-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Genetic Engineering
p-ISSN: 2167-7239 e-ISSN: 2167-7220
2026; 14(4): 133-136
doi:10.5923/j.ijge.20261404.06
Received: Apr. 5, 2026; Accepted: Apr. 25, 2026; Published: Apr. 29, 2026

Bioclimatic Setting, Vegetation Structure and Entomofaunal Sampling Design in Lower Amudarya Tugay Forests, Khorezm Region, Uzbekistan
Madina Allayarova1, Lola Gandjayeva2, Nazirakhon Allayarova3
1PhD., Department of Natural Sciences, Khorezm Ma’mun Academy, Khiva, Uzbekistan
2DSc., Professor, Department of Chemistry, Tashkent International University of Chemistry, Tashkent, Uzbekistan
3PhD., National Centre for Agricultural Knowledge and Innovation, Research Institute of Plant Genetic Resources, Tashkent, Uzbekistan
Correspondence to: Madina Allayarova, PhD., Department of Natural Sciences, Khorezm Ma’mun Academy, Khiva, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

(1) Background: The tugay riparian forests of Khorezm Region, north-western Uzbekistan, represent one of the most ecologically significant yet poorly inventoried woodland systems in arid Central Asia. (2) Methods: A three-year (2023–2025) entomological survey was conducted across three contrasting localities spanning the principal hydrological gradient of the region. Three operationally defined biotope classes — arboreal (closed-canopy Populus euphratica Oliv.–Salix alba L. stands), shrub (Elaeagnus angustifolia L.–Tamarix ramosissima Ledeb. formations) and meadow (open Cynodondactylon (L.) Pers. associations) — were distinguished on the basis of canopy physiognomy, dominant floristic stratum and soil moisture regime. Assemblages were sampled by sweep-net transects, bark-and-litter hand searching and UV light-trapping; population density was expressed as D = N S⁻¹ (ind. m⁻²) and community diversity was characterised through the Shannon–Wiener entropy (H’), Simpson’s concentration index (D) and the Margalef richness estimator (Mg). (3) Results: The combined dataset yielded 19 species referable to four orders; arboreal biotopes accounted for 57.9% of total species richness, with Shannon diversity ranging from H’ = 2.48 (Shovot) to H’ = 2.71 (Gurlan). (4) Conclusions: The three-class biotope typology and multi-method sampling protocol established here constitute a reproducible entomological baseline for detecting faunal changes associated with continuing hydrological degradation of these threatened ecosystems.
Keywords: Lower Amudarya riparian zone, Tugay entomofauna, Phytophysiognomy-based biotope classification, Standardised insect sampling, Khorezm biodiversity inventory, Aridification monitoring
Cite this paper: Madina Allayarova, Lola Gandjayeva, Nazirakhon Allayarova, Bioclimatic Setting, Vegetation Structure and Entomofaunal Sampling Design in Lower Amudarya Tugay Forests, Khorezm Region, Uzbekistan, International Journal of Genetic Engineering, Vol. 14 No. 4, 2026, pp. 133-136. doi: 10.5923/j.ijge.20261404.06.
Article Outline
1. Introduction
- Notwithstanding the sustained global interest in Central Asian drylands as laboratories for investigating the ecological consequences of water scarcity and land-use intensification, the invertebrate fauna of the region’s riverine woodlands — the so-called tugay — has attracted remarkably little systematic attention compared with the substantial body of work devoted to their floristic composition and structural dynamics [1,2]. This lacuna is particularly conspicuous in the lower Amudarya valley, where five decades of reduced discharge attributable to upstream irrigation abstraction have reduced tugay extent to a fraction of its historical area [2,3], yet where the faunal legacy of that contraction remains largely unquantified.Within the broader context of Central Asian riparian ecology, insects occupy pivotal positions in the trophic architecture of tugay woodlands: as primary consumers of arboreal and herbaceous biomass, as coleopteran and hemipteran predators suppressing herbivore irruptions, and as detritivores and saproxylic taxa mediating the decomposition of woody debris [4]. Their community composition is demonstrably sensitive to the microhabitat complexity of the vegetation matrix, fluctuations in soil moisture and the degree of anthropogenic modification — properties that collectively endow entomofaunal surveys with considerable diagnostic power as instruments of ecosystem health assessment [5]. Earlier reconnaissance surveys in the region [6,7] documented isolated representatives of the principal functional guilds, but none was conducted across a replicated biotope gradient, none extended across multiple growing seasons and none produced the quantitative density and diversity metrics necessary to detect temporal change.The present study — the methodological companion to a parallel faunistic account [Allayarova et al., in press] — addresses this deficit through: (i) the bioclimatic and floristic characterisation of three contrasting tugay localities spanning the principal hydrological gradient; (ii) the establishment of an operationally rigorous, replicable biotope classification grounded in canopy physiognomy, dominant floristic elements and soil moisture regime; and (iii) a transparent account of the sampling and analytical protocol applied across the three-year survey period, with sufficient methodological detail to permit future replication and inter-site benchmarking.
2. Materials and Methods
2.1. Geographic and Climatic Setting
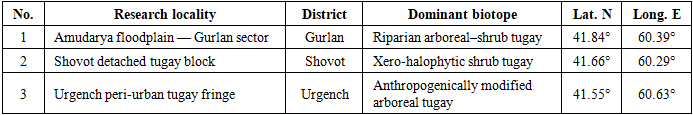
- Khorezm Region (41°30’–41°55’ N, 60°15’–60°45’ E) occupies the north-western extremity of Uzbekistan and is traversed by the lower Amudarya some 300 km above its delta. The climate is emphatically continental: mean annual temperature 12–14°C, with summer diurnal maxima routinely exceeding 40°C and episodic extremes approaching 47°C, set against winter minima of −15 to −20°C; mean annual precipitation does not exceed 80–100 mm, concentrated predominantly in the November–March period, rendering the growing season effectively rainless [7]. Relative humidity fluctuates between 40 and 60%, and spring–summer wind events at 3–5 m s⁻¹ episodically facilitate passive displacement of aerial insects across the landscape [8]. These parameters compress the effective entomological activity window to the five-month interval May–September, which accordingly defined the sampling season throughout the survey.Soils across the research area are predominantly alluvial and sandy-alluvial in texture, with localised occurrence of meadow-saline and takyr-like formations; electrical conductivity varies considerably among biotope types and constitutes a recognised constraint on plant community development and, by extension, on the diversity of phytophage guilds dependent on specific host taxa [9]. Progressive salinisation of formerly productive floodplain soils — a well-documented consequence of irrigation-related waterlogging and subsequent evaporative salt accumulation [10] — represents one of the principal threats to the persistence of structurally diverse tugay in this part of the Amudarya basin. Three research localities were established to span the principal gradient of vegetation integrity and hydrological connectivity within the study region (Table 1).
|
2.2. Vegetation Physiognomy and Biotope Classification
- Recognition of discrete biotope classes for entomological purposes required a classification criterion that was simultaneously ecologically meaningful and operationally reproducible across all three localities. After field reconnaissance during spring 2023, a three-class scheme based on canopy physiognomy, dominant floristic stratum and soil moisture regime was adopted.Arboreal biotope (AB). Defined by the presence of a closed to semi-closed woody canopy (crown cover exceeding 40%), with Populus euphratica Oliv. and/or Salix alba L. (with S. songarica Anderss. as a secondary associate) comprising the dominant stratum. Litter accumulation is appreciable, soil moisture is relatively high owing to capillary rise from the shallow water table and structural complexity — in the sense of the diversity of substrates for bark-associated, wood-boring and foliar guilds — is greatest within this class. Across the combined dataset, the arboreal biotope yielded 57.9% of all species recorded.Shrub biotope (SB). Characterised by the absence of a continuous arboreal canopy and by the dominance of multi-stemmed shrub formations, principally Elaeagnus angustifolia L., Tamarix ramosissima Ledeb. and, locally, Alhagi pseudalhagi (M. Bieb.) Desv. Phytophagous Hemiptera of the families Pentatomidae and Pyrrhocoridae constitute the characteristically dominant guild; this class is most extensively developed at Shovot.Meadow biotope (MB). Open formations of Cynodon dactylon (L.) Pers., Elytrigia repens (L.) Nevski and Bromus inermis Leyss., occupying seasonally or permanently desiccated depressions between woody stands. Litter is negligible, soil moisture is consistently lowest within this class, and the fauna is dominated by acridid Orthoptera.
2.3. Sampling Programme
- All fieldwork was conducted during the growing seasons of 2023, 2024 and 2025, spanning the interval from early May to late September in each year. Each locality received a minimum of five visits per season at approximately four-week intervals, so that phenological stages from vernal emergence to autumnal retraction were adequately represented in the combined sample. Three collection methods were deployed concurrently at each sampling event.Sweep-net sampling was conducted along fixed transects of 10–20 m through the herbaceous and lower shrub stratum, with 10–15 lateral sweeps per transect executed at a standardised pace. This technique efficiently captures soft-bodied active taxa — adults of Pentatomidae, Pyrrhocoridae and Nymphalidae in particular — but systematically under-samples bark-associated and cryptic taxa, a deficiency addressed by the second method.Bark-and-litter hand searching involved direct visual inspection and manual removal of individuals encountered on bark surfaces, woody stem undersides, leaf litter and the soil surface within a defined search area adjacent to each transect. This method is indispensable for the recovery of cerambycid larvae and adults concealed beneath bark, carabid beetles exploiting the litter–soil interface, and sluggish pentatomids resting on abaxial leaf surfaces.UV light-trapping was conducted on two non-consecutive nights per sampling visit, with a portable UV lamp operated from approximately 30 min post-sunset until midnight local time. Night-flying Lepidoptera — Noctuidae principally — are most efficiently sampled by this method; the consistent operational protocol (fixed exposure duration, standardised lamp position at ca. 1.2 m above ground) reduced inter-visit variability attributable to meteorological factors. Soft-bodied and larval material was fixed in 70% ethanol (v/v); adult Coleoptera and macromoths were dry-pinned in the field. All voucher specimens are deposited in the Entomology Reference Collection of the Khorezm Ma’mun Academy, Khiva.
2.4. Specimen Identification and Quantitative Analysis
- Identifications to species level were performed under a stereobinocular microscope (magnification range 10–40×) using the standard determinative keys of Bey-Bienko [13] for general Insecta and those of Kerzhner & Yachevskiy [14] for Heteroptera. Nomenclature was reconciled against the Catalogue of Palaearctic Coleoptera (Löbl & Löbl, 2017) and Fauna Europaea (de Jong et al., v.3).Population density per sampling event was estimated as D = N S⁻¹, where N denotes the total individual count and S denotes the effective swept or searched area (m²; estimated as 2 m² per 10 sweeps for the net method and as the measured search area for hand-searching). Encounter-frequency-based dominance categories followed Pesenko [15]: dominant (>15% of all encounters), subdominant (5–15%) and rare (<5%). Community diversity was quantified by the Shannon–Wiener entropy H’ = −Σ(pᵢ ln pᵢ), Simpson’s concentration D = 1 − Σpᵢ² and the Margalef richness estimator Mg = (S − 1)(ln N)⁻¹. Pearson product-moment correlation (r) and Pearson’s chi-squared (χ²) were used to assess the association between vegetation cover density and species richness, computed in R v.4.3.0.
3. Result and Discussion
- Pooled across localities and seasons, the survey yielded 19 species assignable to four orders (Heteroptera, Coleoptera, Orthoptera, Lepidoptera), eight families and 18 genera — a figure consistent with the species totals recovered from comparably intensive inventories conducted in structurally comparable riparian fragments elsewhere in the Aral Sea basin [6,7]. Site-level species tallies of 15 (Gurlan, 78.9% of total), 14 (Urgench, 73.7%) and 12 (Shovot, 63.2%) declined in parallel with decreasing vegetation structural complexity, a result anticipated on theoretical grounds given the well-documented positive scaling of arthropod diversity with habitat heterogeneity in woody riparian systems.Within the biotope framework, the arboreal class was overwhelmingly the most species-rich, accounting for 11 species (57.9%), compared with five in the shrub biotope (26.3%) and three in the meadow biotope (15.8%). The fourfold excess of arboreal over meadow species richness is attributable, at least in part, to the structural complexity of tall-canopy stands, which simultaneously offer bark and cambium as substrates for xylophagous guilds, foliar resources for pentatomids and lepidopteran larvae, litter and soil for carabid and saprophagous taxa, and aerial space for active fliers. Shannon diversity ranged from H’ = 2.48 (Shovot) to H’ = 2.71 (Gurlan), with Simpson indices of 0.84–0.89 indicating moderate to high evenness at all sites.Notably, despite the appreciable anthropogenic disturbance documented at Urgench, the site retained 93% of the species recorded at the near-pristine Gurlan locality — a result suggesting that even structurally degraded but topographically connected tugay fragments retain disproportionate entomological value and merit active protection rather than abandonment. This finding has direct implications for regional conservation planning, arguing for the retention and active restoration of shrub and tree cover even in heavily modified riparian corridors [11].The methodological combination deployed here — sweep-net transects, bark searching and UV trapping — proved effective across all three activity guilds encountered. The fixed-transect design, with a standardised sweep count and consistently defined search area, confers inter-visit and inter-locality comparability; the three-year replication provides a phenological baseline that single-season surveys cannot supply [12]. Full species accounts and seasonal dynamics are reported in the companion paper.
4. Conclusions
- The lower Amudarya tugay of Khorezm Region presents a thermally extreme, precipitation-deficient continental environment in which insect activity is confined to a brief but intense growing season, and in which the structural quality of the woody vegetation mosaic emerges as the principal determinant of local species richness. The three-class biotope typology developed here — arboreal, shrub and meadow — captures the principal gradient of microhabitat complexity across the study region and provides an operationally transparent basis for future comparative work. The multi-method sampling protocol, applied across five visits per season over three years, constitutes a reproducible entomological baseline for detecting the anticipated faunal changes associated with continuing hydrological degradation and land-use transformation in these threatened riparian woodlands.
ACKNOWLEDGEMENTS
- The fieldwork reported here was conducted with the logistical support of the Khorezm Ma’mun Academy and benefited from the assistance of the laboratory staff of the Research Institute of Plant Genetic Resources in specimen curation. The authors declare no competing interests.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML