Nafisa Husanova1, Madina Khamidova1, Aslbek Yulchiev2, Qamar Serkayev1, Shoirakhon Isroilova1, Saida Gulimova1
1Tashkent Institute of Chemical Technology, Tashkent, Uzbekistan
2Andijan State University, Andijan, Uzbekistan
Correspondence to: Nafisa Husanova, Tashkent Institute of Chemical Technology, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
In the fat processing industry, one of the main sources of fatty acids is soapstock, obtained as a result of alkaline refining of vegetable oils. The aim of the study was to analyze the effect of concomitant substances of cotton and sunflower coapstock on the process of obtaining fatty acids, as well as to determine the content of concomitant substances in coapstock obtained by various refining methods. It was found that the emulsion refining of cottonseed and sunflower oils produces up to 16.5-24.8% of the oil content relative to the oil weight, and its fat content is 45.2-48.6% (7.2% of the oil weight). When processing these oils with monoethanolamine (MEA) and subsequent refining, the fat content in the soapstock increases to 52.7-57.2%, and the content of gossypol and phospholipids decreases to 1.7 and 2.9%, due to their preliminary separation. This composition allows the effective use of coapstocks obtained from IEA-treated oils in the production of fatty acids.
Keywords:
Fatty acids, Molecular weight, Salomas, Hydrolysis, Saponification, Glitserin, Soap
Cite this paper: Nafisa Husanova, Madina Khamidova, Aslbek Yulchiev, Qamar Serkayev, Shoirakhon Isroilova, Saida Gulimova, Change in the Physico-Chemical Properties of Soapstock and the Fatty Acids Obtained from It During a Three-Stage Crushing Process, International Journal of Materials and Chemistry, Vol. 16 No. 2, 2026, pp. 15-26. doi: 10.5923/j.ijmc.20261602.01.
1. Introduction
In our previous studies, we have developed a technology for the separation of raw fatty acids by three-stage processing of cotton and sunflower soapstocks. According to it, a 50/50 ratio mixture of cotton and sunflower soapstocks was formed and soapy for 60 min with an aqueous solution of 35% of calcined soda at a temperature of 90-95℃ in the first stage. In carbonated saponification (in the presence of calcined soda), the triglycerides of soapstock are not completely saponified, and the saponification degree reaches 80%. The soapy mass was washed at this temperature at a rate of up to 50% compared to the soapy mass with a 20% concentrated solution of table salt (NaCl) with electrolyte properties, along with the intermediate product formed in the process - sour soaps, and the aqueous phase was infused for 1-4 hours. During the clarification process, the salt electrolyte solution removes colorants, phospholipids, fatty acid degradation products, and various water-soluble compounds of gossypol from the soap into the salt water phase. After clarification, the aqueous phase contains glycerin, accompanying substances, calcined soda, carbon dioxide, and table salt, and is removed from the bottom of the reactor. In the second stage, the primary soapy mass is completely soapy with a 42% solution of caustic soda at a temperature of 90-95°C for 180 min, and then washed with a 20% concentration solution of table salt at 50% relative to the soap mass and poured into an aqueous phase tincture. After standing for 4 hours and the precipitate was settled, the alkaline soap solution was separated and transferred to an acidic decomposition vessel. The resulting alkaline soap water is sent to separate glycerin or neutralize the water from acid decomposition, along with the water from the first stage. The yield of fatty acids obtained by our developed and industrial methods of processing soapstocks was 97.0% and 90.9%, respectively. The oil phase obtained at the same time contains 2.9% and 5.7% additives, respectively, for the samples. In our further studies, we studied the effect of the amount and concentration of the sodium chloride solution used in the final saponification and acid hydrolysis processes on the physicochemical properties of the resulting oil and aqueous phases.
2. Literature Review
In addition to neutralizing free fatty acids in the process of alkaline refining of vegetable oils to form soapy salts, at the expense of the surface activity of most companion substances and soapstok, which dissolve in the oil, most of the insoluble and non-soapy substances, depending on the adsorbing property, also part of the neutral oil, are brought into the sediment along with In particular, during the refining of cottonseed oil, unlike other soapstocks, a mass is separated that contains a high level of non-oil substances, is dark in color, and contains gossypol and its derivatives [1]. Light-colored soapstocks (soy, kungbaoqar, maxsar, raps, and b.) when used in soap production, fatty acids are extracted from cotton soasptokg [2]. The color of soapstocks depends on the storage of the coloring matter and other accompanying substances of the refined oil and can vary over a wide range. In any case, the soapstok ingredient can be divided into two groups: neutral oils in the form of fat - natural substances - mono -, di-and triglycerides, salts of fatty acids, free fatty acids, waxy substances; non-fatty substances-organic acids soluble in water and their salts, pigments, metallic compounds, hydrocarbons, alcohols, ketones and a number of other compounds. The diverse and complex nature of the substances contained in Soapstok causes its rapid variability in the process of its storage and processing. In its composition, fats, phospholipids are partially hydrolyzed during the storage process. Phospholipids are also degraded by acids [3]. The presence of polar and nonpolar hydrocarbon radicals in phospholipids gives them strong emulsifying properties [1]. During the soapstock decomposition process, they can get stuck in the emulsion layer between the oil and water phases, slowing down the process. Oxidized fatty acids (oxacids) are formed when unsaturated fatty acids react with oxygen. Oxidizing acids can increase by 1-10% during the soapstock degradation process [3]. Oxacids also exhibit high hydrophilicity by reacting with sulfuric acid, such as the soapy salts of glycerophosphoric acid, and reduce the electrolyte effect on the soap mass. Their presence in fatty acids reduces the quality of the product and darkens the color of fatty acid as a result of their synergistic effect with oxygen in the atmosphere [4].Volatile valence metals are involved in fatty acids as active catalysts for the oxidation process [5]. The above negative effects reduce the quality of fatty acids and worsen their color. In the process of alkaline finished soapstock soapy and acid decomposition, as well as inversely affecting the course of the high-temperature distillation process, not only reduces the amount and quality of product output, but also negatively affects the intensity of processes, which leads to an increase in process duration, an increase in energy consumption and an increase in waste. Numerous studies have been conducted and various solutions have been achieved to reduce negative impacts, increase product yield and quality, and achieve energy efficiency. In particular, in order to break down the derivatives of phospholipids and gossipol, the process of complete soapstock soapy can be carried out at high temperature and pressure [6]. As a result, not only glycerides, but also substances such as phospholipids and gossypol are saponified, and the formation of emulsions is reduced during subsequent acid hydrolysis. Gypsum can be separated in the process of neutralizing alkaline water with sour water by carrying out the complete soapy process with the participation of Ca(OH)2 in order to completely dispose of the products of the decomposition process. Many methods of detoxifying gossipol involve converting it to a hydrophilic state during the refining process. However, compounds formed under hydrophilic conditions precipitate with soapstock and enter the soapstock composition, complicating the processing processes. Effective removal of gossypol and phospholipids by refining cottonseed oil before refining can improve the quality of the soapstock after the refining process [7]. The "Sour Soaps" reaction can be used to effectively remove water-soluble impurities from the soap mass. This ensures that the fatty acids are not completely saponified in the reaction medium, but rather form associates, and that the fatty acids are subsequently released cleanly by washing with water. A phased acid decomposition method has been proposed by forming sour soaps of the finished soapy mass. In this case, sour soaps are salted using sodium sulfate, which is formed in the process of acid decomposition. The first stage is carried out in conditions where the pH environment is 4-5. As a result of salting, water-soluble acids (VRKS) and their salts are formed, and they are extracted by clarification. Complete decomposition is carried out in an environment with a pH of 1.5-3.0. Timely removal of VRKs leads to acceleration of the process and a reduction in sulfuric acid consumption by up to 4%. A method has been developed for soapstock decomposition that involves heating it, adding electrolyte and demulsifier while stirring, separating the oily phase, and settling [8]. Heating soapstok to 50-60°C haroart, sodium chloride in the amount of 2-6% is added to it, PAV OP-7 as a deimulgator in the amount of 0.1-0.7%, and 20-40% from isoamyl and isobutyl alcohol, taken in an additional ratio of 2.5:1, compared to the mass of soapstok and mixed 3-5 min. As a result, the separation of the oil emulsion is accelerated, the release of the oil component increases, and the process is accelerated. As can be seen from the analysis, oxidation processes are of great importance during the separation of fatty acids from soapstock with a complex composition, and the decomposition of phospholipids, gossypol, and fatty acids is observed. In the process, the formation of oxycislotes, phospholipids and non-soapy substances have an emulsifying effect in the reacting mixture, reducing the intensity of the process by preventing the separation of the soapy oil and aqueous phase [9]. Despite the fact that certain technologies have been developed to accelerate processes in obtaining fatty acids from soapstock and reduce product output, as a result of the lack of technologies based on a consistent approach, which fully involves the processes of complete soapstock soapy and acid decomposition, the output of fatty acids from soapstock is low, energy consumption is high as a result of In today's era of increasing energy costs, it is important to improve system efficiency and reduce energy consumption.
3. Methods and Materials
The fatty acid composition of soapstock was determined by gas-liquid chromatography. Devices: SHIMADZU Nexis GC - 2030 Gas Chromatography Electronic scale PTXFA210 with accuracy of ± 0.0001g Pipette OMSONS Class A Accuracy ± 0.01ml Mixer MXS Speed - 0 - 2500 R/min Test tube OMSONS A class ± 0.25ml accuracy Thermostat TC-1/80 SPU ± 0.1°C accuracy Reagents: Hexane hc - purity, Sodium methylate - 2 mol/dm3 concentration Sample preparation process. The sample was placed in a glass beaker, heated to 50°C in a thermostat, and passed through a paper filter while stirring. The filtered sample was weighed into a test tube with an accuracy of 0.1 g ± 0.005 and 3 ml of hexane was added to the sample and mixed for 2 min in a mixer. Over the sample being prepared, 0.2 ml of sodium methylate was added and mixed again in a mixer for 2 min. Then it was taken from the top of the sample and a chromatographic analysis was carried out. Chromotography conditions. Detector - flame ionization detector (FID) Mobile phase-helium, Mobile Phase Flow - 2 ml/min, Split - 1: 20, sample sending amount - 1 MCL, injector temperature - 220°C, detector temperature - 250°C, thermostat temperature - 1 )100°C - 4 min cost., 2) It rises from 25°C to 200°C in 8 minutes. 3) It rises from 5°C to 250°C per minute and stays at 6 minutes. Column - Silica Capillary 100meter* 0.25 mm ID* 0.2 mkm. Determination of the color index of fatty acids. The color index was determined on a Lovibond device according to ISO 15305:1998. This method is based on determining the color of oil in a layer of oil or a mixture of fatty acids, i.e. 133.35 mm for light oils and 25.4 mm for dark oils. This method is used to determine the color of refined and unrefined cottonseed oil. With this device, the color determination is very simple, the volume of the sampled cuvette (25.4 or 133.35 mm) is entered into the equipment program, The "read" button is pressed, and in 15-20 seconds the device screen displays 3 indicators of oil by color (red, yellow, blue unit). This method is based on comparing the color of the oil in a specific layer thickness with the color of the glass filters. Lovibond PFX 880. The instrument has lower and upper windows. The upper window is designed to illuminate the colored filters with a beam of light. A cuvette filled with oil is placed in front of the lower window. The light beam is directed to the eyepiece by the upper and lower windows using flat mirrors and prisms. In this case, both halves of the field of view should be equally illuminated. This is achieved by properly positioning the device in front of the light source. The oil color is expressed in red units on a scale with a cuvette thickness and a yellow filter number or sum of numbers. For colorfulness, the average arithmetic value of 3-5 measurements is assumed. The difference between the two parallel dimensions should not exceed 1 unit in the interval of 2-18 units. Determination of acid number by the salt method. The acid number of used soapstocks and fatty acids was determined according to ISO 660:2020 [1]. Equipment, reagents and Materials: Laboratory scales of the 4th grade; A special glass container with a cylindrical volume of 100 or 200 cm3; a Burette with a volume of 25 cm3 with a division of 0.1 cm3; an aqueous or alcohol solution of potassium hydroxide of 0.25 n or 0.1 n; a 35-36% solution of sodium chloride; phenolphthalein. 20 g of soapstock is placed in a 250 ml conical flask with a stopper. 50-60 ml of 15% neutral sodium chloride solution and 0.5 ml of phenolphthalein are poured onto it.The flask is capped with a stopper and shaken, then titrated with 0.1 N con solution (0.25 N con solution can be used for high acid numbers). During titration, the lower layer of the liquid is shaken until the color disappears after adding 4-5 drops of alkali each time. If the color begins to fade gradually after the next shake, the tube is shaken after each 1-2 drops of alkali is added. The titration is continued until a clear pink color appears in the bottom layer of the liquid. The acid number is determined by the following formula (1): 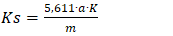 | (1) |
Here: 5.611 - a coefficient expressed as mg KOH corresponding to a 0.1 N solution of caustic alkali;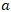 - the volume of 0.1 N solution used for titration, ml;m - the mass of the sample under analysis, g;K - correction factor for the titer.In determining the amount of organic acid salts in soapstock and fatty acids, compounds present in the soapstock that react with sulfuric acid were taken into account. These include low-molecular-weight and oxidized acidic impurities, amino compounds, glycerophosphoric acid, gossypol derivatives, and other saponified substances.The analyses were carried out by potentiometric titration in an alcoholic medium. For the study, a soapstock sample weighing 0.5-1.0 g was taken and dissolved in alcohol. A 0.1 mol/dm³ alcoholic solution of hydrochloric acid was used as the titrant.Based on the titration results, potentiometric titration curves were constructed to determine the equivalent volume of the titrant. The amount of organic acid salts was calculated using the following formula:
- the volume of 0.1 N solution used for titration, ml;m - the mass of the sample under analysis, g;K - correction factor for the titer.In determining the amount of organic acid salts in soapstock and fatty acids, compounds present in the soapstock that react with sulfuric acid were taken into account. These include low-molecular-weight and oxidized acidic impurities, amino compounds, glycerophosphoric acid, gossypol derivatives, and other saponified substances.The analyses were carried out by potentiometric titration in an alcoholic medium. For the study, a soapstock sample weighing 0.5-1.0 g was taken and dissolved in alcohol. A 0.1 mol/dm³ alcoholic solution of hydrochloric acid was used as the titrant.Based on the titration results, potentiometric titration curves were constructed to determine the equivalent volume of the titrant. The amount of organic acid salts was calculated using the following formula: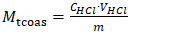 | (2) |
M₍tcoas₎ - total content of organic acid salts, mol·eq/kg;C₍HCl₎ - concentration of the alcoholic HCl solution, mol/dm³;V₍HCl₎ - volume of HCl solution corresponding to the equivalence point, cm³;M - mass of the sample (weighed portion), g.The determination of soap and accompanying salts was based on the extraction of fatty acids from fully saponified soapstock using an organic solvent [3].For this purpose, 5-10 g of fully saponified soapstock, preheated to 80-90°C, was taken and decomposed with sulfuric acid in the presence of methyl orange indicator under continuous stirring.The released fatty acids were then extracted with petroleum ether, and the precipitate was separated by settling. After removal (evaporation) of the solvent from the extract, the residue was dried and its mass was determined.The content of soaps was calculated using two formulas: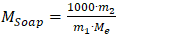 | (3) |
M₍soap₎ - soap content in the soapstock, mol·eq/kg;m₁ - mass of the sample (weighed portion), g;m₂ - mass of the dried extract, g;Mₑ - average equivalent mass of fatty acids in the soapstock, taken as 280 g/mol·eq.The content of salt impurities was determined using Formula 2.3: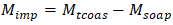 | (4) |
M₍imp₎ - content of salt impurities, mol·eq/kg of soapstock;M₍tcoas₎ - total content of organic acid salts, mol·eq/kg;M₍soap₎ - soap content in the soapstock, mol·eq/kg.In determining the content of water-soluble acids and salts of insoluble compounds, it was taken into account that, at the final stage of soapstock decomposition, water-soluble substances pass into the acidic aqueous phase.Therefore, based on methods proposed by other authors, during the determination of soap content, the separated acidic water phase was filtered and analyzed using the potentiometric method.In this case, the amounts of sulfuric acid and water-soluble acids were determined by acid-base titration according to Formula 4.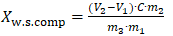 | (5) |
X₍w.s.comp₎ - content of water-soluble compounds, mol·eq/kg;V₁ - volume of alcoholic KOH solution consumed for titration of sulfuric acid corresponding to the first peak, cm³;V₂ - volume of alcoholic KOH solution consumed for titration of sulfuric acid and organic acids corresponding to the second peak, cm³;C - concentration of the alcoholic KOH solution, mol/dm³;m₁ - mass of decomposed soapstock, g;m₂ - mass of the sulfate aqueous phase obtained from decomposition of m₁ amount of soapstock, g;m₃ - mass of the sulfate aqueous phase taken for potentiometric titration, g.
4. Results and Discussion
In particular, the concentration and amount of sodium chloride solution significantly affect the color of the resulting product, the content of impurities, and the consumption of reagents.The separation of the saponified mass into soap and the underlying alkaline aqueous phase is directly dependent on the concentration of the electrolyte solution. The concentration at which complete separation of the oil phase from the aqueous phase is ensured is referred to as the critical concentration (C₍cr₎) [4].This concentration has been determined for individual fatty acids. In our experiments, namely for a 50/50 mixture of cottonseed and sunflower soapstocks, the required electrolyte concentration for washing the saponified mass with saline water was determined by calculation.For this purpose, the following formula was used (6):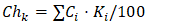 | (6) |
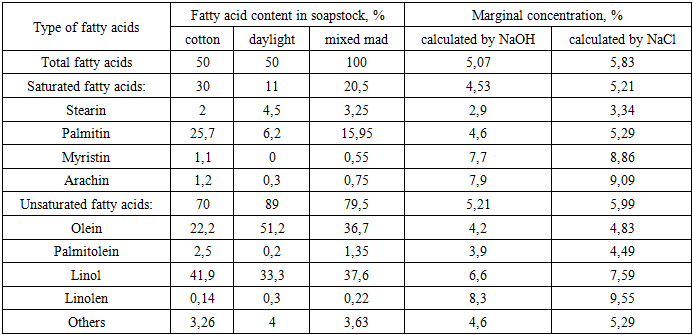
Cᵢ - the fraction (share) of a fatty acid relative to the total amount;Kᵢ - the critical concentration for an individual fatty acid.The critical concentration is calculated relative to sodium hydroxide. Therefore, when selecting different electrolytes, their required amount is determined based on their conversion coefficient relative to sodium hydroxide. For sodium chloride, the conversion coefficient is 1.15. Based on the fatty acid composition of individual soapstocks and their mixtures, as well as their corresponding critical concentrations, the required critical concentration of sodium chloride for separating the alkaline sub-soap aqueous phase after saponification of the studied mixture was calculated. The results are presented in Table 1.Table 1. Critical concentration of sodium chloride solution required for the separation of the alkaline sub-soap aqueous phase from soapstock mixtures
 |
| |
|
As can be seen from Table 1, in the 50/50 cottonseed-sunflower soapstock mixture, unsaturated fatty acids account for 79.5%, while saturated fatty acids constitute 20.5%. Accordingly, the critical concentration is 5.99% for unsaturated fatty acids and approximately 5.99% for saturated fatty acids, with an overall value of 5.83%. It is noteworthy that the critical concentration required for salting out soaps of short-chain fatty acids is significantly lower compared to that of long-chain fatty acids, indicating differences in their phase separation behavior.The first modification introduced in our study to the process of obtaining fatty acids from soapstock involves dividing the complete saponification stage into two steps. In the first stage, 50% of the calculated alkali was replaced with a 35% solution of calcined soda (sodium carbonate). After saponification at 90°C for 60 minutes, the oil and aqueous phases were separated, and water-soluble substances were removed from the oil phase by washing with a 20% sodium chloride solution used as an electrolyte.In laboratory experiments, the amount of electrolyte solution was added visually, based on the free and clear separation of the aqueous and oil phases. However, for industrial application, it is necessary to determine quantitatively the required amount of sodium chloride solution relative to the fatty acid content of the soapstock.Therefore, the concentration and amount of sodium chloride were systematically investigated. Initially, the optimal concentration of sodium chloride solution was evaluated. During the carbonate saponification of soapstock, sodium chloride solutions with concentrations ranging from 5% to 25% were prepared. At the end of the process, heating was stopped, and the system was treated with the salt solution until a clear phase separation between the oil and aqueous layers was achieved.The obtained results are presented in Figure 1.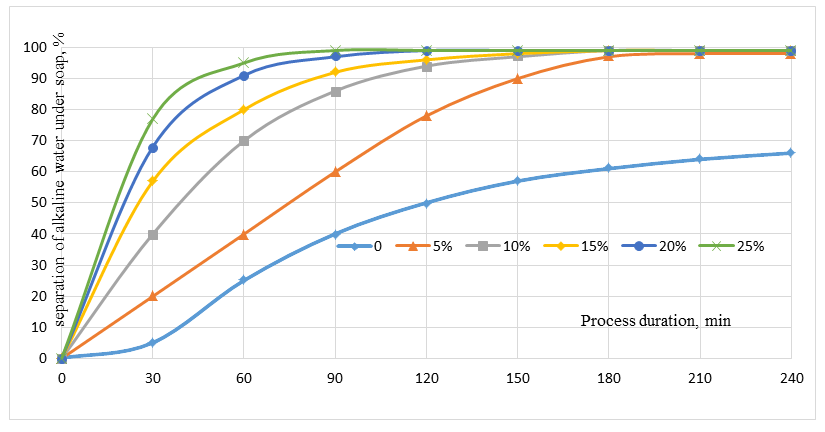 | Figure 1. Dependence of aqueous phase separation on electrolyte concentration |
As shown in Figure 1, during washing of the carbonate-saponified mass, the control sample treated with water without electrolyte exhibited low separation efficiency. Due to the formation of a stable emulsion, phase separation proceeded very slowly, and even after 240 minutes, complete separation of the oil and aqueous phases was not achieved. With an increase in the sodium chloride concentration above 5%, a significant acceleration of aqueous phase separation was observed. In particular, when treated with a 5% NaCl solution, approximately 40% of the aqueous phase separated within 60 minutes, 78% within 120 minutes, and 96% within 180 minutes. Increasing the concentration to 10% and 15% further accelerated the process, leading to 96-97% phase separation within approximately 150 minutes.Treatment with 20-25% NaCl solutions enabled complete separation of the aqueous phase within 90-120 minutes. Since the results obtained using 20% and 25% (saturated) NaCl solutions were very similar, it is considered optimal to use a 20% NaCl solution for washing the carbonate-saponified mass of cottonseed-sunflower soapstock mixtures in the first stage. Similar conclusions have also been reported by other researchers.Thus, a discrepancy exists between the theoretically calculated critical concentration and the practically optimal electrolyte concentration required for effective separation of the alkaline sub-soap aqueous phase. This difference can be attributed to the emulsifying properties of accompanying compounds present in the saponified mass.It is known that soapstock contains three main groups of impurities:1. Alkali-reactive compounds forming soaps.These include phosphatides (lecithin, cephalin, phosphatidic acid), oxidized fatty acids, and certain gossypol derivatives. During refining of cottonseed oil, most phosphorus-containing compounds transfer into the soapstock, whereas in sunflower oil, their content is lower due to prior hydration. Phosphorus-containing compounds possess both polar and nonpolar hydrocarbon groups, giving them strong emulsifying properties, which hinder phase separation. Soapstock may also contain 1-10% oxidized fatty acids (oxyacids) depending on storage conditions [4.N]. These compounds negatively affect both the color of the recovered fatty acids and phase separation efficiency.2. Non-saponifiable compounds.These include ketones, aldehydes, tocopherols, certain water-insoluble gossypol derivatives, and aliphatic alcohols.3. Pigments.Cottonseed oil contains significant amounts of gossypol, while sunflower oil contains chlorophylls and carotenoids. During refining, gossypol forms sodium gossypolate, which is unstable and can readily react with oxygen or interact with phosphatides and proteins to form complex compounds such as gossyproteins and gossyphosphatides.The presence of these compounds in soapstock also affects the subsequent acid decomposition process of the fully saponified mass. In particular:1. During alkaline treatment.Unlike biological degradation, enzymatic β-oxidation does not occur under alkaline conditions. Instead, chemical transformations take place, especially at elevated temperatures. Unsaturated fatty acids undergo degradation reactions leading to the formation of water-soluble salts (soaps), glycerol, aldehydes, and ketones.Saturated fatty acids, under strong alkaline conditions, may undergo destructive reactions, resulting in cleavage of the carbon chain and formation of short-chain carboxylic acids, and in extreme cases, complete degradation to carbon dioxide and water, along with ketone formation.Unsaturated fatty acids are more susceptible to degradation due to the presence of double bonds, which are easily cleaved under alkaline conditions. This leads to the formation of short-chain acids (e.g., butyric acid) or dicarboxylic acids. One of the major losses in the process is associated with such degradation reactions, which continue under the influence of oxygen. Short-chain acids (e.g., butyric acid) can further participate in reactions forming anhydrides and amides. These compounds partially remain in the saponified mass, but a significant portion is removed into the aqueous phase during washing with sodium chloride solution.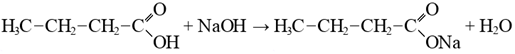 Fatty acid + sodium alkali → sodium butyrate + water
Fatty acid + sodium alkali → sodium butyrate + water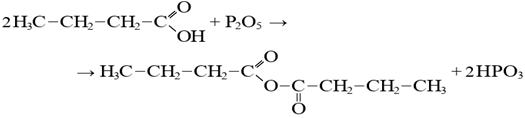 Oil acid + phosphorus oxide → oil anhydride + phosphoric acid
Oil acid + phosphorus oxide → oil anhydride + phosphoric acid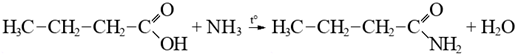 Fatty acid + ammonia → butyramide + water2. Acid decomposition stage.Treatment with sulfuric acid at elevated temperature is an intensive chemical process that not only enables the release of fatty acids from sodium salts formed during caustic saponification, but also leads to destruction of the hydrocarbon chains:2R-COONa + H2SO4 = 2R-COOH + Na2SO4During caustic decomposition, short-chain fatty acid residues formed and retained in the oil phase may recombine, resulting in the formation of fatty acids:
Fatty acid + ammonia → butyramide + water2. Acid decomposition stage.Treatment with sulfuric acid at elevated temperature is an intensive chemical process that not only enables the release of fatty acids from sodium salts formed during caustic saponification, but also leads to destruction of the hydrocarbon chains:2R-COONa + H2SO4 = 2R-COOH + Na2SO4During caustic decomposition, short-chain fatty acid residues formed and retained in the oil phase may recombine, resulting in the formation of fatty acids: 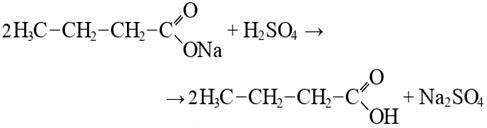 As a result of degradation processes, sulfated fatty acids may also be formed:
As a result of degradation processes, sulfated fatty acids may also be formed: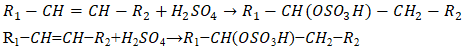 As a result, these processes, firstly, lead to losses during complete saponification and acid decomposition, and secondly, increase reagent consumption due to the formation of additional by-products. Based on the above, it can be concluded that the transfer of hydrophilic compounds reacting with alkali into the aqueous phase contributes to:• improvement of the color of the saponified mass, • reduction in alkali consumption during saponification, and • reduction in acid consumption during decomposition. In the salting-out process of the saponified mass, not only the concentration of sodium chloride but also its amount is of great importance. In our study, since the first stage involved carbonate saponification, the amount of electrolyte was selected to ensure complete separation of the aqueous phase from the system.For this purpose, the effect of adding a 20% sodium chloride solution in the range of 10-70% relative to the mass of the oil phase in the soapstock on aqueous phase separation was investigated.The experimental results showed that the saponified mass can exist in different structural states, as illustrated in Figure 2.
As a result, these processes, firstly, lead to losses during complete saponification and acid decomposition, and secondly, increase reagent consumption due to the formation of additional by-products. Based on the above, it can be concluded that the transfer of hydrophilic compounds reacting with alkali into the aqueous phase contributes to:• improvement of the color of the saponified mass, • reduction in alkali consumption during saponification, and • reduction in acid consumption during decomposition. In the salting-out process of the saponified mass, not only the concentration of sodium chloride but also its amount is of great importance. In our study, since the first stage involved carbonate saponification, the amount of electrolyte was selected to ensure complete separation of the aqueous phase from the system.For this purpose, the effect of adding a 20% sodium chloride solution in the range of 10-70% relative to the mass of the oil phase in the soapstock on aqueous phase separation was investigated.The experimental results showed that the saponified mass can exist in different structural states, as illustrated in Figure 2.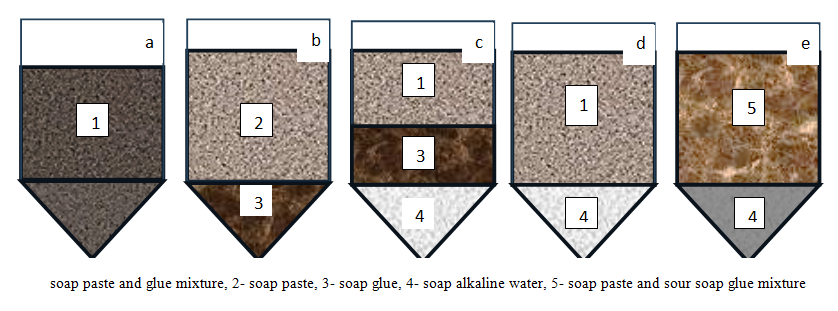 | Figure 2. Cases of fractionation of the saponified mass |
As shown in Figure 2, in the absence of separation of the alkaline sub-soap aqueous phase, the system (case a) consists of a mixture of soap curd and soap gel (1). Upon settling, this mass separates into soap curd (2) and soap gel (3), as illustrated in case b. If the saponified mass contains a high amount of water, or if an electrolyte solution with a concentration above the critical level and in sufficient quantity is added, the system may further separate into distinct phases:• either into soap curd (2), soap gel (3), and alkaline sub-soap aqueous phase (4) (case c), • or into soap curd (2) and alkaline sub-soap aqueous phase (4) (case d). In our experiments, during the first stage of complete saponification, a calcined soda solution, which has lower activity compared to sodium hydroxide, was used. As a result, the process did not proceed to completion, and only about 80% of the mass was saponified.Under these conditions of incomplete saponification, the formation of acid soaps hinders the separation of soap curd (2) and soap gel (3). Consequently, a heterogeneous mixture (case e) containing hydrophobic acid soaps (5) is formed. When a sodium chloride solution (electrolyte) is added to this mixture, the transfer of coloring substances and water-soluble compounds into the aqueous phase is facilitated, and their separation from the oil phase becomes easier.In our experiments, the aqueous phase obtained from mixture (e) exhibited a darker color compared to that separated from systems (c) and (d). This can be explained by a higher diffusion of coloring impurities into the aqueous phase. The results obtained by treating the soap mixture with a 20% sodium chloride solution in the range of 10-70% (relative to the mass) are presented in Table 2.Table 2. Effect of electrolyte dosage on the phase separation of carbonate-saponified mass
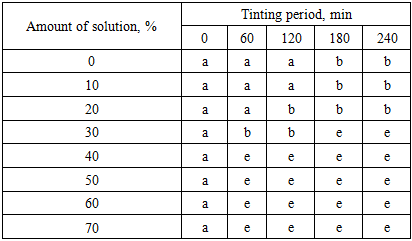 |
| |
|
As shown in Table 2, in the absence of electrolyte addition, no clear separation of the aqueous phase was observed even after 4 hours (240 min) of settling. Similar results were obtained when 10-20% sodium chloride solution was added to the saponified mass. However, when the amount of sodium chloride solution exceeded 30% (relative to the mass), the desired process was achieved, and a distinct separation of the alkaline sub-soap aqueous phase was observed within 60-240 minutes. Specifically:• at 30% electrolyte addition, clear separation occurred within 180-240 min, • at 40%, within 60-180 min, • at 50%, within 60-120 min. Under these conditions, the minimum residual water content in the soap phase and the best color characteristics were also achieved. Further increase of the electrolyte amount to 60-70% resulted in similar outcomes. Based on these findings, it was concluded that washing the carbonate-saponified mass with up to 50% electrolyte solution is optimal.According to the proposed method, the carbonate-saponified mass, previously washed (salted-out) using a 20% sodium chloride solution at 50% dosage, is subjected in the second stage to complete saponification with a 42% sodium hydroxide solution at 90°C for 180 minutes.In conventional technology, the fully saponified mass is treated with a sodium chloride solution and then allowed to settle for 4 hours. However, in the proposed process, the separation of the soap and aqueous phases is accelerated, similar to the acceleration of the saponification stage itself.As demonstrated in the previous experiments, the addition of 0.01-0.02% antioxidant (ionol) prior to complete saponification reduces the formation of oxidized fatty acids. In addition, washing the carbonate-saponified mass with the optimal amount of electrolyte (50%) allows the majority of accompanying substances to be transferred into the alkaline aqueous phase and removed, followed by caustic saponification.The significant reduction of emulsifying, water-insoluble, and non-saponifiable substances in the reaction mass enhances the efficiency of the process, leading to:• reduced consumption of caustic soda for complete saponification, and • reduced electrolyte requirement (or increased effectiveness) for phase separation. To verify these assumptions experimentally, in the second stage, the fully saponified mass obtained using caustic soda was washed with a 20% sodium chloride solution in the range of 10-70% relative to the mass, in order to determine the optimal electrolyte dosage and process duration. The experimental results are presented in Figure 3.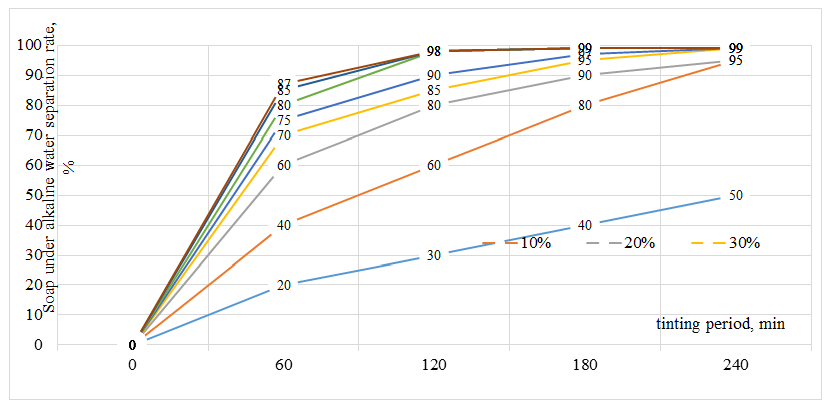 | Figure 3. The effect of the amount of electrolyte on aqueous phase separation from the caustic-finished soapy mass |
As can be observed, the separation of the aqueous phase from the carbonate-saponified mass using a saline electrolyte leads to a significant acceleration of both the caustic complete saponification process and the subsequent phase separation, due to the prior removal of a large portion of accompanying substances. As shown in Figure 3, in the conventional industrial process (control), where complete saponification is carried out in a single stage using caustic soda followed by washing with a salt solution, the settling process is considerably slower. This is due to the presence of accompanying substances, particularly emulsifying compounds that hinder phase separation. Under these conditions, only about 50% phase separation is achieved after 4 hours (240 min), while complete separation requires 6-8 hours.In contrast, the proposed two-stage saponification process, with washing of the soap mass using saline solution at each stage, significantly facilitates the removal of accompanying substances from the fully saponified mass. In particular, the use of 10-50% saline solution (relative to the mass) accelerates the settling process, resulting in 40-98% separation of the aqueous phase within 60-120 minutes. Further increase in electrolyte dosage to 60-70% does not significantly affect the rate of separation. Based on these findings, it is considered optimal in the second stage to wash the caustic fully saponified mass using a 20% sodium chloride solution at 50% dosage (relative to the mass).Thus, as a result of the two-stage complete saponification process, the composition of the obtained saponified mass is presented in Table 3.Table 3. Characteristics of the mass obtained after two-stage complete saponification
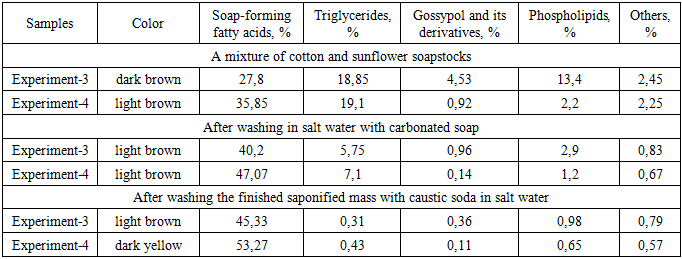 |
| |
|
As shown in Table 3, after complete caustic saponification in the second stage, the degree of saponification relative to the total oil fraction reached 45.33 (94.9%) for Experiment 3 samples and 53.27 (95.8%) for Experiment 4 samples. The content of unreacted triglycerides was correspondingly low, amounting to 0.31% and 0.43%, respectively. In addition, it can be observed that the content of gossypol derivatives, phospholipids, and other accompanying substances, including coloring impurities, present in the oil phase, decreased significantly after the first and second stages of saponification and subsequent washing with electrolyte solution.The average molecular weight of fatty acids in the soapstock mixtures varies depending on the ratio of individual components. In this study, the average molecular weights of fatty acids in the cottonseed-sunflower soapstock mixtures were determined by calculation based on their known fatty acid compositions. The obtained results are presented in Figure 4.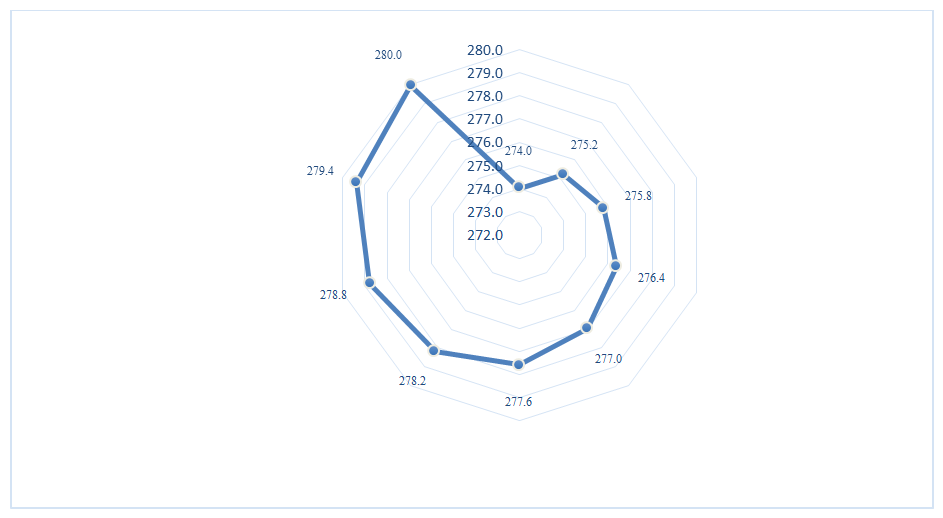 | Figure 4. Influence of soapstock composition ratio on the average molecular weight of fatty acids |
As shown in Figure 4, the average molecular weight of fatty acids in cottonseed soapstock is 274 g/mol. In contrast, the fatty acids in sunflower soapstock have a higher average molecular weight of 280.0 g/mol. With the addition of sunflower soapstock to cottonseed soapstock, the average molecular weight of the fatty acid mixture increases accordingly, reaching 277 g/mol at the 50/50 ratio used in this study. Based on the determined average molecular weight of fatty acids, the neutralization number of the soapstock mixture was evaluated using both calculation and titration methods. The calculation was performed according to the following formula (7):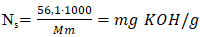 | (7) |
Nₛ - neutralization number of the soapstock mixture;Mₘ - average molecular weight of the soapstock mixture;56.1 - molecular weight of KOH;1000 - conversion factor.The results obtained by calculation using the formula and by experimental determination were in good agreement, and the results are presented in Figure 5.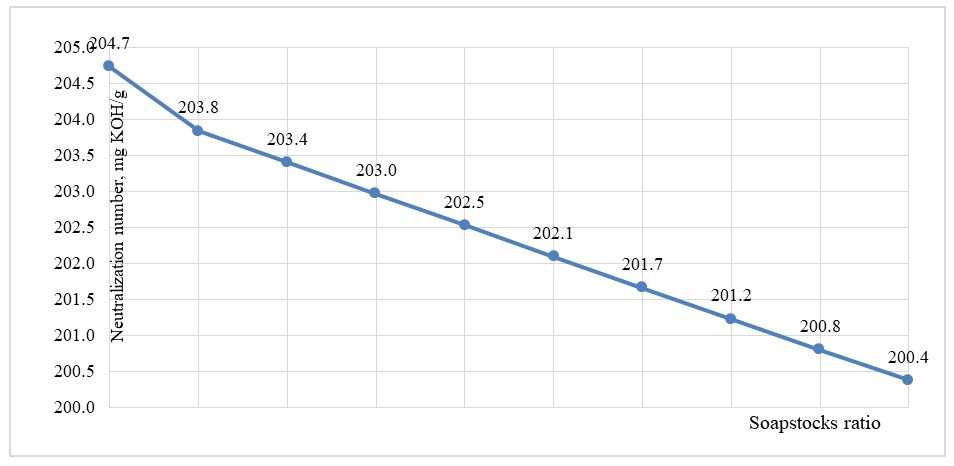 | Figure 5. Change in neutralization number depending on the ratio of soap stocks |
As shown in Figure 5, in contrast to the increase in average molecular weight, the neutralization number decreases with an increasing proportion of sunflower soapstock in the cottonseed soapstock mixture. Specifically, the neutralization number of cottonseed soapstock is 204.7 mg KOH/g. With the addition of 50% sunflower soapstock, this value decreases to 202.5 mg KOH/g, while at a 10/90 ratio, it further decreases to 200.8 mg KOH/g, reaching 200.4 mg KOH/g for pure sunflower soapstock. This trend can be explained by the fact that sunflower soapstock contains a higher proportion of unsaturated fatty acids, characteristic of this type of oil. As a result, with increasing sunflower soapstock content in the mixture, the iodine value increases correspondingly.The results of the iodine value determination experiments are presented in Figure 6.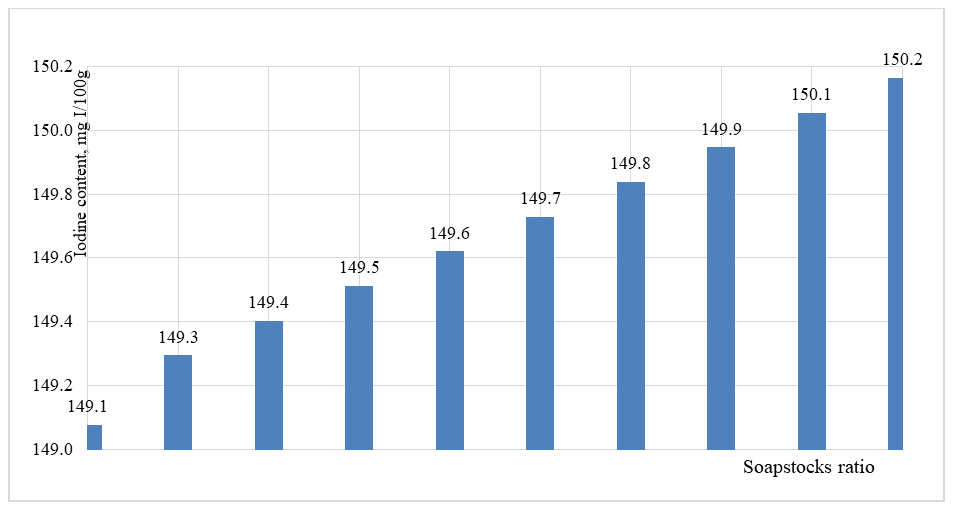 | Figure 6. The effect of the change in the ratio of soapstocks on the iodine number of fatty acids |
As shown in Figure 6, the iodine value of cottonseed soapstock is 149.1 mg I₂/100 g. For the 50/50 soapstock mixture, this value slightly increases to 149.6 mg I₂/100 g, while for pure sunflower soapstock, it reaches 150.2 mg I₂/100 g. The relatively small variation in iodine value can be explained by the fact that both soapstocks have similar contents of unsaturated fatty acids, which primarily determine the iodine value. Since the target fatty acids obtained in this study are intended for applications in the production of soaps and detergents, as well as in the chemical industry, lubricants, and biodiesel, the melting and solidification temperatures of the fatty acids are of significant importance. The melting temperature (titer value) of the fatty acids was determined experimentally for different soapstock mixture ratios. The results are presented in Figure 7.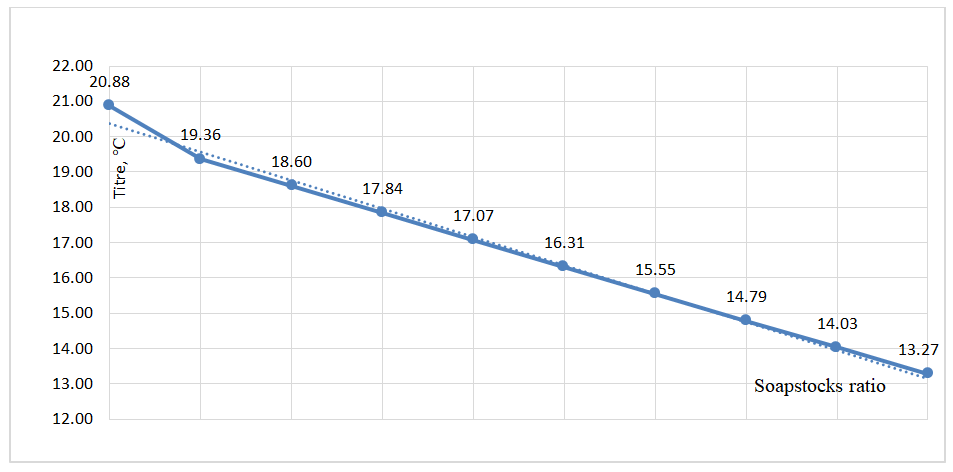 | Figure 7. The effect of the change in the ratio of soapstocks on the titer of fatty acids |
As shown in Figure 7, the titer (melting point) of fatty acids derived from cottonseed soapstock is 20.88°C, whereas for sunflower soapstock, it is significantly lower at 13.27°C. Upon mixing the two soapstocks, the titer value decreases accordingly, reaching 17.07°C at a 50/50 ratio.
5. Conclusions
The conducted research demonstrates that the two-stage saponification approach combined with electrolyte-assisted phase separation is an effective method for improving the processing of soapstock and enhancing the yield and quality of fatty acids. It was established that the composition of soapstock mixtures (cottonseed/sunflower) significantly influences key physicochemical properties, including average molecular weight, neutralization number, iodine value, and titer (melting point). An increase in the proportion of sunflower soapstock leads to an increase in the average molecular weight and iodine value, while simultaneously decreasing the neutralization number, which is consistent with the higher content of unsaturated fatty acids.The study confirmed that the electrolyte concentration and dosage (NaCl solution) play a decisive role in the efficiency of phase separation. The optimal conditions were determined to be:• 20% NaCl concentration, • - 50% dosage relative to the mass, which ensure:• rapid separation of the aqueous phase (within 60-120 min), • improved removal of emulsifying impurities, • reduction of residual water in the soap phase, • improved color characteristics of the final product. A key scientific contribution of this work is the demonstration that preliminary carbonate saponification followed by electrolyte washing significantly reduces the content of phospholipids, gossypol derivatives, oxidized fatty acids, and other emulsifying substances, which are known to hinder phase separation and increase reagent consumption. This leads to:• acceleration of the caustic saponification stage, • reduction in alkali and acid consumption, • improved process efficiency. The degree of saponification achieved in the second stage reached up to 94.9-95.8%, with minimal residual triglycerides (0.31-0.43%), confirming the effectiveness of the proposed method. In addition, it was shown that technological parameters (temperature, electrolyte concentration, and process staging) directly affect not only the yield but also the quality indicators of fatty acids, which are critical for their application in:• soap and detergent production, • chemical industry, • lubricants, • biodiesel production. Thus, the proposed two-stage saponification technology with optimized electrolyte treatment provides a scientifically justified and industrially applicable solution for:• increasing fatty acid yield, • improving product quality, • reducing environmental impact and reagent consumption.
References
| [1] | Chemistry of Fats lecture notes - R. M. Markevich, J. V. Bondarenko. Chemistry of fats: lecture notes for students specializing in Biotechnology (Technology of fats, essential oils, and perfumery-cosmetic products). Minsk: BGTU, 2011. 220 p. ISBN 978-985-530-057-2. |
| [2] | Soap base from secondary resources of oil and fat industry - D. T. Ruzmetova, Sh. R. Kurambayev, U. S. Boltayev, P. R. Sharipov. Soap base from secondary resources of oil and fat industry. Molodoy Uchenyy, 2016, No. 7 (111), pp. 161-164. |
| [3] | Study of cottonseed oil soapstock - M. S. Nair, Ramachandran, S. Parakasa Rao. Study of cottonseed oil soapstock. Indian Oil and Soap Journal, 1965, Vol. 4, pp. 85-88. |
| [4] | Barbusiński, K., Kołodziejczyk, K., & Filipek, K. (2021). Optimization of soapstock splitting process to reduce environmental impact. Journal of Cleaner Production, 279, 123456. https://doi.org/10.1016/j.jclepro.2020.123456. |
| [5] | Kalyna, V., Koshulko, V., & Ilinska, O. (2021). Development of soapstock processing technology to ensure waste-free and safe production. Eastern-European Journal of Enterprise Technologies, 6(11), 45-53. https://doi.org/10.15587/1729-4061.2021.245678. |
| [6] | Sytnik, N., Kunitsia, E., & Melnyk, O. (2021). Technology development for obtaining fatty acids from soapstock using saponification methods. Chemical Engineering Transactions, 88, 235-240. https://doi.org/10.3303/CET2188039. |
| [7] | Casali, B., Rossi, M., & Ricci, A. (2021). Valorization of soapstock for fatty acid-based products: Process optimization and applications. Processes, 9(1), 112. https://doi.org/10.3390/pr9010112. |
| [8] | Ruzmetova, D. T., Kurambayev, S. R., Boltayev, U. S., & Sharipov, P. R. (2025). Study of quality indicators of fatty acids obtained during soapstock processing by multi-stage technology. Journal of Chemical Technology and Biotechnology, 100(2), 567-575. https://doi.org/10.1002/jctb.7890. |
| [9] | Nair, M. S., Ramachandran, S., & Rao, S. P. (2020). Advances in the study of cottonseed oil soapstock processing and fatty acid recovery. Journal of the American Oil Chemists’ Society, 97(5), 567-575. https://doi.org/10.1002/aocs.12345. |


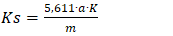
 - the volume of 0.1 N solution used for titration, ml;m - the mass of the sample under analysis, g;K - correction factor for the titer.In determining the amount of organic acid salts in soapstock and fatty acids, compounds present in the soapstock that react with sulfuric acid were taken into account. These include low-molecular-weight and oxidized acidic impurities, amino compounds, glycerophosphoric acid, gossypol derivatives, and other saponified substances.The analyses were carried out by potentiometric titration in an alcoholic medium. For the study, a soapstock sample weighing 0.5-1.0 g was taken and dissolved in alcohol. A 0.1 mol/dm³ alcoholic solution of hydrochloric acid was used as the titrant.Based on the titration results, potentiometric titration curves were constructed to determine the equivalent volume of the titrant. The amount of organic acid salts was calculated using the following formula:
- the volume of 0.1 N solution used for titration, ml;m - the mass of the sample under analysis, g;K - correction factor for the titer.In determining the amount of organic acid salts in soapstock and fatty acids, compounds present in the soapstock that react with sulfuric acid were taken into account. These include low-molecular-weight and oxidized acidic impurities, amino compounds, glycerophosphoric acid, gossypol derivatives, and other saponified substances.The analyses were carried out by potentiometric titration in an alcoholic medium. For the study, a soapstock sample weighing 0.5-1.0 g was taken and dissolved in alcohol. A 0.1 mol/dm³ alcoholic solution of hydrochloric acid was used as the titrant.Based on the titration results, potentiometric titration curves were constructed to determine the equivalent volume of the titrant. The amount of organic acid salts was calculated using the following formula: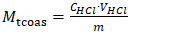
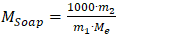
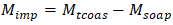
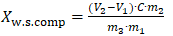
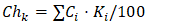

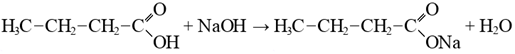 Fatty acid + sodium alkali → sodium butyrate + water
Fatty acid + sodium alkali → sodium butyrate + water Oil acid + phosphorus oxide → oil anhydride + phosphoric acid
Oil acid + phosphorus oxide → oil anhydride + phosphoric acid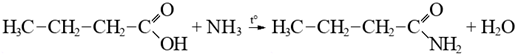 Fatty acid + ammonia → butyramide + water2. Acid decomposition stage.Treatment with sulfuric acid at elevated temperature is an intensive chemical process that not only enables the release of fatty acids from sodium salts formed during caustic saponification, but also leads to destruction of the hydrocarbon chains:2R-COONa + H2SO4 = 2R-COOH + Na2SO4During caustic decomposition, short-chain fatty acid residues formed and retained in the oil phase may recombine, resulting in the formation of fatty acids:
Fatty acid + ammonia → butyramide + water2. Acid decomposition stage.Treatment with sulfuric acid at elevated temperature is an intensive chemical process that not only enables the release of fatty acids from sodium salts formed during caustic saponification, but also leads to destruction of the hydrocarbon chains:2R-COONa + H2SO4 = 2R-COOH + Na2SO4During caustic decomposition, short-chain fatty acid residues formed and retained in the oil phase may recombine, resulting in the formation of fatty acids: 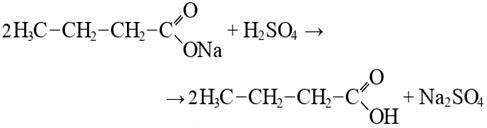 As a result of degradation processes, sulfated fatty acids may also be formed:
As a result of degradation processes, sulfated fatty acids may also be formed: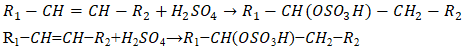 As a result, these processes, firstly, lead to losses during complete saponification and acid decomposition, and secondly, increase reagent consumption due to the formation of additional by-products. Based on the above, it can be concluded that the transfer of hydrophilic compounds reacting with alkali into the aqueous phase contributes to:• improvement of the color of the saponified mass, • reduction in alkali consumption during saponification, and • reduction in acid consumption during decomposition. In the salting-out process of the saponified mass, not only the concentration of sodium chloride but also its amount is of great importance. In our study, since the first stage involved carbonate saponification, the amount of electrolyte was selected to ensure complete separation of the aqueous phase from the system.For this purpose, the effect of adding a 20% sodium chloride solution in the range of 10-70% relative to the mass of the oil phase in the soapstock on aqueous phase separation was investigated.The experimental results showed that the saponified mass can exist in different structural states, as illustrated in Figure 2.
As a result, these processes, firstly, lead to losses during complete saponification and acid decomposition, and secondly, increase reagent consumption due to the formation of additional by-products. Based on the above, it can be concluded that the transfer of hydrophilic compounds reacting with alkali into the aqueous phase contributes to:• improvement of the color of the saponified mass, • reduction in alkali consumption during saponification, and • reduction in acid consumption during decomposition. In the salting-out process of the saponified mass, not only the concentration of sodium chloride but also its amount is of great importance. In our study, since the first stage involved carbonate saponification, the amount of electrolyte was selected to ensure complete separation of the aqueous phase from the system.For this purpose, the effect of adding a 20% sodium chloride solution in the range of 10-70% relative to the mass of the oil phase in the soapstock on aqueous phase separation was investigated.The experimental results showed that the saponified mass can exist in different structural states, as illustrated in Figure 2.


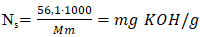



 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML

