-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Ecosystem
p-ISSN: 2165-8889 e-ISSN: 2165-8919
2026; 15(1): 1-13
doi:10.5923/j.ije.20261501.01
Received: Jan. 22, 2026; Accepted: Feb. 16, 2026; Published: Mar. 10, 2026

Effects of Anthropogenic Activities on Woody Species Diversity, Composition, and Structure in Biharamulo-Kahama Forest Reserve, Tanzania
Ng’hwale Ozano Bubinza, Paulo John Lyimo, Ezekiel Edward Mwakalukwa
Department of Ecosystems and Conservation, College of Forestry, Wildlife and Tourism, Sokoine University of Agriculture, Chuo Kikuu, Morogoro, Tanzania
Correspondence to: Ng’hwale Ozano Bubinza, Department of Ecosystems and Conservation, College of Forestry, Wildlife and Tourism, Sokoine University of Agriculture, Chuo Kikuu, Morogoro, Tanzania.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Miombo woodlands are ecologically and socio-economically important ecosystem, yet they are increasingly degraded by unsustainable human activities. The Biharamulo–Kahama Forest Reserve (BKFR), one of Tanzania’s largest dry miombo reserves, faces mounting pressures from logging, charcoal production, grazing, and recurrent fires. This study examined the effects of such disturbances on woody species diversity, composition, and structural attributes. Data were collected from 24 May 2024 to 25 July 2024 using a systematic cluster sampling design across the forest area of 30,712ha. Woody individuals were identified in 53 circular sample plots of 15 radius, counted, and measured for diameter at breast height (dbh) (1.3m above ground). Various anthropogenic disturbances were also recorded in a 15 m radius plot. Species diversity was quantified using the Shannon–Wiener diversity index, while stem density and basal area were derived from the dbh measurements for each tree. Generalized Linear Models (GLM) was applied to assess the effects of disturbances on structural attributes (species composition, stem density and basal area). A total of 37,245 individuals of all categories (Adults = dbh ≥ 5 cm and regenerants, dbh < 5 cm) were recorded, representing 66 species, 56 genera, and 27 plant families, with Fabaceae and Combretaceae being dominant plant families. Species diversity was relatively high (H′ = 3.17 for adults (dbh ≥ 5 cm); H′ = 3.30 for regenerants (dbh < 5 cm), although 73 harvested stumps highlighted ongoing exploitation of valuable 19 species. Mean stem density was 461 ± 165 stems ha⁻¹ for adults and 9,716 ± 4,036 stems ha⁻¹ for regenerants, while basal area for adults averaged 7.43 ± 2.38 m² ha⁻¹ and that’s of regenerants was 0.59 ± 0.26 m2ha-1. GLM results revealed that tree cutting significantly reduced species diversity (p < 0.05) whereas stem density was not affected by the identified anthropogenic activities. Grazing and tree cutting altered basal area patterns. These results indicate that persistent anthropogenic disturbances are reshaping forest structure and threatening regeneration in BKFR. Enhanced enforcement, community-based forest management, and restoration strategies are essential to safeguard biodiversity and ensure long-term ecological resilience of this dry miombo ecosystem.
Keywords: Human activities,Miombo woodlands, Lake zone, Disturbances, Forest degradation, Biodiversity loss
Cite this paper: Ng’hwale Ozano Bubinza, Paulo John Lyimo, Ezekiel Edward Mwakalukwa, Effects of Anthropogenic Activities on Woody Species Diversity, Composition, and Structure in Biharamulo-Kahama Forest Reserve, Tanzania, International Journal of Ecosystem, Vol. 15 No. 1, 2026, pp. 1-13. doi: 10.5923/j.ije.20261501.01.
Article Outline
1. Introduction
- Forests are vital ecosystems that cover about 31% of the Earth’s surface and provide crucial ecological, social, and economic services to human wellbeing [1]. Forests regulate climate, conserve biodiversity, stabilize soils, and supply resources essential for human well-being. Tropical forests, in particular, are renowned for their exceptional biodiversity and ecological productivity, making their conservation critical for both local livelihoods and global sustainability [2].Among tropical ecosystems, miombo woodlands are especially significant due to their vast distribution and high woody species diversity [3]. Spanning approximately 2.7 million km² across eastern, central, and southern Africa, these woodlands are dominated by Brachystegia, Julbernardia, and Isoberlinia species [4]. They are classified into wet and dry miombo based on rainfall regimes, with wet miombo receiving over 1000 mm annually and dry miombo less than 1000 mm [5]. Miombo woodlands are ecologically and socio-economically important, providing timber, fuelwood, grazing land, honey, medicinal plants, and climate regulation services [6].Despite their significance, miombo ecosystems face escalating anthropogenic pressures such as logging, shifting cultivation, uncontrolled grazing, and charcoal production [7]. These activities lead to forest degradation, biodiversity loss, and a reduced carbon sequestration potential [8]. In Tanzania, an estimated 372,816 hectares are lost annually due to unsustainable human activities [9]. This rapid decline threatens ecosystem resilience, biodiversity conservation, and the provision of vital ecosystem services [10].The Biharamulo-Kahama Forest Reserve (BKFR), one of Tanzania’s largest dry miombo forest reserves, covers about 134,680 hectares and provides critical ecological functions while supporting local livelihoods [11]. Despite its ecological and socioeconomic importance, the reserve faces increasing pressure from anthropogenic activities, including illegal logging, charcoal production, shifting cultivation, livestock grazing, and recurrent fires. These disturbances not only threaten the diversity and structure of woody species but also undermine natural regeneration and reduce the reserve’s capacity to deliver ecosystem services in the long term. Assessing the extent of these impacts is therefore crucial for guiding sustainable forest management and conservation interventions in the area [11]. This study aimed to assess these impacts in BKFR by (i) analyzing woody species richness, diversity, and composition, (ii) evaluating structural characteristics of the reserve and (iii) examining the effects of anthropogenic activities on woody species diversity and forest structure. The results will provide baseline information for sustainable forest management and inform conservation strategies aligned with Tanzania’s national goals and international commitments under the Convention on Biological Diversity (CBD) and United Nations Framework Convention on Climate Change (UNFCCC).
2. Materials and Methods
2.1. Description of the Study Area
- The Biharamulo-Kahama Forest Reserve (BKFR) is located in north western Tanzania, approximately 25 km from Biharamulo district, 55 km from Chato district, and 40 km from Bukombe district [11]. Geographically, the reserve lies between 31.9°E to 32.6°E and 2.1°S to 3.6°S (Figure 1). The total area of the BKFR is 134,680 ha, of which 64,924 ha is covered by natural forests. This study specifically focused on a 30,712ha portion in the north western section of the reserve within the natural forest zone [11]. BKFR lies at an elevation of 1,000–1,400 m above sea level and experiences a tropical savanna climate with a unimodal rainfall regime between November and May. Average annual rainfall ranges from 700 mm to 1,200 mm, while mean temperatures vary between 26°C in the cool season and 30°C in the hot season [11]. The vegetation is dominated by typical miombo species such as Julbernardia globiflora, Ficus natalensis, Brachystegia spiciforms, Brachystegia bohemii, Pericopsis angolensis and Combretum collinum accompanied by Combretum and Terminalia species, in low land areas with clay mbuga soils supporting the growth of shrubs and grasses [11]. In swamps and riverine areas, the vegetation cover is riverine tree species such as Ficus natalensis and Syzygium spp [11]. The soils are primarily ferrosols and sandy loams, which support both woody vegetation and small-scale cultivation in encroached areas [11]. Ecologically, BKFR is significant for its role in biodiversity conservation, carbon storage, soil stabilization, and watershed protection, being part of the Lake Victoria catchment. It provides a habitat for a wide variety of woody species and wildlife while sustaining local livelihoods through the provision of fuelwood, timber, honey, fruits, medicinal plants, and grazing land [11].
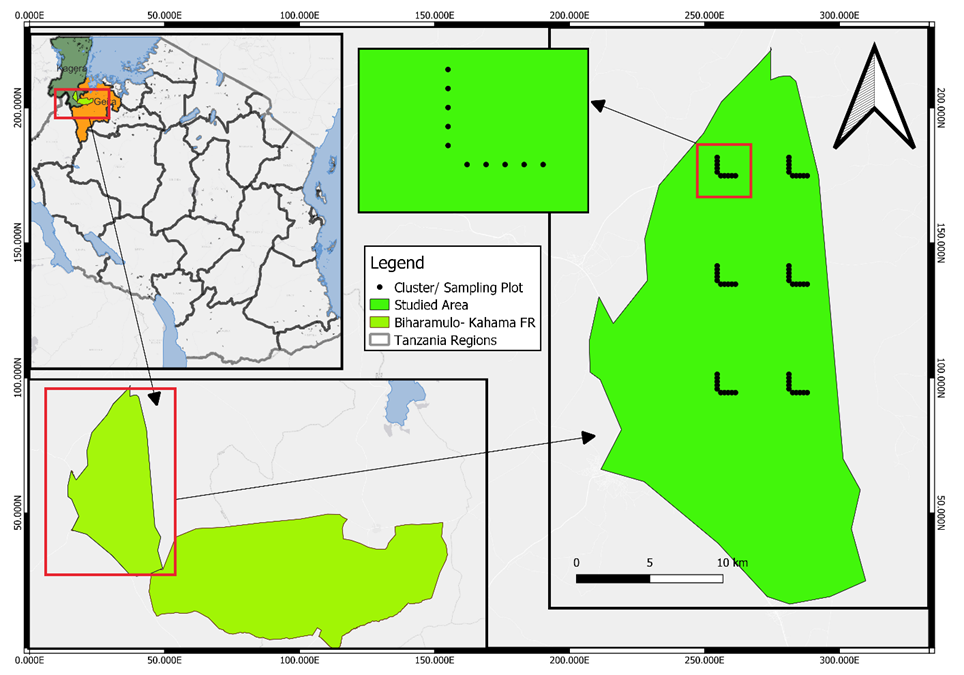 | Figure 1. A map of Tanzania showing the location of the study area and plots layout in the Biharamulo-Kahama Forest Reserve in Biharamulo District, Tanzania |
2.2. Sampling Design
- The field survey was conducted from 24 May 2024 to 25 July 2024 within the Biharamulo-Kahama Forest Reserve (BKFR) to assess the effects of anthropogenic activities on woody species diversity, composition, and structure. A systematic cluster sampling design was employed, consisting of six clusters arranged in an L-shaped layout following the National Forestry Resources Monitoring and Assessment of Tanzania (NAFORMA) guidelines [9]. Each cluster contained 10 subplots, giving a total of 60 subplots, with a sampling intensity of 0.001%. However, only 53 subplots were successfully surveyed due to challenges such as cliffs, rugged terrain, and limited accessibility, which restricted full coverage. Within each cluster, nested circular sample plots of radius 2 m, 5 m, 10 m, and 15 m were established at 250 m intervals between subplots. The spacing between clusters was 4,900 m, while the distance between transects was 7,400 m, ensuring broad spatial representation and reducing the risk of sampling bias [12].
2.3. Data Collection
- Data collection focused on recording woody species diversity, composition, and structure within the established subplots. In each subplot, all woody plants were identified and classified into size categories based on diameter at breast height (Dbh). All trees and shrubs (seedlings) with Dbh < 1 cm were counted and identified within the 2 m radius plot, while saplings (Dbh ≥ 1 cm but ≤ 5 cm) were measured and recorded within the 5 m radius. Young trees (Dbh ≥ 5 cm but < 20 cm) were identified and measured within a 10 m radius, whereas adult trees (Dbh ≥ 20 cm) were recorded in a 15 m radius [13]. For each tree, Dbh was measured at 1.3 m above the ground using a diameter tape. Species identification was done in the field using standard floras and field guides. Unknown species were collected, pressed, and later verified at the National Herbarium of Tanzania. In addition to vegetation parameters, all visible anthropogenic disturbances were recorded in each subplot to assess their influence on woody species. This included evidence of footpath, tree debarking, grazing, farming, charcoal making, fire incidents, and tree cutting. Disturbance intensity was assessed by counting the frequency of observed signs within a 15 m radius plots. Recording both vegetation characteristics and human activities ensured that the study captured not only the diversity and structural attributes of woody species but also the pressures influencing forest structure, composition and regeneration. This integrated approach provided a comprehensive dataset to evaluate the effects of anthropogenic activities on woody vegetation in Biharamulo-Kahama Forest Reserve.
2.4. Data Analysis
- Data were analyzed using both descriptive and inferential statistics. Species diversity was quantified using the Shannon–Wiener diversity index (H′), which accounts for both species’ richness and evenness. The index was computed using the formula [14];
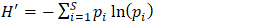 | (1) |
 | (2) |
 | (3) |
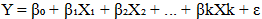 | (4) |
3. Results and Discussion
3.1. Results
3.1.1. Species Richness and Composition
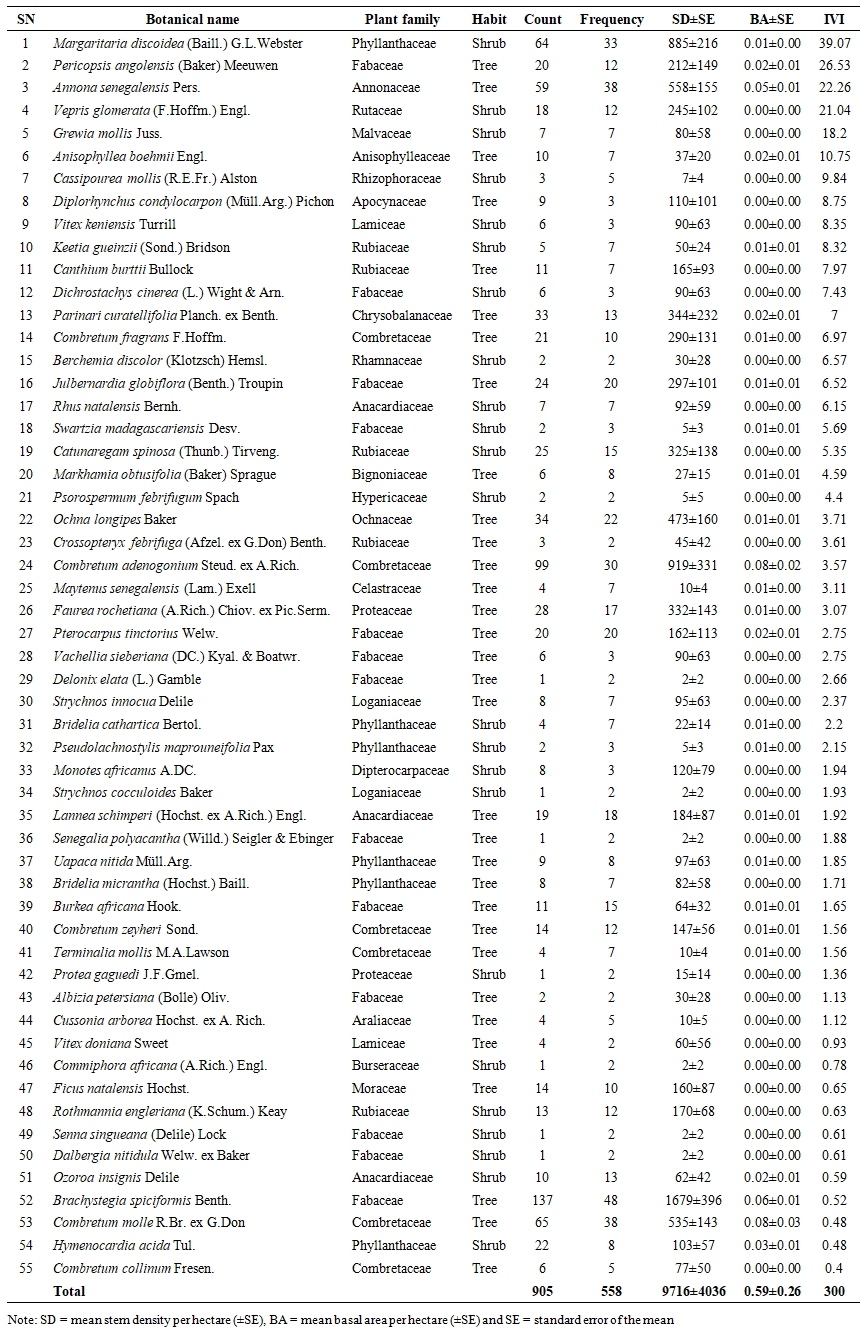
- A total of 37,245 woody individuals (trees and shrubs combined) with diameters ranging from 1 to 51 cm were recorded in Biharamulo–Kahama Forest Reserve (BKFR), representing 66 species, 56 genera, and 27 plant families. Trees accounted for 62% of all species across 19 families, while shrubs contributed 38% across 16 families. The flora was dominated by the Fabaceae family (35% of species), followed by Combretaceae (24%), Phyllanthaceae (8%), and Annonaceae (6%), reflecting a floristic composition characteristic of dry miombo ecosystem. When adult individuals (Dbh ≥ 5 cm) were considered separately, a total of 891 stems representing 56 species, 46 genera, and 24 families were recorded, with Fabaceae remaining dominant (27% of species), followed by Combretaceae (13%), Phyllanthaceae (11%), and Anacardiaceae (5%) (Table 1). Regenerants (Dbh < 5 cm) comprised 905 individuals belonging to 55 species, 48 genera, and 24 families, where Fabaceae contributed 24%, Combretaceae 11%, and Phyllanthaceae 11% (Table 2). Additionally, 73 stumps of illegally harvested trees were recorded, representing 19 species, 17 genera, and 12 families, with diameters ranging from 4 to 52 cm (mean diameter = 21 cm) (Table 3). The families Combretaceae (26%) and Fabaceae (21%) contributed the largest proportion of harvested stems, emphasizing their high economic value for timber and fuelwood extraction.
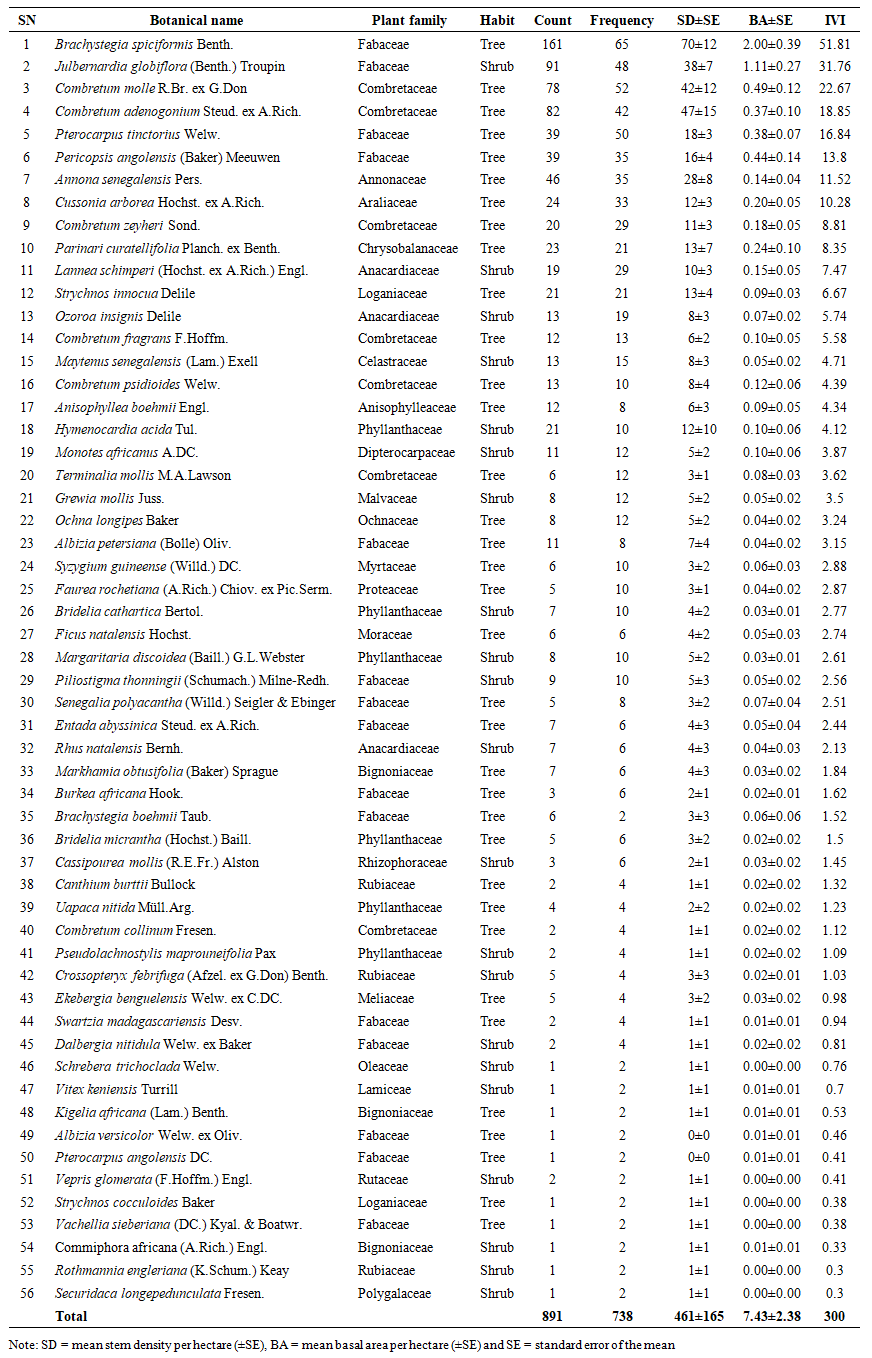 | Table 1. Species diversity and structural attributes of adult woody individuals (Dbh ≥ 5 cm) sorted by the Importance value index (IVI) recorded in the Biharamulo–Kahama Forest Reserve, Tanzania |
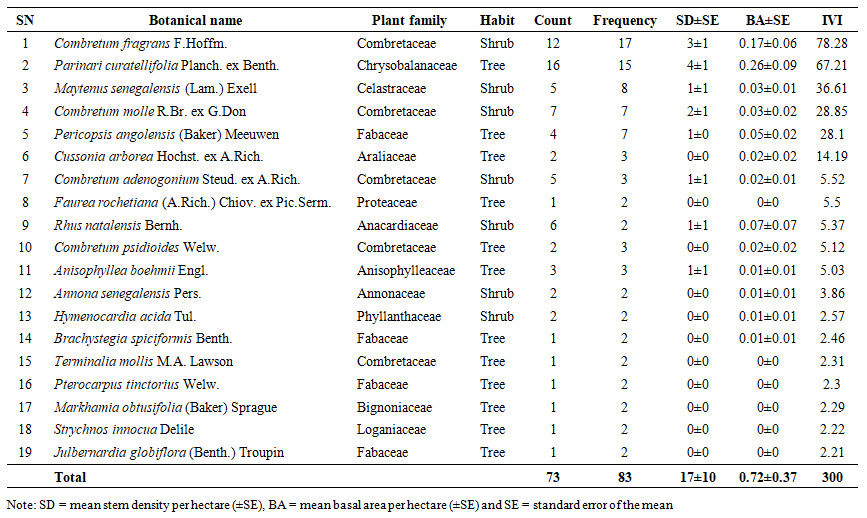 | Table 3. Summary of illegally harvested woody species (stumps) sorted by the Importance value index (IVI) identified in the Biharamulo–Kahama Forest Reserve, Tanzania |
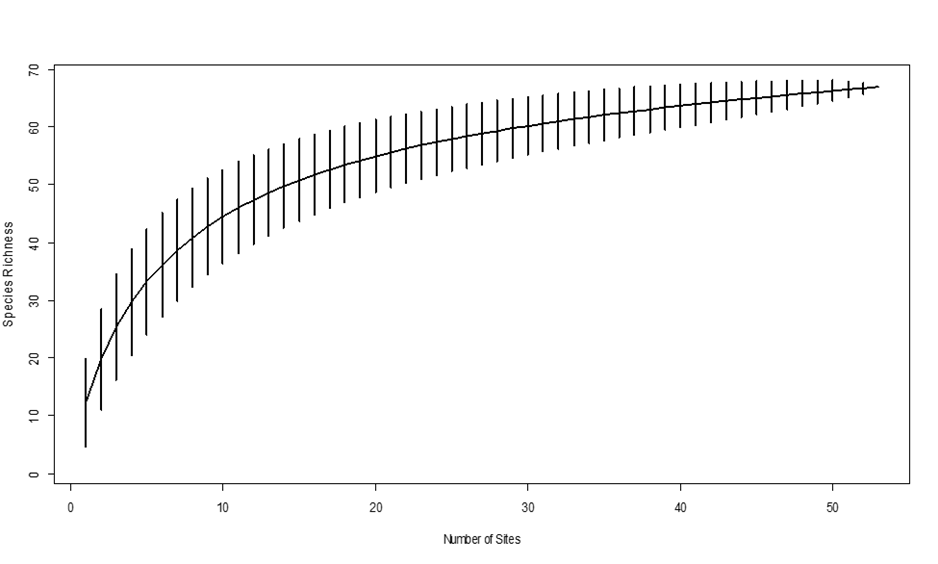 | Figure 2. Species accumulation curve for large individuals (Dbh ≥ 5cm) measured within circular plots with a radius of 15m in Biharamulo–Kahama Forest Reserve, Tanzania |
3.1.2. Species Diversity
- The Shannon-Wiener diversity indices for large individuals (Dbh ≥5cm) and small individuals (Dbh <5cm) were 3.17 (Table 1) and 3.30 (Table 2), respectively. The following species had the greatest contribution to the Shannon-Wiener diversity index of large individuals (Dbh ≥5cm): Brachystegia spiciformis Benth (H’ = 0.31), Julbernardia globiflora (Benth.) Troupin (H’= 0.23), Combretum adenogonium Steud. ex A. Rich (H’= 0.22), Combretum molle R.Br. ex G.Don (H’ = 0.21) and Annona senegalensis Pers (H’=0.15). Meanwhile, for regenerants, the species with the highest Shannon-Wiener diversity index was Brachystegia spiciformis Benth with (H’ = 0.29), followed by Combretum adenogonium Steud. ex A.Rich (H’=0.24) and Combretum molle R.Br. ex G.Don (H’=0.19). This result shows the BKFR supports a relatively high and well-balanced woody species diversity.
3.1.3. Stem Density
- The mean stem density of BKFR for an adult individual (Dbd ≥ 5cm), was 461 ± 165 stems/ha, and that for small individuals (Dbh <1cm) was 9,716 ± 4,036 stems/ha. Among these species, Brachystegia spiciformis Benth had 15% of the total stem density, followed by Combretum adenogonium Steud. ex-A.Rich. at 10%, Combretum molle R.Br. ex G.Don at 9%, and Julbernardia globiflora (Benth.) Troupin at 8%, whereas among small individuals (Dbh< 5cm), the most abundant species were Brachystegia spiciformis Benth (17%), Combretum adenogonium Steud. ex A.Rich (10%), Margaritaria discoidea (Baill.) G.L.Webster (9%), Annona senegalensis Pers (6%), and Combretum molle R. Br. ex G. Don (6%).For stumps, the overall mean density was 17 ± 10 stem/ha (Table 3), where the most abundant species was Pericopsis angolensis (Baker) Meeuwen with 22% of total stump density, followed by Brachystegia spiciformis Benth (17%), Combretum adenogonium Steud. ex A. Rich (10%), Combretum psidioides Welw (8%), and Annona senegalensis Pers (7%). The density distribution shows an inverted ‘J’ shape, which is common for natural forests with active regeneration and recruitment (Figure 3). Regenerant trees and shrubs dominate in number, while adult trees and shrubs are fewer due to competition and slower growth rates. However, higher stump density in larger diameter classes suggests selective logging of bigger trees, possibly due to their higher timber value. If harvesting continues to focus on larger trees, it may impact forest structure, regeneration, and long-term sustainability.
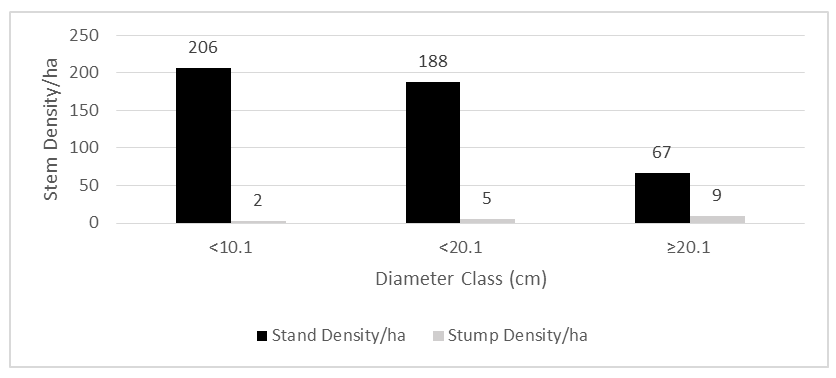 | Figure 3. Density of standing trees ≥5 cm Dbh and stumps ≥4 cm by diameter classes in Biharamulo-Kahama Forest Reserve (BKFR), Tanzania (𝑛 = 53) |
3.1.4. Basal Area
- The mean basal area for the individuals with Dbh ≥5 cm was 7.43 ± 2.38 m2ha-1 (Table 1). The species contributing most to the basal area of large individuals was Brachystegia spiciformis Benth, which contributes 26.97% of the total basal area, followed by Julbernardia globiflora (Benth.) Troupin (14.91%), Combretum molle R.Br. ex G.Don (6.55%) and Pericopsis angolensis (Baker) Meeuwen (5.92%). Meanwhile, for small individuals with Dbh <5 cm mean basal area was 0.59 ± 0.26 m2ha-1. The species with the most basal area for regenerants was Combretum adenogonium Steud. ex A. Rich (14.1%), Combretum molle R.Br. ex G.Don (13.93%), Brachystegia spiciformis Benth (10.65%), and Annona senegalensis Pers (8.86%). For harvested stems, the mean basal area was 0.72 ± 0.37 m2ha-1, in which Pericopsis angolensis (Baker) Meeuwen has the greatest contribution of basal area for stumps, which contributes 35.8% of all basal area, followed by Brachystegia spiciformis Benth (23.41%), Combretum psidioides Welw (9.87%) and Pterocarpus tinctorius Welw (6.49%), and Combretum adenogonium Steud. ex A. Rich (4.16%) (Table 3). Generally, the distribution of trees to diameter classes shows the normal distribution shape (Figure 4) where the basal area for the stand tend to increase with the increase in the diameter size. The trend in stump basal area suggests minimal harvesting in smaller diameter trees, while harvesting intensity increases for larger trees (≥ 20.1 cm).
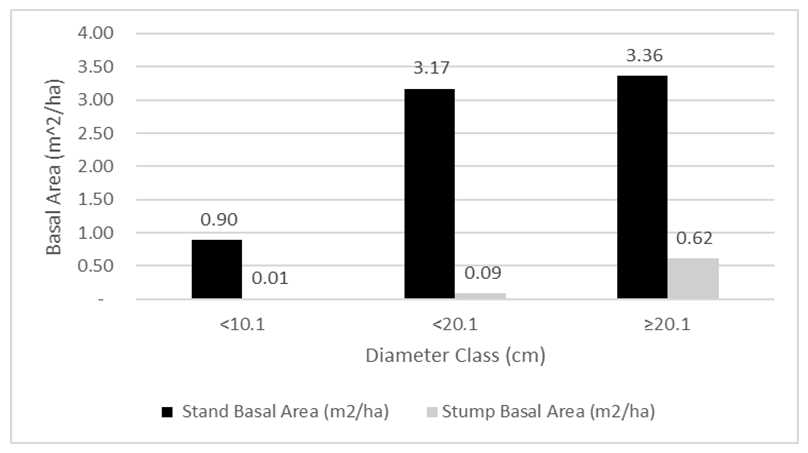 | Figure 4. Distribution of basal area per hectare for standing trees with Dbh ≥5 cm and stumps ≥4 cm by diameter classes at Biharamulo-Kahama Forest reserve (n=53) |
3.1.5. Effects of Anthropogenic Activities on Woody Species Diversity and Forest Structure
3.1.5.1. The Frequency of Occurrences of Anthropogenic Activities
- The assessment of the disturbance frequencies in BKFR show that tree cutting was the most dominant disturbances, contributing (47.16%) of the seven (7) disturbances observed (Figure 5). This was followed by grazing (28.83%) and fire (15.09%). Moderate level of disturbances were attributed to footpaths and charcoal making, both representing (9.43%) of the observations. Farming contributed a smaller proportion 7.5%, while debarking was the least recorded disturbances, with only (1.88%). These finding indicate that extraction and land use activities such as tree cutting and grazing are the primary anthropogenic pressures affecting the reserves.
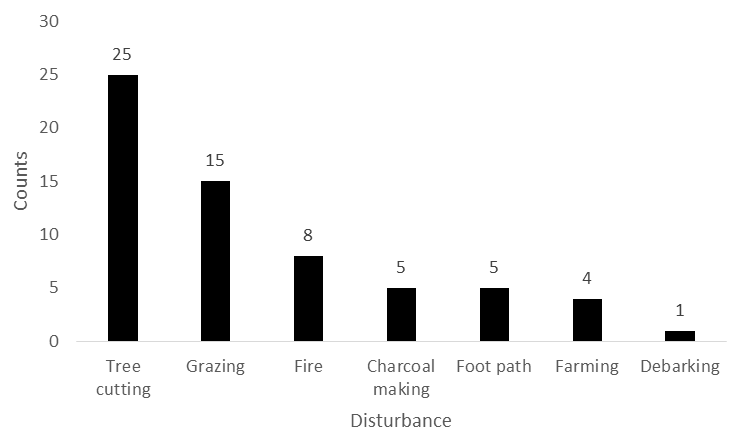 | Figure 5. Distribution of disturbances recorded in the Biharamulo-Kahama Forest Reserve, Tanzania |
3.1.5.2. Species Diversity
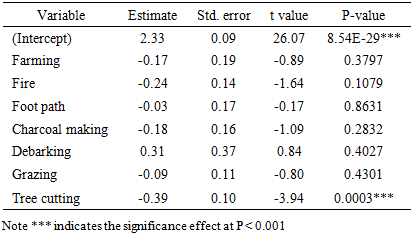
- The Generalized Linear Model (GLM) revealed that tree cutting had a highly significant negative effect on woody species diversity (Estimate = –0.388, p < 0.001), indicating that plots affected by cutting supported fewer species and reduced evenness compared to undisturbed plots. Other disturbances, including farming, fire, charcoal making, grazing, and foot paths, showed negative but non-significant relationships with diversity. Interestingly, debarking showed a positive but non-significant effect, suggesting that light bark harvesting may not immediately reduce species richness (Table 4). Generally, tree cutting emerged as the strongest driver of diversity loss in the BKFR.
|
3.1.5.3. Stem Density
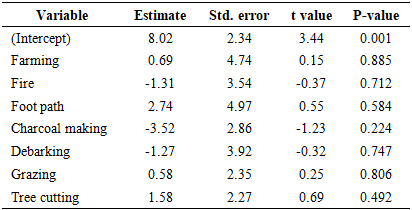
- Analysis for the anthropogenic activities shows that, none of the assessed disturbances showed statistically significant effects (Table 5). Although tree cutting (Estimate = 1.576, p = 0.492) and foot paths (2.735, p = 0.584) tended to increase stem density, the effects were weak and not significant. Similarly, farming (0.687, p = 0.885) and grazing (0.580, p = 0.806) showed slight positive associations, whereas charcoal making (–3.520, p = 0.224), fire (–1.314, p = 0.712), and debarking (–1.270, p = 0.747) were negatively related to stem density. The lack of significant results suggests that stem density may be regulated by compensatory regeneration and natural recruitment processes, which buffer the impacts of anthropogenic disturbances (Table 5).
|
3.1.5.4. Basal Area
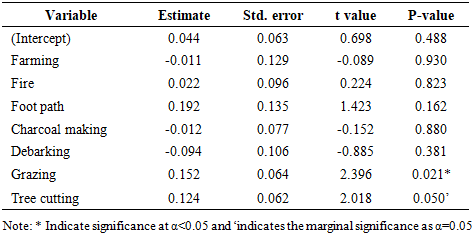
- The disturbance analysis indicated that grazing had a significant positive effect on basal area (Estimate = 0.152, p < 0.05), implying that light to moderate grazing may favor the persistence of larger trees by reducing competition from seedlings and saplings. Tree cutting also exhibited a marginally significant positive relationship with basal area (Estimate = 0.124, p = 0.05), which may reflect selective removal of small or medium-sized individuals, leaving behind trees with higher basal area. Conversely, debarking, charcoal making, and farming had weak and non-significant negative effects while fire and foot paths were positively associated with basal area but not significantly (Table 6).
|
3.2. Discussion
3.2.1. Species Richness and Composition
- The species richness of 56 adult species (≥ 5cm Dbh) recorded in BKFR is lower than reported in several other Tanzanian dry miombo woodlands. For instance, [17] reported 80 species in Kitulanghalo, [18] recorded 88 species from Gangalamtumba VLFR, [19] reported 71 species in Kitulanghalo, and [7], documented 122 species in Chunya. In contrast, species richness in BKFR is higher than that reported by [20] who reported 46 species in Ihombwe and comparable to other dry forests in Tanzania. Differences in richness may reflect variations in sampling intensity, forest size, and disturbance regimes. For example, studies with larger sampling areas and higher numbers of plots generally record more species [21]. Nevertheless, the richness observed in BKFR lies within the expected range for miombo woodlands (40–229 species) [21], [7], [25], [18].The dominance of Fabaceae and Combretaceae plant families aligns with typical floristic patterns in dry miombo [23]; [24], indicating that despite disturbance, BKFR retains key ecological signatures of miombo woodlands. However, the presence of 73 illegally harvested stumps underscores the ongoing threat of selective logging. The high proportion of Fabaceae and Combretaceae plant families among stumps highlights their economic and fuelwood value, suggesting that these families are under disproportionate pressure.The sufficient sampling coverage indicated by the species accumulation curve (Figure 2) strengthens confidence that the recorded richness reflects the standing species pool in BKFR. Thus, while BKFR remains relatively species-rich, anthropogenic activities are reshaping community composition and may threaten long-term regeneration potential if not controlled.
3.2.2. Species Diversity
- The values of the Shannon-Wiener diversity index (H ´ = 3.17) for adult trees and shrubs in the present study are lower than those reported by [25] who reported an H’ value of 4.27 in the miombo woodland of the Bereku forest reserve in Tanzania, [26] who reported an H’ value of 4.06 and 4.26 in Kibutuka miombo woodland in Liwale District, Tanzania, and Kitulanghalo Miombo woodland in Morogoro District, Tanzania, respectively. However, H´ values in this study are much higher than those documented by [27], who reported an H’ value of 3.13 in the Miombo Woodlands of the Kitulanghalo Forest Reserve in Morogoro, Tanzania. However, the H´ value of 3.17 in this study falls in the range of H’ values commonly found in miombo woodland, ranging from 1.05 - 4.27 [28], [18], [7]. The H´ values normally vary between 1.5 and 4.5 but rarely exceed 5. A threshold value of 2 has been cited to be the minimum value, above which an ecosystem can be regarded as medium to highly diverse [29], [20]. Therefore, the value of 3.17 found in this study suggests that the Biharamulo Kahama Forest Reserve exhibits high diversity in tree and shrub species.
3.2.3. Stem Density
- The stem density of 461 ± 165 stems ha-1 for the woody species with Dbh ≥ 5 cm reported in this study is higher than that documented by [31], who reported a mean density of 77 ± 52 stems ha-1 from Esilalei Village Land Forest Reserve in Northern-Eastern Tanzania. However, the stem density of 461 ± 165 stems ha-1 found in this study is lower than the value reported by [25] who reported a mean density of 616 ± 46 stems ha-1 from miombo woodland of Bereku forest reserve in Tanzania; [27], who reported three values of mean stem density of 1,085 ± 115 stems ha-1, 1,027 ± 88 stems ha-1 and 1,495 ± 208 stems ha-1 from Kitulanghalo forest reserve, Tanzania; and [18] who reported a mean stem density of 1521 ± 594stem ha-1 from Gangalamtumba VLFR. This implies that Biharamulo Kahama Forest Reserve is among the lowest stocked dry forests/woodlands in Tanzania and other forests in tropical countries. The higher density reported in other studies might be attributed to the influence of microclimate, which creates favorable conditions for the growth of more species. Agriculture and the tree cutting for charcoal, poles, or timber could also have affected the density of species in the Biharamulo Kahama Forest Reserve. However, the value of 461 ± 165 stem ha-1 reported in this study falls in the range of stem densities commonly found in the dry miombo woodlands of 347- 1,521 stem ha-1 [32]; [33]. This indicates that Biharamulo Kahama Forest Reserve is among the lowest stocked forest in the country and tropical regions due to ongoing anthropogenic activities taking place in the reserve.
3.2.4. Basal Area
- The mean basal area of 7.43 ± 2.38 m² ha⁻¹ of the adults (Dbh ≥ 5 cm) recorded in this study is lower than that reported from other Tanzanian miombo woodlands. For example, [34], found a mean basal area of approximately 12m2ha-1, indicative of somewhat denser woodlands in parts of dry miombo woodland. [17] reported basal area values around 7.61± 4.47m2ha-1 in Kitulanghalo Forest Reserve in Tanzania. The comparatively low basal area observed in BKFR may be attributed to the relatively low stem density observed in the area. Since basal area is directly influenced by both stem density and the size distribution of trees, a lower representation of individuals in larger diameter classes limits the accumulation of basal area. By contrast, forests with higher stem densities and greater representation of large-diameter trees exhibit higher basal area values. These findings suggest that anthropogenic activities, particularly the selective harvesting of larger trees, have reduced the standing basal area in BKFR, thereby lowering its structural integrity compared to other miombo woodlands.
3.2.5. Effects of Anthropogenic Activities on Woody Species Diversity and Forest Structure
- The assessment of disturbance frequencies in BKFR revealed that tree cutting was the most frequent disturbance (47.16%), followed by grazing (28.83%) and fire (15.09%). Footpaths and charcoal making each accounted for 9.43% of the disturbances while farming (7.5%), and debarking 1.88% were less frequent. Similar studies have shown that tree cutting is the most commonly observed disturbance in tropical forests, often driven by demand for timber and fuel wood [35]. This pattern reflects the dominance of extraction and use activities as key pressures in the reserve and highlights the need for strategies to manage these disturbances effectively.These disturbance patterns are important because they relate directly to the ecological consequences observed in the forest. The findings of this study demonstrate that anthropogenic disturbances exert variable impacts on woody species diversity and forest structure in BKFR. Among the seven disturbances analyzed, tree cutting had the most significant negative effect on species diversity (p < 0.001), indicating that selective removal of mature individuals reduces both species richness and evenness. This aligns with earlier study in Tanzanian miombo woodlands, which have shown that uncontrolled logging and pole cutting selectively target valuable timber species, thereby diminishing overall diversity [36]. Also the study from Malawi by [37] found that extensive logging in miombo woodland threatens resources nexus potential for diverse values of nature. Other disturbances, such as fire, grazing, charcoal making, and farming, also negatively influenced diversity, although their effects were statistically non-significant. Nevertheless, their ecological implications cannot be overlooked, as fire and charcoal burning in particular is known to kill seedlings and saplings, thereby suppressing regeneration and reducing long-term species diversity [38]. Forest structural attributes were also influenced by anthropogenic activities, though in contrasting ways. For stem density, no disturbance had a statistically significant effect, although negative coefficients for debarking, fire, and charcoal making suggest that these activities suppress regeneration and recruitment. Similar observations have been reported in other miombo ecosystems, where intensive wood extraction and recurrent fires reduce the density of younger cohorts, resulting in structurally simplified forests [39].Basal area was significantly affected by grazing (p = 0.021) and tree cutting (p = 0.05). Grazing pressure often suppresses small stems while favoring the growth of fewer, larger individuals, which may explain the observed increase in basal area despite its negative effects on regeneration. [20], reported similar findings from Central-Eastern Tanzania, showing that high grazing intensity reduced composition and regeneration, while basal area patterns shifted under grazing pressure. Conversely, tree cutting reduced diversity but contributed positively to basal area, suggesting that selective harvesting removes small and medium-sized individuals while leaving behind larger trees that dominate stand structure. Generally, these results indicate that while some disturbances (e.g., grazing and selective cutting) may temporarily enhance certain structural attributes such as basal area, the overall trend is toward reduced species diversity, altered stand composition, and impaired regeneration. This pattern threatens the ecological stability and resilience of miombo ecosystems, as diversity loss and skewed structure reduce the forest’s capacity to recover from further disturbances and maintain ecosystem services.
4. Conclusions and Recommendations
- This study revealed that anthropogenic activities, including tree cutting, grazing, fire, and charcoal production, significantly affect the diversity, composition, and structure of woody species in the Biharamulo–Kahama Forest Reserve. Species diversity was most negatively impacted by tree cutting, which selectively removed ecologically valuable species, thereby reducing overall richness and evenness. For stem density, no anthropogenic activities shows the significance effects, while grazing and tree cutting significantly influenced forest structure, with grazing altering basal area patterns and tree cutting reducing stand diversity. Although other disturbances showed non-significant statistical effects, their ecological consequences, particularly in suppressing regeneration and recruitment, are evident.To mitigate the adverse impacts of anthropogenic activities on woody species diversity and forest structure in Biharamulo–Kahama Forest Reserve, a multifaceted management approach is required. First, strict enforcement of forest protection measures should be prioritized to curb illegal tree cutting, charcoal production, and uncontrolled fires, coupled with strengthened local by-laws and community patrols. Equally important is the promotion of sustainable livelihood alternatives such as beekeeping, agroforestry, and fuel-efficient energy technologies, which can reduce community dependence on destructive practices, while enrichment planting and assisted natural regeneration could be employed in heavily degraded patches to restore forest structure and species composition. In addition, community participation must be enhanced by empowering local groups through Joint forest management (JFM), ensuring equitable benefit-sharing and joint responsibility in conservation. Finally, long-term ecological monitoring of species composition, structure, and carbon dynamics is essential to track disturbance impacts and inform adaptive management strategies that secure both ecological integrity and sustainable use of the reserve.
Disclosure
- All authors read and approved the final manuscript.
Conflicts of Interest
- The authors declare no conflicts of interest.
Funding
- This research was financially supported by the Tanzania Forest Fund (TaFF).
ACKNOWLEDGEMENTS
- We would like to express our gratitude to the Tanzania Forest Fund (TaFF) for their financial support, which made this study possible. We also extend our heartfelt thanks to the Biharamulo-Kahama Forest Reserve TFS staff for their support and cooperation throughout the study, in particular, Goodluck Melisa (District Forest Conservator of Biharamulo), George Thadeus Mwiga (Botanist), Ramadhani Kipokoso (assistant), Bakari Makeh, (GPS handlerer), Rashidi Said Jumanne (security), and Donard Rwabushirombo (Security). Special thanks also go to Jofrey Jacob, who provided essential support in map drawing and data analysis.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML