-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Materials and Chemistry
p-ISSN: 2166-5346 e-ISSN: 2166-5354
2026; 16(1): 6-10
doi:10.5923/j.ijmc.20261601.02
Received: Feb. 24, 2026; Accepted: Mar. 10, 2026; Published: Mar. 16, 2026

Synthesis Preparation and Optical-Dimensional Properties of Luminescent Polymer Nanocomposite Films Based on CuInS2/ZnS Quantum Dots
Islomova Z. R.1, Tursunova N. R.1, Ishankulov A. F.1, Khalilov K. F.1, Galyametdinov Y. G.2
1Samarkand State University, Samarkand, Uzbekistan
2Kazan National Research Technological University, Kazan, Russia
Correspondence to: Ishankulov A. F., Samarkand State University, Samarkand, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In this study, the preparation of nanocomposite materials based on quantum dots (QDs) and polymers was studied. Polymethylmethacrylate (PMMA) was used as a matrix, and semiconductor quantum dots were embedded in it. The importance of the stabilizer in the synthesis of polymer nanocomposites was observed, the dependence of the absorption spectra of hybrid CuInS2/ZnS quantum dots polymer nanocomposites on the octadecene concentration, and the effect of impurities in the oleic acid (OA) composition on the yield of quantum dots. Other additives in OA affect the monodispersity of nanoparticles. The obtained nanoparticles were subjected to surface modification reactions by replacing the stabilizer. The replacement of oleic acid with trioctylphosphine oxide (TOPO) changed the optical-dimensional properties of CuInS2/ZnS colloidal nanocrystals. The addition of TOPO to oleic acid reduced the nanoparticle size from 9.4 to 8.9 nm, which led to a progressive shift in the ultraviolet and photoluminescence spectra.
Keywords: Polymer, Quantum dots, QD synthesis, Polymer, Nanocrystal, Semiconductor, Nanocomposite
Cite this paper: Islomova Z. R., Tursunova N. R., Ishankulov A. F., Khalilov K. F., Galyametdinov Y. G., Synthesis Preparation and Optical-Dimensional Properties of Luminescent Polymer Nanocomposite Films Based on CuInS2/ZnS Quantum Dots, International Journal of Materials and Chemistry, Vol. 16 No. 1, 2026, pp. 6-10. doi: 10.5923/j.ijmc.20261601.02.
Article Outline
1. Introduction
- Colloidal semiconductor QDs are ideal luminescent nanoparticles. Since semiconductor nanocrystals are discrete electronic structures, they are also called artificial atoms. Quantum dots have the ability to control their physical properties, comparable to artificial molecules. In this case, the size of the nanoparticle behaves like artificial molecules based on the interaction of electrons of QDs clusters. The size, shape, and physicochemical properties of these nanoparticles can be controlled through emission processes. Currently, the photostability of QDs has been achieved to be higher than that of organic fluorescent dyes [1-3]. It is known that semiconductor nanocrystals have great potential for biosensors, lasers, quantum dot-based light-emitting diodes, diagnostics, and targeted drug delivery to the body due to their unique optical and electronic properties [4,5].In recent decades, the rapid development of nanotechnology has led to the development of nanomaterials with unique properties for new-generation optoelectronic devices, sensors, and biomedical diagnostics. In this direction, semiconductor quantum dots are distinguished by their size-dependent optical properties, broad absorption spectrum, and narrow luminescence lines. Due to the quantum confinement effect, it is possible to control the physicochemical properties of QDs by changing their size, shape, and composition [6-8]. However, the presence of toxic heavy metals in the composition of traditional II-VI group quantum dots (e.g., CdSe, PbS) limits their widespread use. Therefore, there is growing interest in hybrid quantum dots, which belong to the class of environmentally friendly and "green" materials, in particular, core-shell structures coated with multicomponent copper-indium sulfide (CuInS₂), zinc sulfide (ZnS), and carbon-based hybrid nanostructures. Such hybrid systems have not only low toxicity, but also high quantum efficiency and thermal stability [9]. In the practical application of quantum dots, their solid-state, flexible and transparent forms, especially nanocomposite films, are of great importance rather than colloidal solutions. Polymer matrices (e.g. PMMA, PVA, PS) simultaneously act as a passivator protecting against the external environment and a medium for evenly distributing particles in the nanocomposite. By introducing hybrid quantum dots into a polymer matrix, it is possible to obtain optical films with high luminescent properties, mechanical strength and the ability to be shaped into various shapes [10-12]. Such films are the main components in the creation of spectral converters for solar cells, light-emitting diodes (WLEDs) and fluorescent sensors.The aim of this research work is to develop a technology for synthesizing hybrid quantum dots and obtaining luminescent polymer nanocomposite films based on them. The article studies in detail the effect of the amount and size of quantum dots in the nanocomposite on the optical properties of the film, in particular, on the absorption and photoluminescence spectra. The optical-size properties of the obtained nanocomposite films and their structural stability are also analyzed. There are also scientific studies on the use of thiol-containing substances as stabilizers in the synthesis of quantum dots [13-15]. The authors found that other methods can be used to create nanocomposite polymer micelles, which rely on the direct coating of QDs with amphiphilic polymers. Amphiphilic and alkyl-modified low molecular weight polyacrylic acids, coated with trioctylphosphine oxide (TOPO)-protected nanocrystals, render quantum dots water-soluble [16,17]. In this study, the synthesis of colloidal QDs, the preparation of nanocomposites, and their main optical and dimensional properties are briefly discussed. Methods for increasing the quantum yield are described. It should be noted that nanocomposites are an important component of biomaterials, which are used in nanomedicine, targeted drug delivery systems, and biosensors, as well as in a wide range of applications. In medicine, bioactive compounds can be effectively used for various purposes, in particular as biomarkers.
2. Experimental
- Materials and reagentsZinc acetate dihydrate ((CH3COO)2Zn∙2H2O, 99%, Vekton, Russia), cadmium oxide (CdO, Reahim, Russia), zinc oxide (ZnO, Acros, Belgium), oleic acid (90%, Vekton, Russia), sulfur (ODE, 90% Acros, Qatar), dodecanethiol-1 (DT, 98%, Alpha Aesar, UK), octadecene (ODE) rhodamine B (99%, Lenreactiv, Russia), distilled degassed water (pH=7), toluene (analytical grade, HimReactiv), selenium metal powder (99.5%).The hydrodynamic sizes of nanoparticles were determined on a “Malvern Zetasizer Nano” photon correlation spectrometer based on the principle of dynamic light scattering.In the experiments, a JEOL JEM-2100 transmission electron microscope was used to determine the shape and size of the nanoparticles of the samples.Synthesis of CuInS2 quantum dotsCadmium oxide was used as the cadmium precursor in the synthesis of CuInS2 quantum dots. The synthesis process was carried out under a nitrogen atmosphere with constant stirring. In the hot injection method, copper (CuI or CuCl) and indium (InCl3) salts are heated in high-temperature solvents such as oleylamine or 1-octadecene, initially at 100-120°C under vacuum. The temperature is then raised to 180-240°C, and a sulfur source (DDT or S powder in ODE) is rapidly injected into the system; nucleation occurs at this stage, and particle growth is controlled. In this process, dodecanethiol (DDT) acts as both a sulfur source and a ligand (surfactant) that passivates the particle surface, greatly facilitating the synthesis process.On the other hand, the solvothermal or hydrothermal synthesis method is characterized by the fact that it is carried out in hermetic autoclaves under laboratory conditions. In this case, all the reactants, together with the solvent, are placed in a Teflon-lined stainless steel autoclave and heated for several hours at a temperature of 150-200°C. The synthesis of CuInS2 quantum dots was carried out by mixing solutions 1 and 2 at 270°C for 2 minutes. To quickly cool the resulting mixture, 5 ml of ODE and 5 ml of toluene were added to the solution. To purify the mixture from unreacted precursors, ethanol and acetone were added to the solution until the transparency changed to turbidity. Then the mixture was centrifuged for 15 minutes at 5000 rpm to separate the precipitate. The resulting precipitate was dissolved in toluene to purify it. The purification process was repeated 3 times.Synthesis of CuInS2/ZnS quantum dotsPreparation of precursors and thermal treatment: First, 2 mmol zinc acetate, 0.15 mmol copper (I) iodide (CuI) and 5 ml oleic acid were mixed in a three-necked flask. In order to remove oxygen and moisture from the system, the reaction medium was heated to 160°C under a nitrogen stream and held at this temperature for 20 minutes. After that, the temperature was gradually increased to 300°C.Injection and formation of the core/shell structure: After reaching the specified temperature (300°C), a mixture of 0.1 mmol selenium and 1.75 mmol sulfur dissolved in 1 ml trioctylphosphine (TOP) was rapidly injected into the reaction medium. The reaction mixture was kept isothermally at 300°C for 7 minutes to ensure complete formation and crystallization of core/shell heterostructure quantum dots (QDs).Method for preparing QD nanocomposite based on CuInS2/ZnS with polymethylmethacrylate.Preparation of components: In the study, polymethyl methacrylate (PMMA) was chosen as the polymer matrix of the nanocomposite. PMMA is characterized by its high optical transparency, mechanical stability, and the ability to maintain luminescence properties in hybrid systems. To prepare the nanocomposite, initially, CuInS₂/ZnS quantum dots (QDs) and PMMA polymer were each dissolved separately in toluene solvent at a concentration of 2 mg/ml.Formation of hybrid solutions: In order to study the interaction of the components in the composite and the dependence of the optical properties on the concentration, the prepared QD and PMMA solutions were mixed in the following mass ratios: 100/0, 75/25, 50/50, 25/75, and 0/100. The resulting hybrid solutions were subjected to ultrasonic treatment or intensive mixing until they became homogeneous. Nanocomposite films were obtained by the "spin-coating" method using a Spin Coater Laurell WS-400-6NPP-LITE device. Quartz glasses with high transparency in the ultraviolet (UV) and visible spectral ranges were used as substrates. Hybrid solutions were deposited on the surface of quartz glass at a speed of 800 rpm, which ensured the formation of a uniform film with the required thickness. The diameter of the quantum dots is determined by the semi-empirical equation using the absorption spectra of particles in the ultraviolet and visible ranges of the obtained spectra:
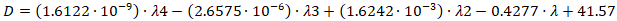 where D is the particle size (nm), λ is the wavelength of the maximum absorption spectrum peak (nm).
where D is the particle size (nm), λ is the wavelength of the maximum absorption spectrum peak (nm).3. Results and Discussion
- Figure 1a displays the spectral characteristics of the synthesized core and hybrid QDs. The deposition of the shell layer significantly intensifies the luminescence, accompanied by a noticeable peak shift from 582 nm to 555 nm. This shift is primarily driven by the altered confinement effects and hydrodynamic dimensions of the QD core. The emission spectra typically exhibit a dual nature: a narrow, symmetric peak representing excitonic recombination and a broad, low-energy tail attributed to trap-state emissions from surface defects. After the shell is grown on the core surface, it can be observed that the spectral maximum shifts towards the shorter wavelength range (Fig. 1a). With increasing synthesis temperature, the symmetry of the spectral peaks can broaden. This phenomenon is associated with an increase in the polydispersity of nanocrystals. Increasing the synthesis time had a negative effect on the photoluminescent properties of quantum dots, which in turn led to an increase in the intensity of defect luminescence relative to the excitation luminescence. The contribution of nanoparticle surface defect levels to the luminescence changes with changing synthesis temperature.After the ZnS shell was grown on the surface of the CuInS2 core, the absorption spectra of the hybrid quantum dots were obtained. (Fig. 1b) It can be seen that the maximum of the exciton peak in the spectrum shifted to the short wavelength range.The exciton peak range is observed in the 500-600 nm wavelength range of the visible region, which corresponds to 537 nm for the hybrid CuInS2/ZnS.From the comparison of the absorption spectra of CuInS2 and CuInS2/ZnS QDs, it can be seen that after the shell was grown on the surface of the core, the exciton peak shifted to the short wavelength range (Fig. 1b).
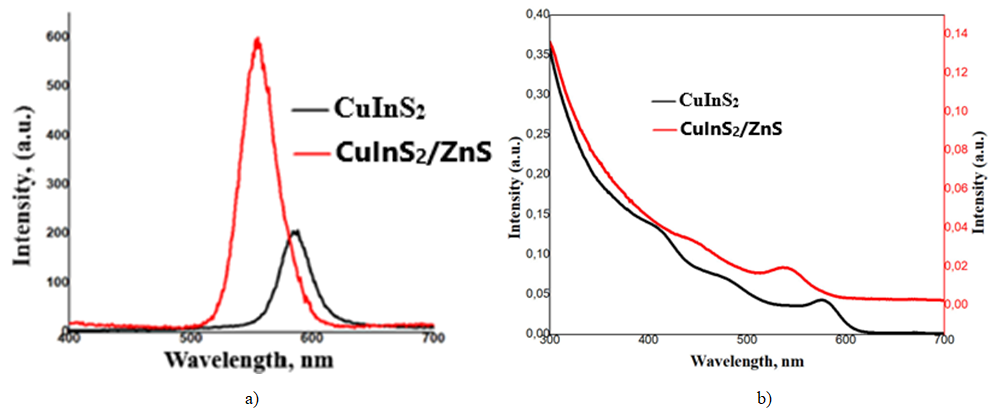 | Figure 1. Luminescence (a) and absorption spectra (b) of CuInS2 and CuInS2/ZnS QDs |
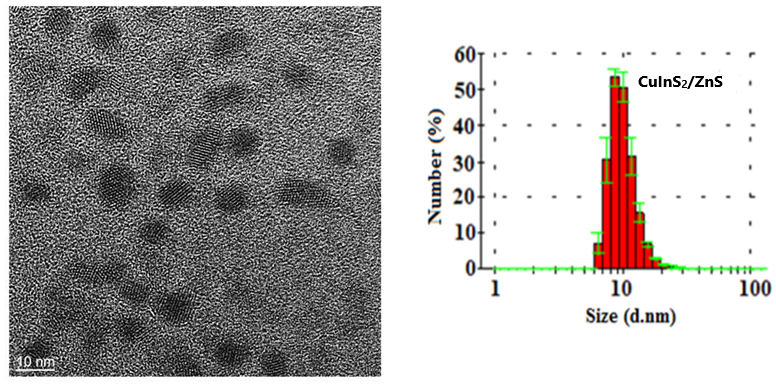 | Figure 2. TEM image and histogram of the average hydrodynamic size distribution of CuInS2/ZnS QDs obtained at 300°C |
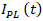 on time for CuInS2 QDs at a wavelength of 555 nm was described exponentially, and the kinetics of the intensity of the nanoparticles were calculated by the following equation (1):
on time for CuInS2 QDs at a wavelength of 555 nm was described exponentially, and the kinetics of the intensity of the nanoparticles were calculated by the following equation (1):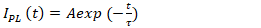 | (1) |
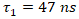 represents the fluorescence decay time.To analyze the fluorescence decay kinetics of the CuInS2/ZnS core/shell quantum dots, the time-dependent intensity was fitted using a biexponential decay model rather than a monoexponential function (2):
represents the fluorescence decay time.To analyze the fluorescence decay kinetics of the CuInS2/ZnS core/shell quantum dots, the time-dependent intensity was fitted using a biexponential decay model rather than a monoexponential function (2):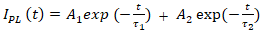 | (2) |
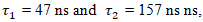 where the latter corresponds to the slow decay component. The contribution of this long-term process is about 11%, which is presumably associated with the recombination of excitons trapped at the interface states between the core and the shell.To further elucidate the charge carrier dynamics, PL decay kinetics were analyzed. The decay profiles of both pristine CuInS2 and core/shell CuInS2/ZnS QDs exhibited an excitation-power-independent nature, confirming the negligible contribution of Auger recombination to the overall relaxation mechanism.In the studies, thin-layer (film) nanocomposites based on PMMA containing CuInS2/ZnS QDs synthesized at a temperature of 270–300°C were obtained (Fig. 3).
where the latter corresponds to the slow decay component. The contribution of this long-term process is about 11%, which is presumably associated with the recombination of excitons trapped at the interface states between the core and the shell.To further elucidate the charge carrier dynamics, PL decay kinetics were analyzed. The decay profiles of both pristine CuInS2 and core/shell CuInS2/ZnS QDs exhibited an excitation-power-independent nature, confirming the negligible contribution of Auger recombination to the overall relaxation mechanism.In the studies, thin-layer (film) nanocomposites based on PMMA containing CuInS2/ZnS QDs synthesized at a temperature of 270–300°C were obtained (Fig. 3).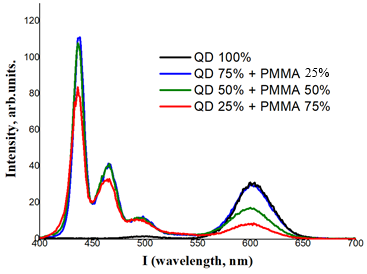 | Figure 4. Photoluminescence spectra of PMMA-based composite films with blue (left) and green (right) CuInS2/ZnS quantum dots at an excitation energy of 𝛼=300 nm |
4. Conclusions
- CuInS2 and hybrid CuInS2/ZnS QDs were synthesized, nanocomposite films were obtained based on an optically active PMMA polymer matrix, and their optical and dimensional properties were studied.The growth of the shell on the surface of the CuInS2 core led to a significant increase in the luminescence intensity of the hybrid QDs obtained. A shift of the maximum wavelength of the luminescence spectrum from 582 nm to 555 nm was observed. This phenomenon can be explained by a change in the hydrodynamic size of the QD core.The optimal time for synthesizing quantum dots with high luminescence intensity was set to 7 minutes and the temperature to 300°C. The concentration of CuInS2/ZnS quantum dots and PMMA matrix in a ratio of 75/25 led to an increase in the luminescence intensity of the nanocomposite.It was found that the absorption spectra of hybrid CuInS2/ZnS quantum dots polymer nanocomposites depend on the octadecene concentration, and it was observed that impurities in the oleic acid) content also affect the performance of quantum dots.
Formatting of Funding Sources
- The author declare that he did not receive any grants for this research.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML