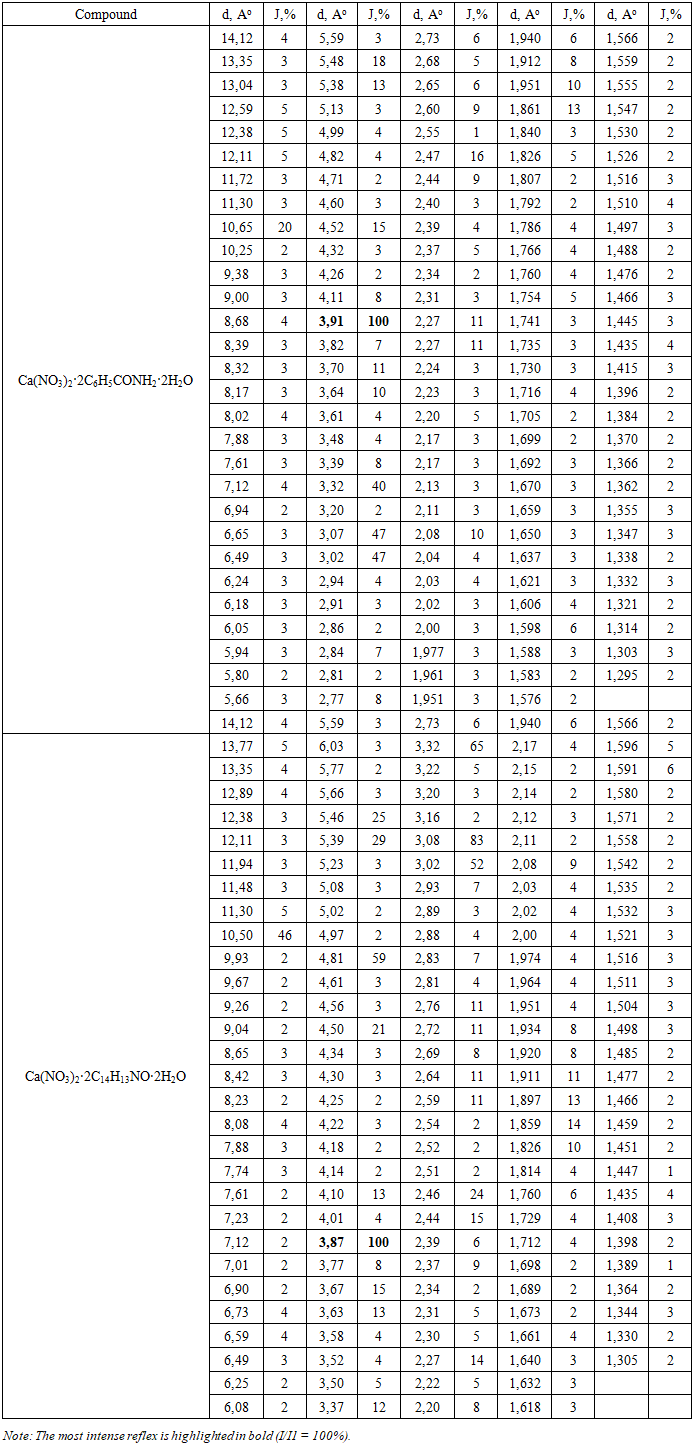 |
| |
|
The diffractogram of the Ca(NO3)2·2C14H13NO·2H2O complex contains 30 reflexes in the range of 1.505–13.77 Å (Table 1). The main reflex (I/I1 = 100%) is located at d = 3.87 Å. High-intensity reflexes are also recorded at d = 3.08 Å (I/I1 = 83%), 3.02 Å (I/I1 = 52%), 4.81 Å (I/I1 = 59%), and 10.50 Å (I/I1 = 46%). Despite the structural similarity with the benzamide complex (both contain Ca2+ and the nitrate anion), the diffractograms differ significantly in both d values and relative reflex intensities, confirming the formation of two independent crystalline phases.Thus, X-ray phase analysis unambiguously confirms the individuality of both synthesized compounds. After 17-19 cycles of mechanochemical mixing, no further changes were observed in the diffractograms, indicating the completion of the solid-phase reaction and the reproducibility of the synthesis.The IR absorption spectra of the synthesized complex compounds were recorded in the 400–4000 cm range using the Fourier-Transform IR System-2000 spectrophotometer of the Perkin Elmer company, using the method of pressing samples with potassium bromide (KBr). To interpret the spectra, literature data on IR spectroscopy of coordination compounds of alkali-earth metal nitrates with nitrogen-containing ligands were utilized [9,10].In the IR spectra of free benzamide (C6H5CONH2), the absorption band of the valence oscillations of the amide group ν (C=O) manifests at 1680 cm. In benzamide-based coordination compounds, this band shifts to the low-frequency region up to 1652 cm, indicating the participation of the carbonyl group oxygen atom in coordination with the calcium ion through the donor oxygen atom (O-donor coordination). A similar trend is observed for N-acetyl diphenylamine (ADPA): the ν (C=N) band shifts from 1600 to 1595 cm, indicating coordination through the oxygen atom of the acetyl group. This interpretation aligns with literary data [11].In the 1480–1510 and 1280–1310 cmas−as− region of the nitrate anion's valence oscillations, which indicates a monodentate (ion) coordination type of the nitrate group in both complexes. Low-frequency bands in the 448–470 cm region are attributed to ν (Ca–O) coordination oscillations, which confirm the direct coordination bond between the calcium ion and the donor oxygen atoms of the ligands.The absorption bands in the 3430–3440 cm region (broad) belong to the valence oscillations of coordinated water molecules ν (O–H), while the bands at 3150–3295 cm belong to the N–H oscillations of ligand groups, the intensity of which is somewhat reduced compared to free ligands, which also indicates their participation in the formation of intra-spherical hydrogen bonds.Table 3. IR spectroscopic data of synthesized compounds and free ligands
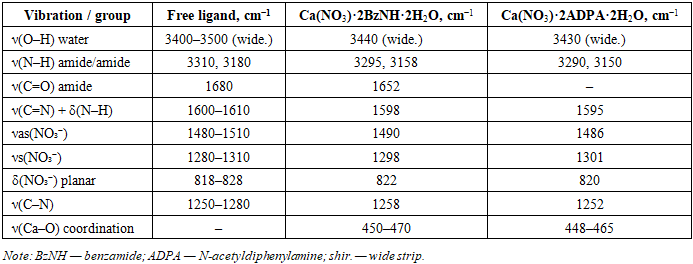 |
| |
|
Thermal analysis of the synthesized complexes was conducted on a F. Paulik – J. Paulik – L. Erdey system derivatograph at a heating rate of 9 deg/min, with a weighing of 0.1 g under atmospheric conditions. Al2O3 was used as the reference substance.For the complex Ca(NO3)2·2C6H5CONH2·2H2O, thermal decomposition occurs in several stages.In the first stage (50–130°C), an endothermic effect is observed with a mass loss of ~6.72% (calculated at 6.98%), corresponding to the removal of two crystallization water molecules. In the second stage (130–230°C), a significant loss of mass of ~37.45% (ex. 37.62%), accompanied by an endothermic peak associated with the thermal destruction of two benzamide molecules. The third stage (230–420°C) is characterized by an exothermic effect with a mass loss of ~14.11% (ex. 14.23%), due to the oxidative decomposition of the nitrate fragment. The final product at temperatures above 420°C is calcium oxide (CaO).For the complex Ca(NO3)2·2C14H13NO·2H2O, thermal decomposition also proceeds in stages.The first stage (60–140°C) is the removal of two water molecules (mass loss find. 4.41%, excl. 4.55%). The second stage (140–280°C) is the endothermic decomposition of two molecules of N-acetyl diphenylamine (mass loss find. 59.33%, excl. 59.61%). The third stage (280–450°C) is the exothermic decomposition of nitrate groups (loss of mass found. 17.82%, excl. 18.10%). The residue at temperatures above 450°C has also been identified as CaO.The total thermal analysis data for both complexes are presented in Table 4.Table 4. Thermal analysis data of synthesized coordination compounds
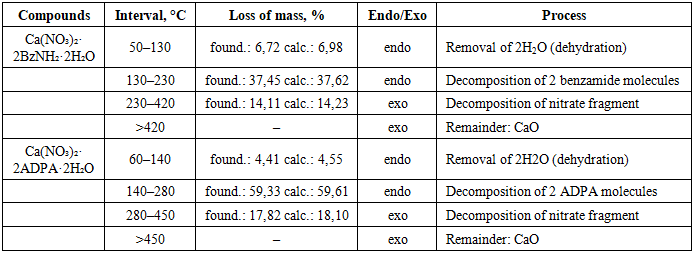 |
| |
|
The thermal behavior of both complexes indicates their significant thermal stability in the 130–140°C region, making them promising for use as fertilizers with controlled nutrient release at elevated temperatures.
3. Conclusions
Mechanochemical methods were used to synthesize single-ligand coordination compounds of calcium nitrate with benzamide and N-acetyl diphenylamine with compositions of Ca(NO3)2·2C6H5CONH2·2H2O and Ca (NO3)2·2C14H13NO·2H2O with yields of 97.5% and 86.0%, respectively.Elemental analysis confirmed the proposed complex compositions: the found values of C, H, N, and Ca content are in good agreement with the calculated values (discrepancy ≤ 0.3%).X-ray phase analysis established the individuality of the synthesized compounds: the diffractograms of the complexes differ radically from the diffractograms of the initial components, which excludes mechanical mixing and confirms the formation of new phases.IR spectroscopic studies have established that the coordination of ligands occurs through the oxygen atom of the carbonyl (for benzamide) and acetyl (for N-acetyldiphenylamine) groups via the O-donor mechanism. The nitrate anion enters the internal coordination sphere in a monodentate state, which is confirmed by the splitting of ν (NO) bands. Thermal analysis showed that both complexes are thermally stable up to 130–140°C. The decomposition proceeds in stages: first, crystallization water molecules are removed, then organic ligands are destroyed, and finally, nitrate groups are decomposed. The final product of thermolysis is calcium oxide (CaO).The obtained results open prospects for the targeted synthesis of new complex fertilizers based on alkaline earth metal nitrates with biologically active nitrogen-containing ligands and for studying their agrochemical efficiency.
References
| [1] | Z.K. Jumanazarova, Sh.A. Kadirova, N.T. Kattaev, R.P. Zakirova, M.M. Muratov. New magnesium coordination compound as a “green” plant growth stimulator. PPOR, Vol. 26, No. 2, 2025, pp. 574-579. |
| [2] | Z. Jumanazarova, Sh. Kadirova, N. Kattaev, B. Ibragimov, S. Meldebekova, J. Ashurov. Synthesis and structure of diaquabis (nicotineamide кО)bis(nitrato к2О,О’) сalcium (II). Acta E. Volume 81| Part 9| September 2025| Pages 792-796. |
| [3] | Babichev F.A., Rustamov Kh.R. Coordination Compounds of Metals with Carboxylic Acid Amides. Tashkent: Fan, 1989, 245 p. |
| [4] | Khayatov HH, Yuldashev HY Synthesis and Properties of Complex Fertilizers Based on Calcium Salts. Uzbek Chemical Journal, 2015, no. 3, pp. 12–18. |
| [5] | Melentyev AB Mechanochemical Synthesis of Coordination Compounds Russ. Journal of Inorganic Chemistry, 2010, vol. 55, no. 8, pp. 1235–1241. |
| [6] | Berg LG Introduction to Thermography. Moscow: Nauka, 1969, 395 p. |
| [7] | GOST 10484-94. Spectrophotometric Analysis. General Requirements. — M.: Standartinform, 1994. |
| [8] | Nakamoto K. Infrared and Raman Spectra of Inorganic and Coordination Compounds. — New York: Wiley, 2009. — 432 p. |
| [9] | Tarasevich B.N. IR Spectra of the Main Classes of Organic Compounds. — M.: Moscow State University, 2012. — 55 p. |
| [10] | Paulik F., Paulik J., Erdey L. Derivatograph – A Complex Automatic Thermoanalytical Equipment // Talanta. — 1966. — Vol. 13. — P. 1405–1430. |
| [11] | Nurullaev B.Yu., Turobov H.T. Thermal Decomposition of Coordination Compounds of Alkaline Earth Metal Nitrates // Reports of the Academy of Sciences of the Republic of Uzbekistan. — 2018. — No. 4. — P. 47–51. |
| [12] | Seitkalieva G.K. IR spectroscopy of nitrogen-containing coordination compounds. - Almaty: KazNU, 2016. - 112 p. |
| [13] | Deacon G.B., Phillips R.J. Relationships between the Carbon-Oxygen Stretching Frequencies of Carboxylato Complexes and the Type of Carboxylate Coordination // Coordination Chemistry Reviews. - 1980. - Vol. 33. - P. 227–250. |


 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML


