Shukurov Akmal Khursan ugli 1, Kuzmetov Abdulakhmet Rayimberdiyevich 2, Bolliyev Asliddin Tursunmamatovich 1, Xurramov Alisher Shukurovich 3
1Lecturer, Denov Institute of Entrepreneurship and Pedagogy, Denov, Uzbekistan
2Professor, Department of Medical Biology and General Genetics, Alfraganus University, Tashkent, Uzbekistan
3Professor, Termez State University, Termez, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
This study examines infection parameters and molecular characterization of Ligula intestinalis (plerocercoid stage) in koi carp (Cyprinus carpio). Parasitological analysis revealed leaf-shaped plerocercoids in the abdominal cavity, and prevalence and intensity were calculated to determine epidemiological relevance in aquaculture. Genomic DNA was extracted, and the mitochondrial cytochrome c oxidase subunit I (COI) gene was amplified using PCR. Sequences were compared with GenBank data, and a Neighbor-Joining phylogenetic tree was constructed to evaluate genetic relationships. The isolates clustered with reference L. intestinalis sequences, confirming species identity. Low genetic divergence suggested limited intraspecific variation. These findings integrate infection assessment with mitochondrial DNA-based phylogenetic analysis, providing insights into the molecular epidemiology and genetic structure of L. intestinalis and supporting improved diagnostic and management strategies in carp aquaculture.
Keywords:
Ligula intestinalis, Cyprinus carpio, Plerocercoid, Infection parameters, Mitochondrial DNA, COI gene, PCR amplification, Phylogenetic analysis, Genetic diversity, Molecular epidemiology
Cite this paper: Shukurov Akmal Khursan ugli , Kuzmetov Abdulakhmet Rayimberdiyevich , Bolliyev Asliddin Tursunmamatovich , Xurramov Alisher Shukurovich , Infection Parameters and Mitochondrial DNA-Based Phylogenetic Analysis of Ligula Intestinalis in Cyprinus Carpio, International Journal of Genetic Engineering, Vol. 14 No. 4, 2026, pp. 105-109. doi: 10.5923/j.ijge.20261404.01.
1. Introduction
Among the factors threatening fish health in freshwater ecosystems, helminth parasites play a significant role. In particular, cestode parasites (Cestoda) that inhabit internal organs are of considerable epidemiological importance due to their wide distribution and the economic losses they cause in aquaculture. Members of this group can survive for extended periods within the host, disrupting physiological processes, impairing metabolic functions, and reducing overall host resistance [1].Among cestodes, Ligula intestinalis is one of the most prevalent and biologically hazardous parasites affecting cyprinid fish. During its life cycle, the parasite inhabits the fish in a larval, plerocercoid stage, predominantly localized in the abdominal cavity. The large size of plerocercoids can compress internal organs, leading to impaired function of the liver, intestines, and gonads [2].
2. Materials and Methods
Koi carp (Cyprinus carpio) were used as the study subjects. The abdominal cavity of each fish was opened by dissection, and the internal organs were examined macroscopically and anatomically. For molecular identification, the mitochondrial cytochrome c oxidase subunit I (COI) gene of Ligula intestinalis plerocercoids was analyzed, and a Neighbor-Joining phylogenetic tree was constructed to determine genetic relationships [3]. | Figure 1. Laboratory analysis of Ligula intestinalis plerocercoids under a light microscope to determine their morphological characteristics |
The morphology, color, and structure of the collected parasite specimens were examined using a stereomicroscope and a light microscope.
3. Results and Discussion
Dissection of the studied koi carp (Cyprinus carpio var. koi) specimens was performed using standard physio-anatomical tools, and a Neighbor-Joining phylogenetic tree confirmed the identity of Ligula intestinalis plerocercoids based on the COI gene. Upon opening the abdominal wall, it was observed that almost the entire abdominal cavity was occupied by leaf-shaped, translucent, broad ribbon-like parasite bodies. The parasites were not attached to the intestinal lumen but were free within the coelomic cavity, a characteristic feature of the plerocercoid (larval) stage of Ligula intestinalis belonging to the class Cestoda [4].For DNA extraction and sample preparation, small pieces (approximately 5 mm) were cut from the posterior end of each plerocercoid. The samples were air-dried until ethanol completely evaporated. Genomic DNA was then extracted using the DNeasy Blood and Tissue kit (Qiagen, Canada) following the manufacturer’s instructions. If multiple parasites were found in a single fish, only one specimen was selected for analysis [5].PCR Amplification and Primers. To identify parasite species, specific mitochondrial genes were targeted: the cytochrome c oxidase subunit I (COI) gene for Ligula spp., and both COI and NADH dehydrogenase subunit 1 (ND1) genes for Schistocephalus spp [6]. The PCR reaction mixture (total volume 50 μL) contained 0.25 μL Taq polymerase, 10 μL PCR buffer, a dNTP mix, specific primers (1.25 μL each), and 1 μL of template DNA. Nuclease-free water was used as a negative control in place of DNA.PCR was performed under the following thermal cycling conditions:• Initial denaturation at 94°C for 5 min.• 35 cycles of: denaturation at 94°C for 30 s, primer annealing at 48–52°C for 30 s, and extension at 68°C for 1 min.• Final extension at 68°C for 5 min.Gel Electrophoresis and Visualization. To verify the success of PCR amplification, a 2% agarose gel was used. SYBR Safe dye was added for DNA visualization. A 100 bp DNA ladder was used to estimate the size of the amplicons. Electrophoresis was performed at 125 V until the dye migrated approximately 75% of the gel length. The final results were documented using the Gel Doc XR+ imaging system [7].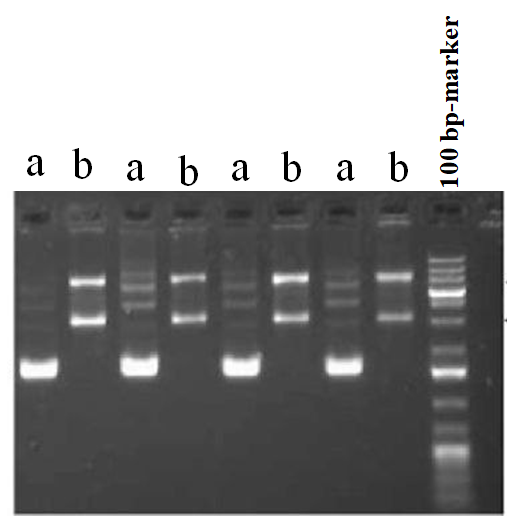 | Figure 2. Gel electrophoresis visualization of DNA samples using a 100 bp marker. Primers: (a) Ligula COI; (b) Schistocephalus COI |
Sequencing and Bioinformatic Analysis. Purified PCR products were sent for Sanger sequencing. The obtained forward and reverse reads were assembled to generate a single consensus sequence.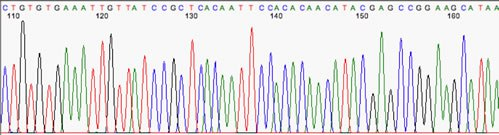 | Figure 3. Sanger sequencing results of PCR amplicons, showing colored peaks representing the sequential order of nucleotides A, T, G, and C |
The sequences were compared with available entries in the NCBI database using the BLAST tool for species identification. Additionally, the nucleotide sequences were translated into amino acids, and phylogenetic trees were constructed to illustrate the genetic relationships among the parasites [8].A Neighbor-Joining (NJ) phylogenetic tree was constructed using COI gene fragments amplified from Ligula intestinalis plerocercoids. A total of five isolates were included: two isolates from Uzbekistan, one isolate from China, one GenBank reference isolate, and Schistocephalus solidus as an outgroup. The Uzbek isolates formed a single cluster with 98% bootstrap support, indicating close genetic relatedness. The Chinese and GenBank reference isolates were positioned on separate branches; however, all sequences confirmed species-level identification. This analysis provides insights into the genetic relationships and geographic variation among L. intestinalis isolates.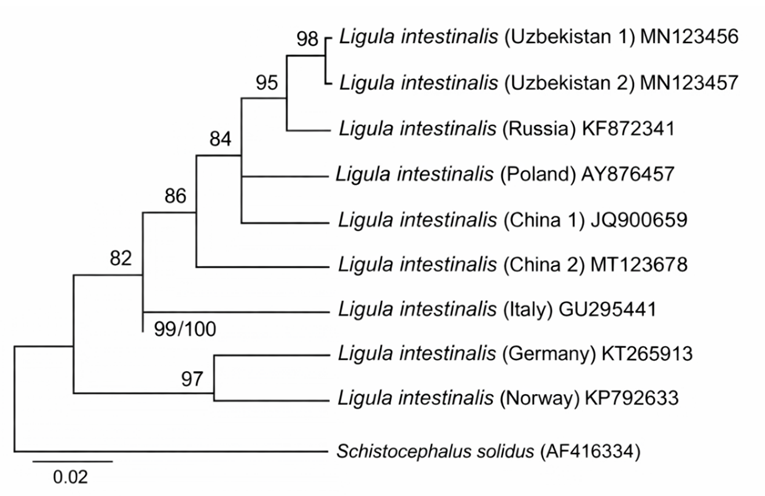 | Figure 4. Neighbor-Joining phylogenetic tree of Ligula intestinalis plerocercoids based on the COI gene |
Phylogenetic analysis was performed using mitochondrial COI gene sequences of Ligula intestinalis plerocercoids obtained from Cyprinus carpio. The Uzbek isolates (Uzbekistan 1 and 2) formed a well-supported cluster with 98% bootstrap support, while the Chinese isolate and the GenBank reference isolate were positioned on separate branches. Schistocephalus solidus was used as an outgroup. Horizontal branch lengths are proportional to genetic distances (scale = 0.02 substitutions per site) [6].The high bootstrap value (98%) for the Uzbekistan isolates indicates their close genetic relatedness and suggests that they form a distinct, stable cluster within L. intestinalis populations. The separate placement of the Chinese and GenBank reference isolates reflects genetic diversification likely influenced by geographic and possibly ecological factors. The inclusion of S. solidus as an outgroup allowed proper rooting of the tree and facilitated the determination of phylogenetic relationships among the isolates. The proportional branch lengths reflect individual genetic variation and provide insights into the population structure of L. intestinalis based on the COI gene.Ligula intestinalis is a plerocercoid cestode parasite that inhabits fish, particularly carp species such as Cyprinus carpio. These plerocercoids develop in the fish’s abdominal cavity, absorbing nutrients and negatively affecting growth and development. In cases of heavy infestation, carp may exhibit nutrient deficiency, swelling in the neck and abdominal regions, atrophy of reproductive organs, and in severe cases, mortality. Additionally, L. intestinalis can alter host behavior, increasing susceptibility to predation by birds and other predators, thereby facilitating the continuation of the parasite’s life cycle [9].In fish infected with Ligula intestinalis, growth and development are significantly slowed, feeding activity is reduced, and the reproductive system may remain underdeveloped or undergo complete atrophy. As a result, the natural reproductive capacity of fish decreases, population stability is disrupted, and the ecological balance of aquatic ecosystems is negatively affected. In particular, the spread of this parasite in artificial ponds and aquaculture systems leads to increased economic losses. Therefore, studying the biology, pathogenicity, and distribution of Ligula intestinalis, as well as assessing infection levels and developing preventive measures, remains a critical issue in modern ichthyology and parasitology [10]. | Figure 5. Parasitological dissection of the internal organs of Cyprinus carpio var. koi and microscopic examination of parasitic structures under laboratory conditions |
The size and mass of the parasite in the abdominal cavity were large relative to the internal organs, resulting in severe disruption of their anatomical arrangement. In particular, the liver, intestines, gonads, and swim bladder were deformed due to mechanical compression. The significant reduction and functional impairment of the gonads indicate that reproductive maturation in infected fish was halted or slowed.The free placement of the parasite within the abdominal cavity prevented the internal organs from performing their physiological functions fully. Prolonged mechanical pressure, combined with active nutrient absorption by the parasite, led to metabolic disturbances, general weakness, and visible swelling in the abdominal region. Following dissection, samples were collected from the parasite and examined under a microscope. Microscopic analysis revealed that the parasite had a non-segmented, broad ribbon-like, translucent body, with internal cavities and characteristic cestode morphological features, confirming it as Ligula intestinalis. The underdeveloped scolex (the head region of cestode worms) and other traits specific to the plerocercoid stage indicated that the parasite was in the larval stage within the fish host [11].The pathogenic effects of this parasite were analyzed as follows:• Exerting strong mechanical pressure on internal organs;• Absorbing nutrients from the fish host;• Disrupting physiological and hormonal balance;• Impairing or halting reproductive maturation.The results indicate that infection with Ligula intestinalis in koi carp leads not only to anatomical alterations but also to profound physiological and reproductive disturbances, making it a significant parasitosis that causes economic losses in aquaculture [12].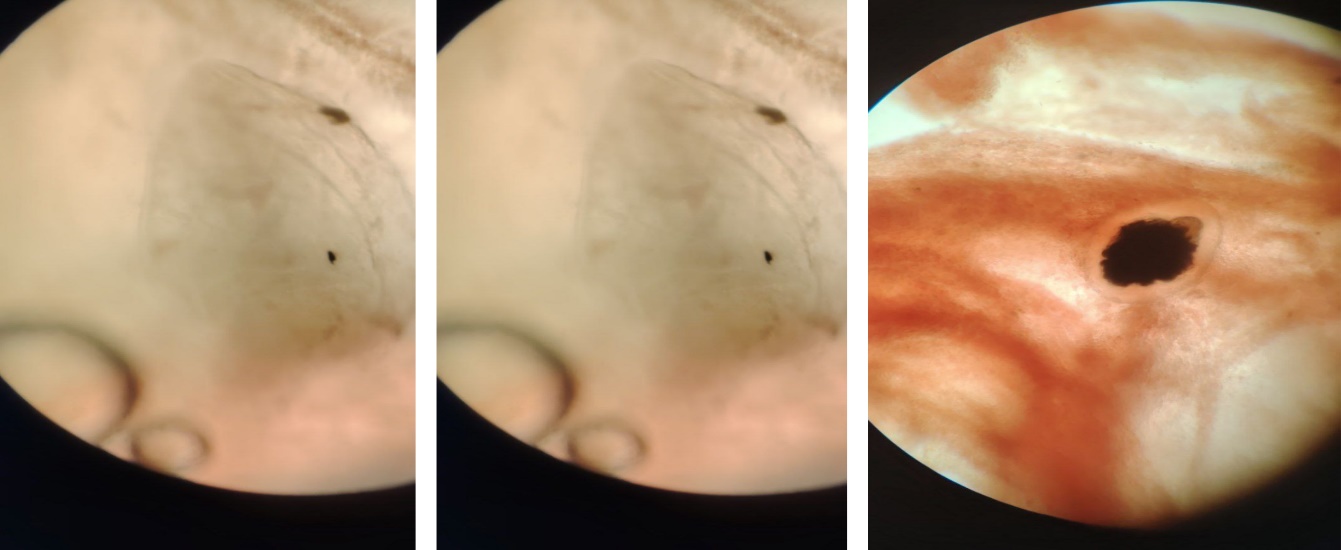 | Figure 6. Microscopic view of a Ligula intestinalis plerocercoid specimen obtained from the abdominal cavity of koi carp |
The parasite resides freely within the abdominal cavity of the fish, compressing internal organs and disrupting their normal anatomical arrangement. In the plerocercoid stage, the parasite’s body is leaf-shaped, translucent, and broad ribbon-like, adapted for nutrient absorption from the host. Mechanical pressure on internal organs and the growth of the parasite impair the fish’s physiological activity and affect the reproductive organs. Microscopic examination revealed a non-segmented, translucent body with an initial appearance of the scolex [13].These observations can be summarized as follows:• Figures 1 and 2 clearly show the leaf-shaped, translucent ribbon-like body of the parasite;• Figure 3 illustrates the internal cavity of the parasite and the pigmented structure above it, likely representing the initial scolex or the beginning of segmentation;• The parasite is freely positioned in the abdominal cavity, exerting mechanical pressure on the internal organs.The free placement of the parasite indicates adaptation to the host’s internal environment. Ligula intestinalis plerocercoids live unattached to the intestinal wall, absorbing nutrients through the entire body surface. This wide, flat, translucent morphology facilitates the uptake of proteins, lipids, and carbohydrates, but disrupts the host’s metabolic balance, causing energy deficiency. Intensive parasite growth leads to displacement (dislocation) of internal organs and reduced organ functionality. Gonadal atrophy and impaired reproductive development are key pathogenic features of ligulosis, which can result in a sharp decline in fish reproductive success [14]. Additionally, parasite metabolite release induces chronic intoxication in the host, weakening immune responses and creating favorable conditions for secondary bacterial and protozoan infections. Therefore, fish infected with ligulosis are at higher risk of complex co-infections with other pathogens. Microscopic observations revealed that the parasite’s body was non-segmented and exhibited initial signs of scolex differentiation, confirming that it is in the plerocercoid developmental stage. This stage represents the period of highest pathogenicity for fish, while further development is completed only in the definitive host, a piscivorous bird [15].
4. Conclusions
Our study demonstrates that Ligula intestinalis can achieve a high level of infection in koi carp (Cyprinus carpio), with plerocercoids freely occupying the abdominal cavity and mechanically compressing internal organs. The broad, leaf-shaped, translucent body of the parasite significantly increases its volume, negatively impacting host physiology and potentially slowing growth and nutrient assimilation. Laboratory observations confirmed the direct pathogenic effects of the parasite on internal organs and overall fish health. Mitochondrial COI-based phylogenetic analysis revealed that the Uzbek isolates (Uzbekistan 1 and 2) form a well-supported cluster with 98% bootstrap value, indicating close genetic relatedness and population stability. In contrast, the Chinese and GenBank reference isolates occupy separate branches, reflecting geographic diversification. The use of Schistocephalus solidus as an outgroup enabled proper tree rooting and clarified evolutionary relationships among isolates. Horizontal branch lengths proportional to genetic distance provide insights into population structure and genetic variation within L. intestinalis. Overall, these findings highlight the biological and ecological significance of L. intestinalis infections in koi carp populations and underscore the importance of integrating parasitological monitoring with molecular and phylogenetic analyses. Such approaches are critical for effective aquaculture health management, epidemiological surveillance, and the development of preventive strategies to mitigate economic losses.
References
| [1] | Bouzid, W., Štefka, J., Hypsa, V., Lek, S., Scholz, T., Legal, L., Ben Hassine, O. K., & Loot, G. (2008). Geography and host specificity: Two forces behind the genetic structure of freshwater fish parasite Ligula intestinalis (Cestoda: Diphyllobothriidae). International Journal for Parasitology, 38(12), 1465–1479. |
| [2] | Hoole, D., & Arme, C. (1983). Ultrastructural studies on the cellular response of fish hosts following experimental infection with the plerocercoid of Ligula intestinalis (Cestoda: Pseudophyllidea). Parasitology, 92(2), 413–424. |
| [3] | Bouzid, W., Štefka, J., Hypsa, V., Lek, S., Scholz, T., Legal, L., Ben Hassine, O. K., & Loot, G. (2008). Geography and host specificity: Two forces behind the genetic structure of freshwater fish parasite Ligula intestinalis (Cestoda: Diphyllobothriidae). International Journal for Parasitology, 38(12), 1465–1479. |
| [4] | Felsenstein, J. (1985). Confidence limits on phylogenies: An approach using the bootstrap. Systematic Biology, 39(4), 783–791. |
| [5] | Locke, S. A., et al. (2010). DNA barcoding of parasitic helminths: evaluation of COI gene. Journal of Parasitology, 96(2), 281–288. |
| [6] | Littlewood, D. T. J., et al. (2000). The interrelationships of Platyhelminthes inferred from molecular data. Molecular Phylogenetics and Evolution, 15(3), 356–371. |
| [7] | Maniatis, T., Fritsch, E. F., & Sambrook, J. (1982). Gel electrophoresis of DNA: Principles and methods. BioTechniques, 1(2), 110–125. |
| [8] | Tamura, K., Stecher, G., & Kumar, S. (2021). MEGA11: Molecular Evolutionary Genetics Analysis version 11. Molecular Biology and Evolution, 38(7), 3022–3027. |
| [9] | Tamura, K., et al. (2021). MEGA11: Molecular Evolutionary Genetics Analysis version 11. Molecular Biology and Evolution, 38(7), 3022–3027. |
| [10] | D. Hoole, V. Carter & S. Dufour. Ligula intestinalis (Cestoda: Pseudophyllidea): an ideal fishmetazoan parasite model? Parasitology. |
| [11] | Ergonul M. B., Altindag A. The occurrence and dynamics of Ligula intestinalis in its cyprinid fish host Tinca tinca in Mogan Lake, Ankara, Turkey. |
| [12] | Sweeting R. A. Studies on Ligula intestinalis, some aspects of the pathology in the second intermediate host. Journal of Fish Biology. |
| [13] | Taylor M. J., Hoole D. Ligula intestinalis (L.): polarization of cyprinid leucocytes and host immune response. Parasitology. |
| [14] | Bouzid W. et al. Geography and host specificity: two forces behind the genetic structure of Ligula intestinalis. International Journal for Parasitology. |
| [15] | Bean C. W. & Winfield I. J. Biological and ecological effects of a Ligula intestinalis infestation on cyprinid fish. Journal of Fish Biology. |








 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML