-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Materials and Chemistry
p-ISSN: 2166-5346 e-ISSN: 2166-5354
2025; 15(4): 83-87
doi:10.5923/j.ijmc.20251504.03
Received: Nov. 6, 2025; Accepted: Nov. 23, 2025; Published: Nov. 25, 2025

Amination of Cotton Pulp and Dyeing with Metal Complexes of D–element Ions
Matluba Djumaeva1, Mukhtar Amonov2
1Independent Researcher, Bukhara State University, Bukhara, Uzbekistan
2Professor, Bukhara State University, Bukhara, Uzbekistan
Copyright © 2025 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The study investigates the rheological and chemical modification behavior of cellulose-based dye compositions under varying conditions. The relevance of this work lies in the growing demand for improved dyeability and functionalization of cellulose fibers in textile processing. The main objective was to determine the viscosity and thixotropic characteristics of dye compositions and to examine the chemical interactions occurring during cellulose modification. Experimental methods included rheological measurements, acetylation of cellulose using acetic anhydride, and subsequent amination through the addition of ethylenediamine. The resulting cellulose acetate and aminated cellulose derivatives were analyzed for cation exchange capacity and nitrogen content, particularly when incorporating metal ions into the modified cotton fabric. The results showed that viscosity and thixotropy were strongly dependent on temperature, shear rate, and composition ratio, indicating significant structural rearrangements within the dye system. The introduction of ethylenediamine enhanced the degree of substitution and provided active nitrogen sites for metal ion binding. Overall, the study demonstrates that controlled acetylation and amination reactions can effectively improve the physicochemical and dye-adsorption properties of cotton cellulose, paving the way for the development of functionalized textile materials with enhanced performance characteristics.
Keywords: Amination, Cotton pulp, Dye composition, Metal complexes, D–element ions, Cellulose, Thixotropic reduction, Acetic anhydride, Ethylenediamine, Fabric, Modification, Viscosity, Silk, Cotton fabrics
Cite this paper: Matluba Djumaeva, Mukhtar Amonov, Amination of Cotton Pulp and Dyeing with Metal Complexes of D–element Ions, International Journal of Materials and Chemistry, Vol. 15 No. 4, 2025, pp. 83-87. doi: 10.5923/j.ijmc.20251504.03.
Article Outline
1. Introduction
- Many domestic and foreign researchers are conducting scientific research on obtaining a dye composition based on the amination of cotton cellulose and dyeing with complex d–metal ions, and the determination of their properties is of great fundamental and practical importance. Today, it is important to achieve high efficiency through the use of such dyeing compositions in the light and textile industries for dyeing silk and cotton fabrics [1–3].In recent years, research institutes and universities in the Republic of Uzbekistan have been conducting research on the synthesis and study of the properties of new oxidation–binding compounds based on soluble amino–containing organic compounds, as well as their practical application. Particular attention is paid to the synthesis of target compositions aimed at improving high performance and coloristic properties when dyeing various fabrics [4].
2. Materials and Methods
- The research was carried out using cotton cellulose as the main substrate, acetic anhydride, ethylenediamine, and salts of d-element metals such as nickel, cobalt, iron, and copper. The cellulose was pretreated with a dilute alkali solution, washed, and dried. Acetylation was performed by reacting cellulose with acetic anhydride at 45–50°C for two hours at a molar ratio of 1:1 to obtain cellulose acetate. The aminated derivative was then prepared by treating the cellulose acetate with a 5% ethylenediamine solution in an alkaline medium at pH 8–10, temperature 60–70°C, and a molar ratio of cellulose acetate to ethylenediamine of 1:1.2. The reaction lasted three hours, resulting in modified cellulose containing amino groups capable of forming coordination bonds with metal ions. To determine optimal parameters, compositions of cotton cellulose with acetic anhydride, ethylenediamine, and triethylamine were prepared, and their viscosity and thixotropic recovery were measured at 303 K using a rotational viscometer. The degree of thixotropic recovery was determined by comparing viscosity values immediately after preparation and after two days of storage. Metal complexes were obtained by immersing aminated cotton cellulose in 0.05 M aqueous solutions of Ni²⁺, Co²⁺, Fe³⁺, and Cu²⁺ salts at 40–45°C for one hour with triethylamine and zinc acetylacetonate as stabilizers. The content of metal cations and nitrogen in the modified fabric was determined by spectrometric titration with Trilon B and by the Kjeldahl method. The obtained aminated cellulose showed high stability, improved reactivity, and strong coordination with metal ions, confirming its suitability for use in dyeing compositions.
3. The Main Findings and Results
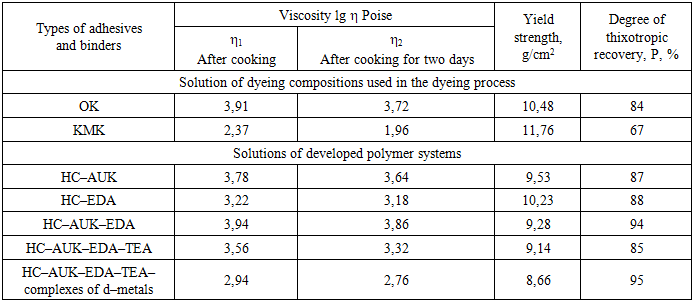
- Most studies have shown that cellulose itself does not have a high chelating capacity. To obtain colored metal complexes within the structure of cellulose fibers, which will contribute to their durable coloration, it is necessary to functionalize it by introducing an amino group into the polymer substrate. Amino groups have an unshared pair of electrons on nitrogen and can form a covalent bond with a metal. The presence of primary amino groups in polymer substrate molecules enables their diazotization to produce highly active diazonium salts. Combining diazonium salts with amines produces colored compounds [5–7].In connection with the above, the development of effective compositions and technology for dyeing cotton cellulose fabrics using polyvalent metal salts based on local raw materials by forming metal complexes is a pressing issue.Today, the textile industry of our country is achieving certain results in the creation of new types of highly effective, inexpensive, import–substituting products based on local raw materials and their introduction into practice as a dyeing composition for dyeing fabrics made of cotton fiber.In this regard, it is of great importance to develop a new composition of dye–binder composition based on local raw materials used in dyeing cotton fabrics, to determine their structure, physical and chemical properties, to create an effective method of dyeing with d–metal complexes and to develop production technologies that are economically advantageous.Dye and binder systems play a crucial role in the cotton fabric dyeing process. The cost of dye and binder systems has a significant economic impact on cotton fabric production. Improvement of the technology for obtaining and using dyeing compositions contributes to an increase in the quality indicators of fabrics dyed with d–element complexes. Currently, many cotton fabric manufacturers predominantly use imported dyes to dye their fabrics. This, in turn, leads to increased fabric costs.In the textile industry, mixtures of oxidized starch with synthetic polymers are mainly used. The technological and mechanical properties of this system are determined by the degree of dispersion and the size of the colloidal solutions.In our country, the textile industry primarily uses oxidized starch in combination with various synthetic polymers. These thickeners are important for improving the color and performance properties of fabrics, but are not economically viable.In our country, in addition to oxidized starch, silk fiber production plants also use imported products, namely sodium alginate and DHT. The use of sodium alginate and DHT in the process of printing silk fiber fabrics provides high efficiency, however, since these polymer systems are imported from abroad, this leads to an increase in the cost of manufactured products.Therefore, the development of dye compositions that can replace imported products and are highly effective is one of our main goals and objectives. In this regard, we studied the properties of various three– and four–component polymer systems based on cotton cellulose (CC), acetic anhydride (AA), and ethylenediamine (EDA). The results of the studies are presented in Table 1.
|
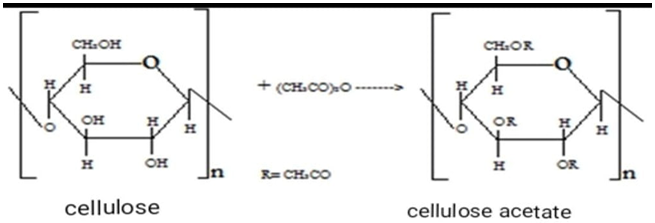 The second reaction mechanism: the resulting intermediate compound cellulose acetate reacts with ethylenediamine:
The second reaction mechanism: the resulting intermediate compound cellulose acetate reacts with ethylenediamine: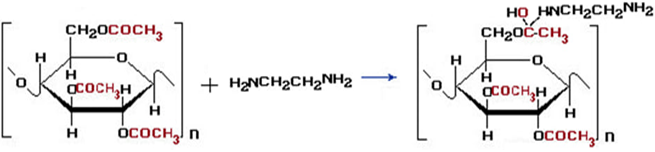 The reactions of cellulose with acetic anhydride and the amination of cellulose acetate obtained by adding ethylenediamine and subsequent dyeing with solutions of d–metal complexes can also be represented by the following mechanism:
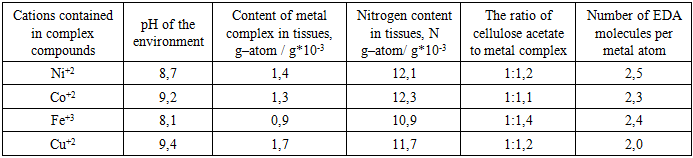
The reactions of cellulose with acetic anhydride and the amination of cellulose acetate obtained by adding ethylenediamine and subsequent dyeing with solutions of d–metal complexes can also be represented by the following mechanism: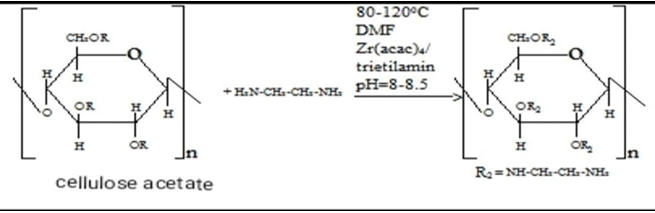 Without these special conditions, normal aminolysis with deacetylation will predominate.Amination is carried out by the following sequence and reaction conditions:1. The amination reaction is carried out at a temperature of 60–70°C. This temperature range is optimal, as determined by studying the reaction kinetics as a function of temperature. The amination reaction was carried out at a cellulose acetate to EDA ratio of 1:1.2 for 3 hours. The EDA solution concentration is 5%;2. An important factor in the formation of hemiacetals during the amination of cotton cellulose is the pH of the medium. Experimental results showed that the reaction should preferably be carried out in an alkaline medium in the pH range of 8–10. It is this pH range of the medium that promotes intensive interaction of acetic anhydride with cellulose and effective interaction with the amino group, which leads to an increase in the degree of amination;3. The duration of treatment of cotton cellulose with acetic anhydride and the interaction of cellulose acetate with ethylenediamine under optimal conditions takes 4–5 hours;4. One important factor is the selection of the optimal ratio of the starting reagents. The molar ratios of cellulose, acetic anhydride, and ethylenediamine are in the range of 1:1.2:1.5, which achieves the maximum degree of modification.Cotton cellulose modified with ethylenediamine contains amino groups that are highly hydrophilic and water–soluble. Due to the hydrogen bonds between these groups and water molecules, the cellulose can exist in a colloidal or gel–like state. It should be noted that modified cellulose disperses well in water, which helps create favorable conditions for the preparation of various bioactive and sorption materials. Organic compounds containing amino groups are used as functional groups in bioactive substances, pharmaceuticals, film materials, and sorbents. Furthermore, the hydrophilic groups of cellulose can lead to volume expansion and the formation of a gel structure.To establish the composition of the complex, the amount of metal cations and nitrogen in cotton fabric dyed with metal complexes was determined.The determination of cations was carried out by spectrometric titration of salt solutions with a 0.01 M solution of Trilon B (sodium salt of ethylenediaminetetraacetic acid) in the presence of an ammonia buffer solution. When Trilon B interacts with metal cations, almost exclusively 1:1 complexes are formed. With an excess of Trilon B, highly charged cations, such as Fe 3+, also form 1:2 complexes, but their stability is lower than that of 1:1 complexes.As can be seen from the obtained data presented in Table 2, the content of nitrogen and cation contained in complex compounds, depending on the pH of the environment, fluctuates within the range of 10.9–12.3 and 0.9–1.7, respectively.
Without these special conditions, normal aminolysis with deacetylation will predominate.Amination is carried out by the following sequence and reaction conditions:1. The amination reaction is carried out at a temperature of 60–70°C. This temperature range is optimal, as determined by studying the reaction kinetics as a function of temperature. The amination reaction was carried out at a cellulose acetate to EDA ratio of 1:1.2 for 3 hours. The EDA solution concentration is 5%;2. An important factor in the formation of hemiacetals during the amination of cotton cellulose is the pH of the medium. Experimental results showed that the reaction should preferably be carried out in an alkaline medium in the pH range of 8–10. It is this pH range of the medium that promotes intensive interaction of acetic anhydride with cellulose and effective interaction with the amino group, which leads to an increase in the degree of amination;3. The duration of treatment of cotton cellulose with acetic anhydride and the interaction of cellulose acetate with ethylenediamine under optimal conditions takes 4–5 hours;4. One important factor is the selection of the optimal ratio of the starting reagents. The molar ratios of cellulose, acetic anhydride, and ethylenediamine are in the range of 1:1.2:1.5, which achieves the maximum degree of modification.Cotton cellulose modified with ethylenediamine contains amino groups that are highly hydrophilic and water–soluble. Due to the hydrogen bonds between these groups and water molecules, the cellulose can exist in a colloidal or gel–like state. It should be noted that modified cellulose disperses well in water, which helps create favorable conditions for the preparation of various bioactive and sorption materials. Organic compounds containing amino groups are used as functional groups in bioactive substances, pharmaceuticals, film materials, and sorbents. Furthermore, the hydrophilic groups of cellulose can lead to volume expansion and the formation of a gel structure.To establish the composition of the complex, the amount of metal cations and nitrogen in cotton fabric dyed with metal complexes was determined.The determination of cations was carried out by spectrometric titration of salt solutions with a 0.01 M solution of Trilon B (sodium salt of ethylenediaminetetraacetic acid) in the presence of an ammonia buffer solution. When Trilon B interacts with metal cations, almost exclusively 1:1 complexes are formed. With an excess of Trilon B, highly charged cations, such as Fe 3+, also form 1:2 complexes, but their stability is lower than that of 1:1 complexes.As can be seen from the obtained data presented in Table 2, the content of nitrogen and cation contained in complex compounds, depending on the pH of the environment, fluctuates within the range of 10.9–12.3 and 0.9–1.7, respectively.
|
4. Discussion
- The results of the study confirm that the amination of cotton cellulose significantly enhances its physicochemical and sorption properties, providing active sites for metal complex formation and improved dye affinity. The introduction of amino groups into the cellulose matrix increases its capacity to interact with polyvalent metal ions due to the presence of lone electron pairs on nitrogen atoms, which facilitates the formation of coordination bonds. This modification changes the surface polarity and increases the hydrophilicity of the cellulose, promoting better dispersion in aqueous systems and higher dye uptake. The rheological analysis of the prepared dye compositions demonstrated that the viscosity and degree of thixotropic recovery depend on the composition and the presence of modifiers. The developed four-component system based on cellulose, acetic anhydride, ethylenediamine, and triethylamine exhibited a higher degree of thixotropic recovery (up to 94–95%) compared with traditional industrial binders, indicating enhanced structural stability and better suitability for technological processes.The study of reaction mechanisms showed that the acetylation of cellulose with acetic anhydride leads to the formation of reactive acetate intermediates, which subsequently react with ethylenediamine, replacing the acetate group with amino functionalities. This stepwise modification process ensures the stable fixation of amine groups in the cellulose chain, forming strong covalent bonds. The alkaline environment and the presence of triethylamine facilitated the nucleophilic substitution reaction, while maintaining the optimal pH range (8–10) prevented cellulose degradation.The analysis of metal complex formation showed that the degree of coordination depends on the valence and ionic radius of the metal cations. The highest content of metal ions was observed for Cu²⁺ and Ni²⁺, while Fe³⁺ showed a lower incorporation level, which can be explained by steric and electrostatic factors. The nitrogen content in the modified cellulose correlated with the amount of incorporated metal ions, confirming the formation of stable metal–amine complexes. These complexes provided uniform and durable coloration, resistant to washing and environmental influences.The obtained results demonstrate that the combined acetylation and amination processes, followed by dyeing with metal complexes, form a promising technological basis for creating new, efficient, and cost-effective dyeing compositions for the textile industry. The developed method allows replacing imported thickening and binding systems with locally produced polymer compositions, reducing production costs while improving the quality and durability of dyed cotton fabrics. The high rheological stability, strong metal binding capacity, and improved dyeing efficiency make the modified cellulose-based compositions a competitive alternative for industrial application in the production of functional textile materials.
5. Conclusions
- Thus, the treatment of cotton fabric with acetic anhydride and ethylenediamine solution helps to obtain aminated cotton cellulose. When treating modified cotton acetate cellulose with a solution containing a salt of a polyvalent metal, triethylamine and zinc acetylacetonate contribute to improving the protonation of ethylenediamine, as well as stabilizing the pH of the medium, which easily forms colored metal complexes with cations of polyvalent metals.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML