-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Genetic Engineering
p-ISSN: 2167-7239 e-ISSN: 2167-7220
2026; 14(3): 77-82
doi:10.5923/j.ijge.20261403.01
Received: Feb. 12, 2026; Accepted: Mar. 8, 2026; Published: Mar. 17, 2026

Assessment of Gametophyte Stability and Seed Index Variability in Cotton Germpasm Accessions for Advanced Breeding Programs
L. A. Azimova1, 2, D. K. Ernazarova3, 4, B. O. Ochilov5, 6, M. D. Kholova1, M. Sh. Bosimov6, K. K. Boboniyazov6, F. N. Kushanov3, 4
1PhD Candidate, Institute of Genetics and Plant Experimental Biology, Academy of Sciences of Uzbekistan, Tashkent, Uzbekistan
2Assistant Lecturer, Impuls Medical Institute Chirchiq Branch, Tashkent, Uzbekistan
3Doctor of Biological Sciences, Institute of Genetics and Plant Experimental Biology, Academy of Sciences of Uzbekistan, Tashkent, Uzbekistan
4Professor, National University of Uzbekistan, Tashkent, Uzbekistan
5Senior Lecturer, Impuls Medical Institute Chirchiq branch, Tashkent, Uzbekistan
6Senior Lecturer, Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: L. A. Azimova, PhD Candidate, Institute of Genetics and Plant Experimental Biology, Academy of Sciences of Uzbekistan, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This study aimed to evaluate the cyto-embryological stability and agro-morphological characteristics of various Gossypium hirsutum genotypes, including semi-wild subspecies and modern cultivars, in order to determine their reproductive efficiency and potential value for breeding programs. Significant genotypic variation was detected (P<0.05). The cultivar Ravnak 1 demonstrated the highest reproductive performance, exhibiting an ovule penetration rate of 94.52 ± 0.42 and pollen fertility 96.78±0.94, indicating high stability of male gametophyte. In contrast, subsp. purpurascens showed the lowest fertility indicators. Meanwhile subsp. gambia demonstrated moderate meiotic stability with a meiotic index of 90.39 ± 1.19, despite the occurrence of micronucleated tetrads and polyads (3.14% and 6.47%, respectively). The analysis of the 1000-seed weight revealed substantial variability among genotypes, ranging from 81.78 ± 1.25 g in subsp. gambia to 133.89 ± 1.16 g in cultivar Ravnak 2. The genotype Genofond 2 exhibited remarkable population uniformity with a coefficient of variation of V=1.07, indicating high genetic stability. Overall, the obtained results demonstrate that the efficiency of pollen tube penetration and seed index stability can serve as reliable indicators for selecting highly productive cotton genotypes. These findings emphasize the importance of integrating reproductive biology parameters into cotton breeding programs aimed at developing high-yielding and genetically stable cultivars.
Keywords: Cotton (Gossypium hirsutum L.), Pollen-ovule interaction, Male gametophyte fertility, Meiotic index, ANOVA, Micronucleated tetrads, Seed index, Cotton breeding
Cite this paper: L. A. Azimova, D. K. Ernazarova, B. O. Ochilov, M. D. Kholova, M. Sh. Bosimov, K. K. Boboniyazov, F. N. Kushanov, Assessment of Gametophyte Stability and Seed Index Variability in Cotton Germpasm Accessions for Advanced Breeding Programs, International Journal of Genetic Engineering, Vol. 14 No. 3, 2026, pp. 77-82. doi: 10.5923/j.ijge.20261403.01.
1. Introduction
- Cotton is one of the most important industrial crops worldwide, serving as a major source of natural fiber and playing a crucial role in the global textile industry. According to recent estimates, global cotton production exceeds 25 million tons annually, highlighting its significant economic value and widespread cultivation in many regions of the world [5]. At present, cotton is grown on all continents except Antarctica, reflecting its high adaptability to diverse climatic conditions [1]. Among cultivated cotton species, Gossypium hirsutum L. is the most widely distributed and economically important. In its wild form, however, the natural distribution range of this species has considerably decreased. Currently, wild populations are mainly found in limited geographical areas such as the northern Yucatán Peninsula, Florida, southeastern Puerto Rico, and several islands of the Caribbean region [8]. The study of wild and semi-wild cotton forms is particularly important because they represent valuable sources of genetic diversity that can be utilized in breeding programs aimed at improving agronomic traits and environmental adaptability.Embryological studies in angiosperms play a fundamental role in understanding the mechanisms of gametogenesis, fertilization, and early embryo development. One of the most critical stages of plant reproduction is the growth of pollen tubes through maternal tissues, which ensures the successful delivery of male gametes to the embryo sac. During fertilization, pollen tubes pass through three main anatomical structures: the stigma, the style, and the ovules located within the ovary. This precisely regulated process enables the transport of sperm cells to the egg cell and central cell, ultimately leading to double fertilization and seed formation [4].The directional growth of pollen tubes toward the ovary is regulated by chemotropic signaling mechanisms. In cotton plants, the maturation of male gametes is completed within the pollen tubes during their elongation through the transmitting tissue of the style. Environmental factors, particularly temperature, also influence pollen tube growth. In most plant species, optimal pollen tube elongation occurs within the temperature range of 20–30°C, although species with delayed flowering periods may exhibit adaptation to lower temperatures. Furthermore, pollen tube growth rates vary considerably among plant taxa and are influenced not only by chemical signals but also by mechanical interactions with surrounding tissues. In this context, pollen tubes may respond to differences in tissue rigidity, a phenomenon referred to as durotropism, which facilitates efficient navigation toward the embryo sac [10].Recent studies have demonstrated that the kinetics of pollen tube elongation plays a decisive role in successful fertilization. For example, Wang et al. (2023) reported that in certain Malvaceae species, failure of pollen tubes to reach the embryo sac within 24 hours often results in seed abortion [12]. In addition, previous embryological investigations conducted by V.A. Rumi provided detailed insights into the morphometric characteristics and elongation dynamics of pollen tubes during the fertilization process in flowering plants. These studies highlighted the importance of pollen tube growth dynamics in determining reproductive success. Furthermore, stigmatic secretions have been shown to influence pollen germination and pollen tube development, emphasizing the complex biochemical interaction between the male gametophyte and maternal tissues [3].In cotton breeding research, yield-related traits such as the weight of seed cotton per boll and the 1000-seed weight (seed index) are considered key agronomic indicators. These parameters are widely used for evaluating the productivity of cultivars and hybrids and for identifying promising genotypes in breeding programs. Previous studies have demonstrated that the variability and heritability of these traits play an essential role in the development of high-yielding cotton varieties through targeted selection strategies. For instance, Meyer (1972) reported significant differences in the number of stamens per flower between New World and Old World cotton species, which are associated with their ploidy levels. According to this study, the tetraploid species G. hirsutum L. possesses an average of 121.6 stamens per flower, whereas the diploid species G. arboreum L. has approximately 78.6 stamens, indicating clear differences in reproductive morphology between these taxa [7].Considering the importance of reproductive stability for cotton productivity, the evaluation of embryological characteristics such as pollen fertility, pollen tube growth, and meiotic stability can provide valuable information for breeding programs. Therefore, the present study aims to assess the cyto-embryological stability and seed index variability in different Gossypium hirsutum genotypes, including both cultivated varieties and semi-wild forms, in order to identify promising genetic resources for future cotton breeding efforts.
2. Materials and Methods
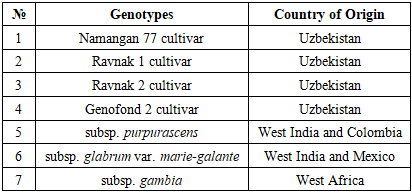
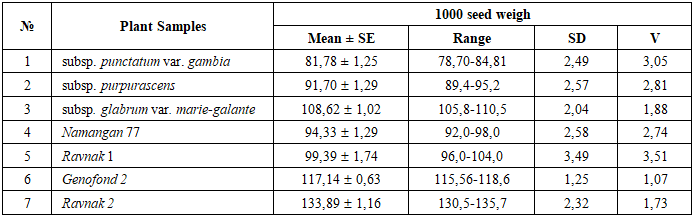
- Plant Materials and Germplasm CollectionThe study was conducted during a three-year period (2022-2024) in the experimental fields of the Institute of Genetics and Plant Experimental Biology of the Academy of Sciences of Uzbekistan (Tashkent region). Several Gossypium hirsutum L. genotypes, including modern cultivars and semi-wild forms, were selected for the investigation. The plant material was obtained from the Uzbekistan Cotton Germplasm Collection maintained at the Institute of Genetics and Plant Experimental Biology. The analyzed genotypes included four cultivated varieties (Namangan 77, Ravnak 1, Ravnak 2, and Genofond 2) and three semi-wild forms (subsp. purpurascens, subsp. glabrum var. marie-galante, and subsp. punctatum var. gambia). These genotypes represent diverse genetic backgrounds and were selected in order to evaluate their reproductive stability and agronomic potential (Table 1).
|
3. Results
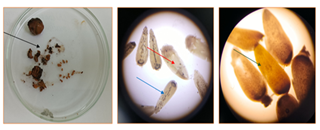
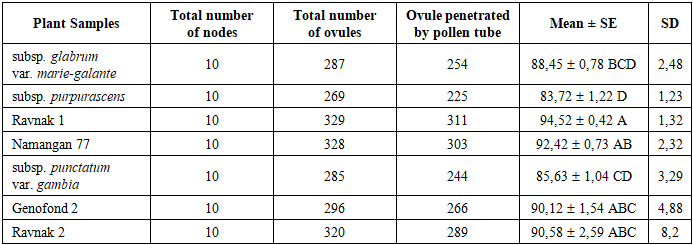
- Embryological Characteristics of the Studied Genotypes Embryological traits were analyzed in both cultivated varieties and semi-wild forms of Gossypium hirsutum. The investigation focused on several reproductive parameters, including the total number of ovules per ovary, the number of ovules penetrated by pollen tubes, and the overall efficiency of fertilization (Fig. 1).
|
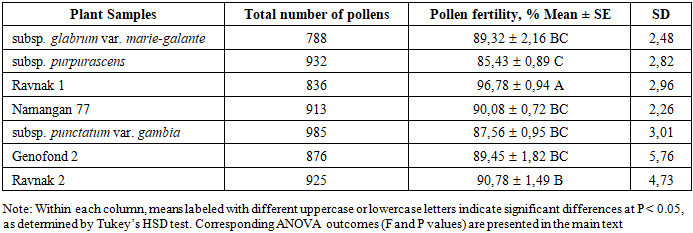
 | Figure 2. Pollen fertility analysis: a) Namangan 77 cultivar (90.08±0.72%); b) subsp.purpurascens (85.43±0.89%) (10× magnification) |
|
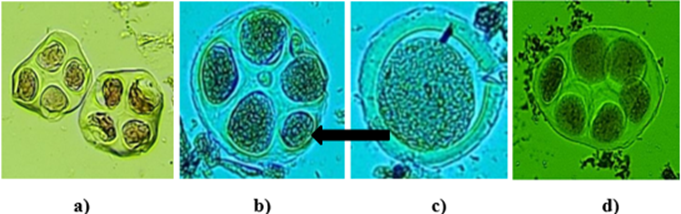 | Figure 3. Microscopic images of tetrads in subsp.punctatum var.gambia: a) normal tetrads; b) micronucleated tetrads (arrows indicate micronuclei); c) monads; d) hexads (at 40×magnification) |
|
4. Discussion
- The experimental results demonstrate a clear physiological and genetic divergence in the reproductive efficiency of Gossypium hirsutum genotypes. The embryological analysis revealed that Ravnak 1 exhibits superior reproductive potential, with a peak ovule penetration rate of 94.52±0.42, significantly outperforming semi-wild forms such as subsp. purpurascens (83.72±1.22). This heightened efficiency in pollen tube navigation to the micropyle is inherently linked to the stability of the male gametophyte. Statistical analysis of our data reveals a significant positive correlation between pollen viability and pollen tube growth (r=0.583, p<0.001), supporting the link between high fertility and successful fertilization processes. Among the studied genotypes, Ravnak 1 exhibited the highest performance, recording the top fertility rate (96.82±2.87%) and superior pollen tube growth (94.52±1.29%). The moderate-to-strong nature of this correlation (R2=0.34) suggests that while pollen viability is a primary determinant of fertilization success, the efficiency of pollen tube penetration also plays a critical role in the overall reproductive stability of these cotton varieties. Recent studies have also emphasized the importance of pollen fertility and pollen tube growth for cotton productivity. For instance, Zhang et al. (2024) reported that environmental stress factors, particularly elevated temperature and drought, significantly reduce pollen fertility and consequently affect boll formation and yield components in cotton [16]. Similarly, Luqman et al. (2025) demonstrated that disruptions in pollen germination and pollen tube elongation are closely associated with metabolic disturbances in anthers and may lead to decreased reproductive efficiency in Gossypium hirsutum under stress conditions [15]. These findings confirm that reproductive biological parameters such as pollen viability and pollen tube growth play a key role in determining cotton yield potential and should be considered in breeding programs aimed at improving stress tolerance and productivity. These findings suggest that directional pollen tube growth, governed by the mechanisms of chemotropism and durotropism discussed in classical literature, is more optimized in modern local cultivars compared to semi-wild accessions. The cytogenetic investigation into subsp. punctatum var. gambia provides a critical perspective on the chromosomal basis of fertility. Although the meiotic index (90.39±1.19) indicates a relatively stable microsporogenesis, the presence of micronucleated tetrads and polyads (up to 6.47%) suggests occasional aneuploid sporads and irregular chromosomal segregation. Such meiotic irregularities explain the intermediate fertility levels observed in wild-type genotypes. Furthermore, the variability in the 1000-seed weight (seed index) highlights the impact of targeted selection. The exceptional seed mass in Ravnak 2 (133.89±1.16 g) and the high population uniformity of Genofond 2 (V=1.07) demonstrate that breeding for embryological stability directly translates into superior agronomic traits. The statistical significance established by Tukey’s HSD test confirms that genotypes with higher embryological performance (Group A) are the most suitable candidates for high-yield breeding and hybridization programs.
5. Conclusions
- The present study demonstrated that significant differences exist among Gossypium hirsutum genotypes in terms of their reproductive efficiency, cytogenetic stability, and seed productivity. The results of embryological analysis showed that modern cotton cultivars generally possess higher fertilization efficiency compared with semi-wild forms. Among the studied genotypes, the cultivar Ravnak 1 exhibited the highest reproductive performance, characterized by a high rate of pollen tube penetration into ovules and superior pollen fertility. These parameters indicate strong stability of the male gametophyte and efficient interaction between pollen tubes and maternal reproductive tissues. Similarly, Namangan 77 also demonstrated relatively high fertilization efficiency, suggesting its potential value as a promising genotype for breeding programs. In contrast, semi-wild forms such as subsp. purpurascens and subsp. punctatum var. gambia showed lower reproductive indices, which may be associated with observed meiotic irregularities, including the formation of micronucleated tetrads and polyads. Such cytogenetic abnormalities can negatively affect gamete viability and may reduce overall fertilization success. The analysis of yield-related traits revealed substantial variability in 1000-seed weight (seed index) among the investigated genotypes. The cultivar Ravnak 2 showed the highest seed weight, whereas Genofond 2 exhibited exceptional population uniformity with a very low coefficient of variation. These characteristics indicate that targeted breeding and selection have contributed to improving seed productivity and genetic stability in modern cotton cultivars.Overall, the results of this study highlight the importance of integrating embryological and cytogenetic indicators into cotton breeding strategies. Parameters such as pollen fertility, pollen tube penetration efficiency, meiotic stability, and seed index variability can serve as reliable criteria for identifying promising genetic resources. The genotypes identified in this study may therefore represent valuable material for future cotton breeding programs aimed at developing high-yielding and genetically stable cultivars.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML