-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Genetic Engineering
p-ISSN: 2167-7239 e-ISSN: 2167-7220
2026; 14(4): 113-116
doi:10.5923/j.ijge.20261404.03
Received: Feb. 26, 2026; Accepted: Mar. 19, 2026; Published: Apr. 7, 2026

The Immunomodulatory and Metabolic Effects of Chlorella Vulgaris Biologically Active Compounds on Experimental Models and Bovine Physiology
Maxliyo M. Teshajonova
Department of Medical Chemistry and Biological Sciences, Central Asian Medical University, Fergana, Uzbekistan
Correspondence to: Maxliyo M. Teshajonova, Department of Medical Chemistry and Biological Sciences, Central Asian Medical University, Fergana, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This study investigates the efficacy of biologically active substances (BAS) derived from Chlorella vulgaris in correcting secondary immunodeficiency induced by toxic hepatitis (CCl4 and D-galactosamine) in laboratory rats and its metabolic impact on calves. Results indicated that a 1.0% concentration of Chlorella BAS significantly restored antibody-forming cells (AFC) in the spleen, comparable to the reference drug Thymalin. In calves, parenteral administration improved T and B-lymphocyte counts and increased body weight gain (up to 62 kg). The findings suggest that Chlorella vulgaris acts as a potent biostimulator of hematopoiesis and protein-carbohydrate metabolism.
Keywords: C. vulgaris, Bioactive compounds, Immunomodulation, Toxic hepatitis, Bovine physiology, Hematopoiesis, Metabolic status
Cite this paper: Maxliyo M. Teshajonova, The Immunomodulatory and Metabolic Effects of Chlorella Vulgaris Biologically Active Compounds on Experimental Models and Bovine Physiology, International Journal of Genetic Engineering, Vol. 14 No. 4, 2026, pp. 113-116. doi: 10.5923/j.ijge.20261404.03.
Article Outline
1. Introduction
- The increasing impact of anthropogenic factors and xenobiotics has led to widespread physiological disruptions in livestock, particularly affecting liver function and immune response. Toxic hepatitis often results in secondary immunodeficiency, reducing animal productivity and increasing susceptibility to infectious diseases. [3]. While synthetic drugs are commonly used, their side effects necessitate the search for natural alternatives. [13]. Chlorella vulgaris is a unicellular green alga rich in proteins (50–55%), vitamins (A, B-group, C, E), and essential minerals. [1]. Its unique "Chlorella Growth Factor" (CGF) and polysaccharides exhibit potent antioxidant and immunomodulatory properties. [2, 11]. This study aims to scientifically substantiate the use of local Chlorella vulgaris strains as biostimulators for restoring physiological and immunological status.
2. Materials and Methods
2.1. Experimental Design
- The research was conducted on outbred white mice (18–22 g), white rats (180–200 g), and young calves. Toxic hepatitis was induced in mice using a CCl4 oil solution (0.2 mg/kg, i.p. for 3 days) and in rats using D-galactosamine hydrochloride (1.1 g/kg, i.p.). [15].
2.2. Immunological and Biochemical Analysis
- Antibody-forming cells (AFC) in the spleen were determined using the local hemolysis method in agarose (Jerne and Nordin). Blood parameters (erythrocytes, leukocytes) were analyzed using a Goryayev chamber. Biochemical markers (ALT, AST, protein fractions, glucose, and minerals) were measured in blood serum using standard diagnostic kits. [23].
3. Results and Discussion
3.1. Impact on Experimental Immunodeficiency
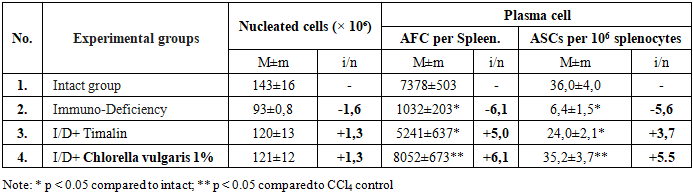
- Induction of hepatitis led to a 2.7-fold decrease in AFC production in the spleen (5,334 ± 725 vs. 14,621 ± 1,029 in intact mice). Administration of Chlorella vulgaris BAS resulted in a significant recovery, increasing AFC counts by 2.5 to 6.1 times depending on the concentration (Table 1). [8,17].
|
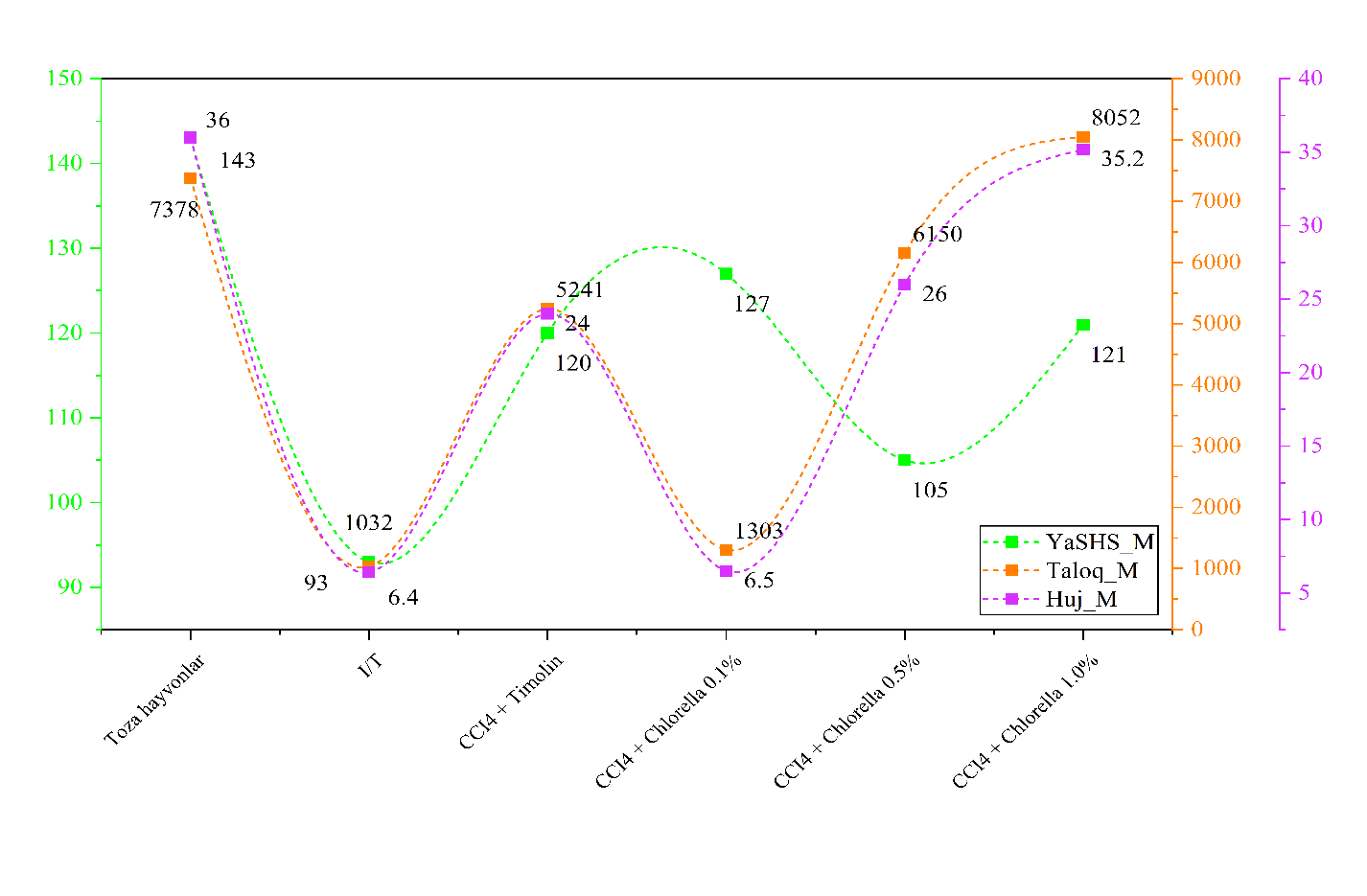 | Figure 1 |
3.2. Physiological Status in Calves
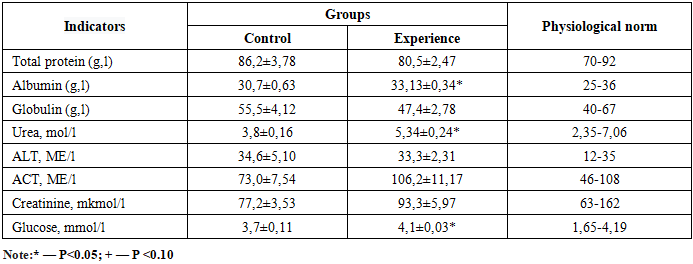
- Parenteral administration of Chlorella to calves stimulated T-lymphocyte (increase of 1.3–3.1%) and B-lymphocyte (1.5% increase) populations. [14,18].Significant improvements were noted in protein metabolism: albumin levels reached 33.13 ± 0.34 g/l in the experimental group compared to the control (P < 0.05). Glucose levels increased by 12.3%, indicating enhanced energy homeostasis (Table 2).
|
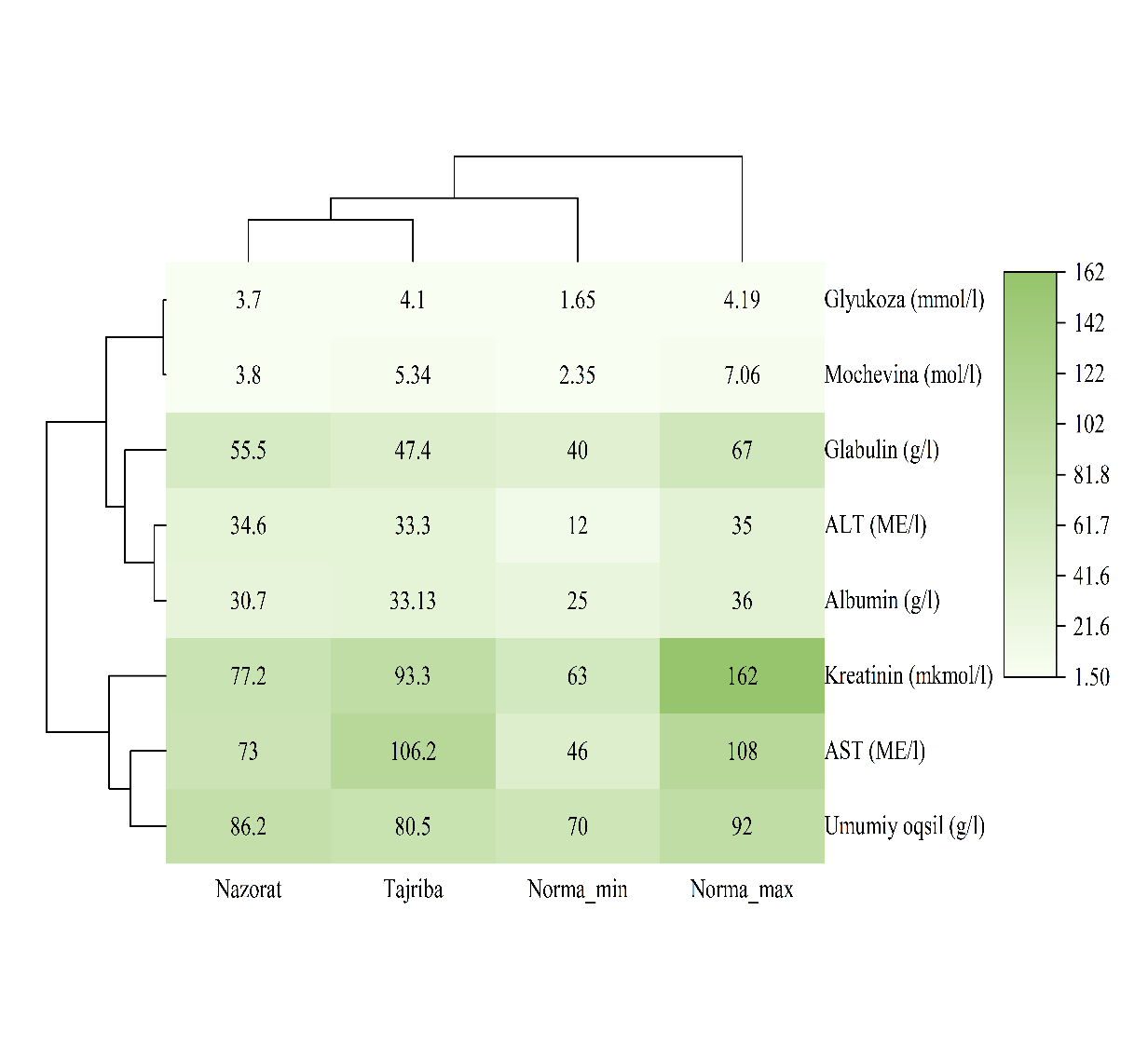 | Figure 2. Dynamics of protein and carbohydrate metabolism under the influence of the drug |
4. Analysis of Protein and Carbohydrate Metabolism
- Biochemical profiling of blood serum demonstrated a distinct predominance of anabolic processes in the experimental group administered with Chlorella vulgaris suspension. Specifically, serum albumin levels in the experimental group exhibited a statistically significant increase (P < 0.05) compared to the control, reaching 33.13 ± 0.34 g/L. This elevation in albumin suggests a robust stimulation of the liver's protein-synthetic function. This physiological shift is attributed to the direct systemic bioavailability of the preparation’s biologically active compounds, specifically bioactive peptides and essential amino acids, which facilitate enhanced hepatic proteosynthesis and optimize molecular transport mechanisms. [24].The protein index (albumin/globulin ratio) reached 0.68 in the experimental cohort, whereas it remained at 0.58 in the control group. This divergence indicates an improved potential for replenishing systemic protein reserves and a superior level of cellular supply with essential plastic materials. [22].Furthermore, a positive trajectory was observed in energy metabolism markers. Glucose concentrations in the experimental group increased by 12.3% (0.45 mmol/L) relative to the control (P < 0.05), signifying an enhanced energetic supply to peripheral tissues and improved glucose homeostasis.The rise in creatinine levels from 77.2 µmol/L (control) to 93.3µmol/L (experimental) indicates an accelerated metabolic turnover within muscle tissues under the influence of the biostimulant. [22]. This correlates with an expansion of the phosphocreatine pool, a vital energy accumulator. Such an increase within the physiological reference range is of paramount importance for ensuring efficient ATP resynthesis and maintaining high stability in intracellular bioenergetics.
5. Conclusions
- The comprehensive physiological and immunological assessment of Chlorella vulgaris biologically active substances (BAS) allows for the following conclusions:1. Potent Immunomodulatory Efficacy: It was established that Chlorella vulgaris biomass serves as a high-efficiency biostimulant for correcting secondary immunodeficiency induced by hepatotropic toxins (CCl4 and D-galactosamine). The administration of a 1.0% suspension demonstrated a superior restorative effect on the splenic immune microenvironment, increasing the population of antibody-forming cells (AFC) by 6.1 times, effectively neutralizing the immunosuppressive effects of toxic hepatitis.2. Hematopoietic and Regenerative Stimulation: The preparation exhibits a pronounced hematopoietic effect, specifically stimulating erythropoiesis and leucopoiesis within the bone marrow. This leads to a stabilized balance of peripheral blood formed elements, ensuring systemic homeostasis without inducing pathological leucocytosis.3. Metabolic Optimization in Livestock: Parenteral administration of Chlorella BAS in calves and cows significantly enhances anabolic processes. The observed increase in serum albumin (33.13 ± 0.34 g/L) and glucose levels (12.3%) indicates an optimization of hepatic proteosynthesis and energy metabolism. Furthermore, the stabilization of the creatinine-phosphocreatine pool confirms improved intracellular bioenergetics and ATP resynthesis capacity.4. Practical Applicability: Given its high biological activity, cost-effectiveness, and absence of adverse side effects compared to synthetic analogs (e.g., Thymalin), the locally derived Chlorella vulgaris strain is a viable candidate for integration into veterinary pharmacology. Its use is recommended for enhancing the nonspecific resistance and productive potential of agricultural animals, particularly in ecologically unfavorable regions.In summary, this research scientifically substantiates Chlorella vulgaris as a multifaceted biostimulant that effectively bridges the gap between hepatoprotection and immunocorrection in animal physiology.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML