-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Agriculture and Forestry
p-ISSN: 2165-882X e-ISSN: 2165-8846
2014; 4(1): 24-33
doi:10.5923/j.ijaf.20140401.04
Effect of Plant Density and Weeding Regime on Population and Severity of Aphids (Aphis craccivora Koch) and Foliage Beetles (Ootheca mutabilis Sahl) on Cowpea in Sierra Leone
Sheku Max Kanteh1, Johnny E. Norman1, Joseph Sherman-Kamara2
1Department of Crop Protection, School of Agriculture, Njala University, Freetown, +232, Sierra Leone
2Department of Agricultural Engineering, School of Technology, Njala University, Private Mail Bag, Freetown, +232, Sierra Leone
Correspondence to: Sheku Max Kanteh, Department of Crop Protection, School of Agriculture, Njala University, Freetown, +232, Sierra Leone.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
Cowpea is one of the major grain legumes grown in Sierra Leone. This trial was conducted in the upland soils of Njala, southern Sierra Leone, in the 2011/2012 cropping season, to determine the effects of plant density and weeding regime on population and severity of aphids and foliage beetles on cowpea. Three plant populations (P1 = 456,522; P2 = 239,130 and P3 = 152,174 plants/ha) and three weeding regimes (no weeding, once at 3 weeks after sowing (WAS) and twice at 3 and 6 WAS) were investigated. The study provides the evidence that- aphids and foliage beetles are better controlled by weeding twice at 3 and 6 WAS, and that planting at lower density (152,174 plants/ha) has the potential to reduce the population of both foliage beetles and aphids. These results have implications for improving cowpea productivity in smallholder farms through the simple manipulation of plant population and weeding regime for the control of aphids and foliage beetles, and probably other cowpea insect pests.
Keywords: Aphids, Foliage beetles, Plant population, Population, Weeding regime
Cite this paper: Sheku Max Kanteh, Johnny E. Norman, Joseph Sherman-Kamara, Effect of Plant Density and Weeding Regime on Population and Severity of Aphids (Aphis craccivora Koch) and Foliage Beetles (Ootheca mutabilis Sahl) on Cowpea in Sierra Leone, International Journal of Agriculture and Forestry, Vol. 4 No. 1, 2014, pp. 24-33. doi: 10.5923/j.ijaf.20140401.04.
Article Outline
1. Introduction
- Cowpea (Vigna unguiculata (L) Walp) is an annual crop that is intensively and widely grown in Africa, and other humid and semi-arid regions of the world[1]. In Sierra Leone, cowpea is the second most important legume crop after groundnut (Arachis hypogea)[2],[3] and many local and improved varieties of the crop are cultivated across the country. Among these, are the popular local varieties like the Tabe (commonly known as black-eyed beans, because of the black colour of its hilium against the background of white testa) mostly grown in Southern Sierra Leone, and the variety Temne which is widely grown in Northern Sierra Leone, as well as other varieties like Holodome/Ladome, and Musia[3],[4]. Among the improved varieties that have been developed and introduced by SLARI (Sierra Leone Agricultural Research Institute) are the Slipea1/TVu 1190, Slipea 2/IT 86D-721 and Slipea 3/IT 86D-1010. Although cowpea can be grown throughout Sierra Leone, studies indicate that Moyamba District (Southern Sierra Leone) is the largest cowpea producing district in the country. The popularity of cowpea spans around many parts of Africa due mainly to its importance to many poor households, whose diets are predominantly carbohydrate rich foods with low amounts of protein[5]. Cowpea provides a cheap source of plant protein for many poor households that cannot regularly afford the high cost of popular protein sources like meat, fish and eggs[6]. In terms of chemical composition, the mature grains of cowpea contain about 23 to 25 % protein, 50 to 67 % starch. The crop is also rich in vitamins such as folic acid (important in preventing birth defects); and essential micronutrients such as iron, calcium, and zinc[7]. Besides being a cheap source of protein, several other benefits of cowpea have been reported. These include; food for man and livestock[8],[9],[10], important for controlling soil erosion, improves on soil fertility by providing ground cover and fixing nitrogen into the soil[11],[12],[13], suppresses weeds depending on growth habit, and also positively impacts on the health of women and children[50]. Women particularly value cowpea because it helps them to bridge the hunger months prior to the main cereal (rice) harvest. Trading fresh produce and processed food provide rural and urban women with the opportunity for earning cash income[14],[15],[16], [17]. The stalks are also very important fodder materials when harvested in the dry season (when fresh grass and other fodder materials are not available)[18]. Despite the high economic and nutritional value of cowpea in Sierra Leone, yields remain generally poor. reported average yield of 1.5 t/ha for improved cowpea varieties, compared to an average of about 4 t/ha for least developed countries. The situation is similar to what prevails in most parts of Africa, or even worse in some Sahelian countries where yields are hardly above 300kg/ha[19],[20]. Damages due to a wide spectrum of insect pests, attack by diseases and weeds infestation have been reported[21-24]. All of these contribute significantly to the widening gap between potential and actual yields of cowpea on smallholder plots. The key insect pests of cowpea identified so far include; cowpea aphid (Aphis craccivora Koch), foliage beetles (Ootheca mutabilis Sahl), flower bud thrips (Megalurothrips sjostedti Tryb), legume pod borer (Maruca vitrata Fab.), and a complex of pod sucking bugs which include Clavigralla tomentosicollis Stal, C. shadabi Doll, Anoplocnemis curvipes Fab and Riptortus dentines Fab [25-31]. Sixty to eighty percent of grain losses in cowpea have been attributed to the activities of insect pests[32], [33-35]. Of these, losses of up to 30 to 70% have been recorded on stored cowpea grains alone[36].Accumulating evidence[37], suggests that the effective control of major cowpea pests will lead to significant increase in yield. However, several limitations to chemical insecticides usage have been reported. These include; damages to the user or crop (phytotoxicity) due to excessive or unwisely use, environmental pollution, toxicity to mammals, destruction of beneficial organisms such as predators and parasitoids[13]. Other problems associated with chemical usage are cost of insecticides and application equipment[13]. Because these problems, a growing awareness of the dangers of chemicals in pest control has resulted in the recommendation that chemical application should be minimized, but not to be discarded[38-39].Ecological approach to insect pest management in some parts of the world, particularly in African countries to reduce pesticide usage tends to receive the attention of farmers[39]. Such approaches include cultural control practices; such as species diversification, manipulation of planting dates, intra-low spacing, intercropping, use of improved and resistant crop varieties, use of trap cropping and mulching[39-40]. The use of such cheap, sustainable and environmentally friendly options for the control of field pests should be well understood if their use must be popularized among poor smallholder farmers. With the extremely low yields reported in Sierra Leone and the dominant role of smallholders in food production in the country, efforts at maximizing cowpea yield through low cost insect pest control will be immensely beneficial. Thus optimizing basic cultural operations such as proper inter-row spacing and weeding regime for maximum cowpea yield could make a useful contribution to national food security. This study therefore aims at identifying the optimum weeding frequency and planting density that best fits for the control of aphids and foliage beetles on cowpea in Sierra Leone.
2. Materials and Methods
- This field trial was conducted in the upland ecology at Njala University, Njala Campus, Southern Sierra Leone in 2011/2012 cropping season. The Njala University is situated at 80 07' north-latitude and 120 05' west-longitudes[59]. The climate in the Njala area is characterized by a pronounced rainy season (May to November), and a pronounced dry season (December to April). The average total rainfall is estimated as 2,750.0 mm annually. The mean monthly air temperature is nearly constant, varying from 76.6o F(24.8°C) in August to 82.4o F (8.0°C) in March. The soils of Njala area have been classified as orthoxic palehumult belonging to the Njala series. The soil texture is usually gravely clay loam on the surface to gravely clay in the subsoil. The soils are never waterlogged and have a low nutrient status[41]. Two cowpea varieties (Slipea 2/IT86D-721 and Musai) were tested in this trial. Planting took place on 5th September, 2011. The experimental design was the randomized complete block design of eighteen (18) treatments, replicated three (3) times. Three (3) seeds per hole were planted, at a spacing of 50 x 30cm...152,174 plants/ha, 50 x 20 cm...239130 plants/ha (recommended spacing) and 50 x 10 cm....456,522 plants/ha; and thinned to two (2) seedling per hole at two (2) weeks after germination (WAG). Weeding treatments include; Wo-no-weeding, W1-weeding once at 3 WAS and W2-weeding twice at 3 and 6 WAS. In all cases, the inter-row spacing was 30.0 cm; 20.0 cm, and 10.0 cm, respectively. A 100 cm space was maintained between replicates, and 50 cm separated plots within each replicate. Plot sizes were 2.3 x 1.2 m, and the whole experimental field measured 49.9 x 5.6 m (279.44 m2).Data was collected at 2, 4, 6 and 8 weeks after sowing (WAS), within the middle row, targeting 5 tagged plant samples per plot and excluding the border plants. Data on key insect pests (aphids and foliage beetles) was collected during the early and late hours of the day, on each sampling day. Aphids, population density was rated based on a visual estimation scale of 1-6, following[21-23] procedure (where 1 = no aphids, 2 = 1-100 aphids per plant, 3 = 100-300 aphids, 4 = 300-600 aphids, 5 = 600-1000 aphids, and 6 = 1000 aphids per plant). Population density of foliage beetles was assessed by physically counting and recording the number of adult beetles found feeding on cowpea plants attacked. The % incidence was assessed on a 0-1 scale (where; 0 = clean plant with no signs of damage, and 1 = plant with signs of damage; expressed as percentage of the total number of sample plants with signs of damage over the total number of plants assessed); while the severity was assessed using a 0-5 visual rating scale (where; 0 = no plant with signs of damage/symptoms, 1=1-5% of plant with signs of damage/symptoms, 2=6-25% of plant with signs of damage/symptoms, 3=26-50% of plant with signs of damage/symptoms, 4=56-75% of plant with signs of damage/symptoms, and 5=76-100% of plant with signs of damage/symptoms). The statistical analysis technique applied was the analysis of variance (ANOVA) using Genstat Statistical Package Version 3. The least significant difference (LSD) test[42] was used for mean separation at 5% probability level (P<0.05).
3. Results
- Results of effects of weeding regime and weeks after sowing (WAS) on mean population of Aphis craccivora are shown in figures 1 and 2. The mean population of A. craccivora recorded in control plots differed significantly (P<0.05), but was not significantly different between plots weeded at 3 WAS and, 3 and 6 WAS. Plots weeded twice- at 3 and 6 WAS (1.058) and once-at 3 WAS (1.097) respectively supported the lowest mean population of A. craccivora, which also had fewer weeds; compared with the zero-weeded plots (control), which on the contrary supported the highest mean population of A. craccivora (1.469) and high weed density (Figure 1). Although there was no significant difference between the mean population of aphids recorded in plots weeded once-at 3 WAS and twice-at 3 and 6 WAS, the least mean population of aphids was however recorded in plots weeded at 3 and 6 WAS, relative to plots weeded at 3 WAS and the control (no-weeding) (Figure 1).
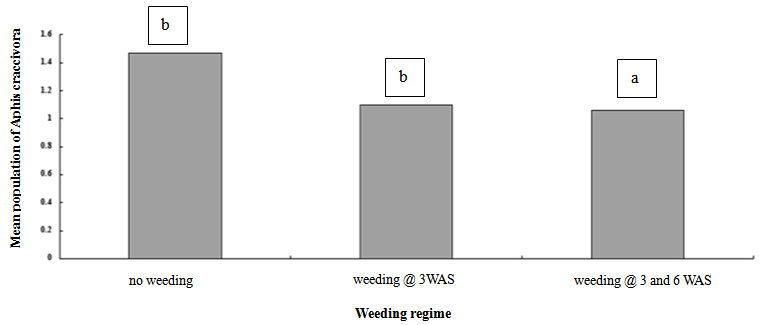 | Figure 1. Means in the same column followed by the same letter are not significantly different at P < 0.05 |
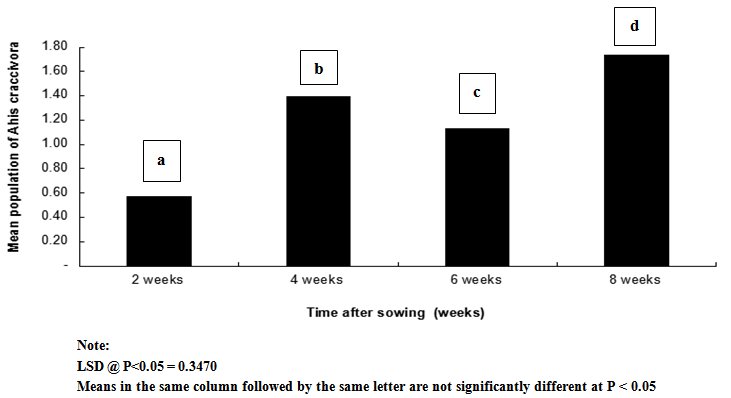 | Figure 2. Influence of time after sowing (weeks) on mean population of Aphis craccivora on cowpea |
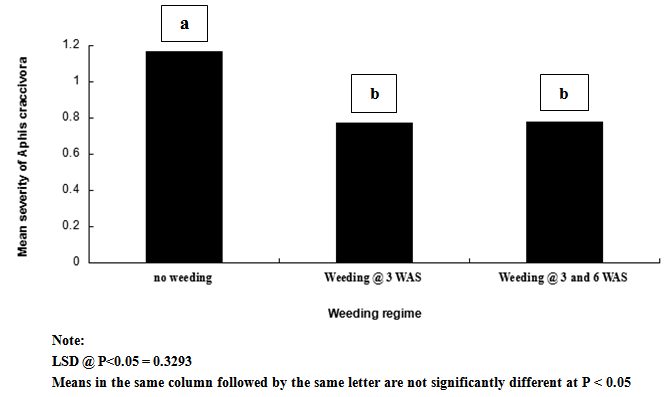 | Figure 3. Influence of weeding regime on mean severity of Aphis craccivora on cowpea |
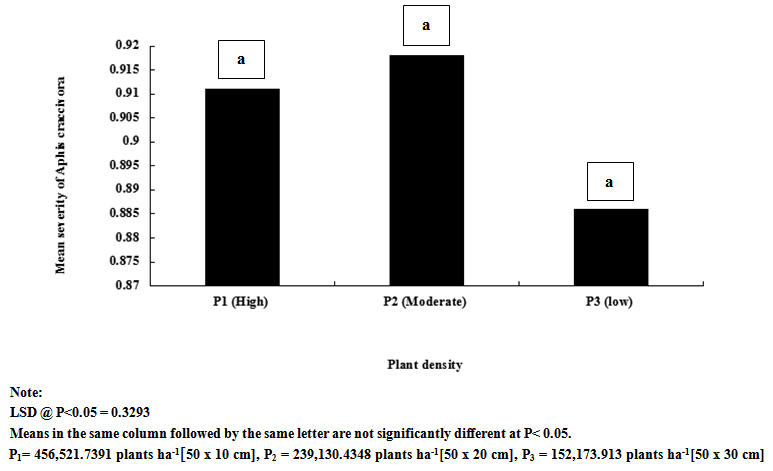 | Figure 4. Influence of plant density on mean severity of Aphis craccivora on cowpea |
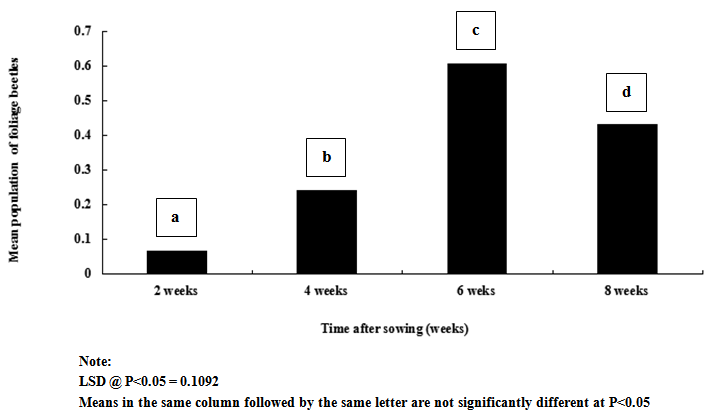 | Figure 5. Influence of weeks after sowing on mean population of foliage beetles on cowpea |
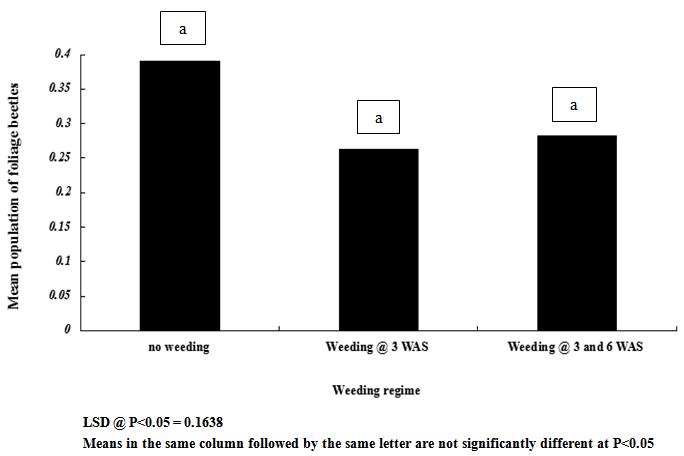 | Figure 6. Influence of weeding regime on mean population of foliage beetles on cowpea |
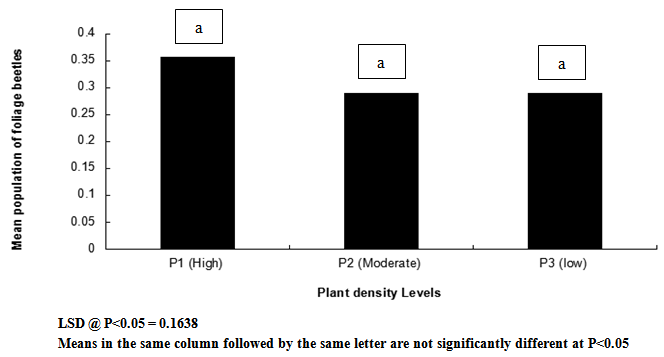 | Figure 7. Influence of plant density on mean population of foliage beetles on cowpea |
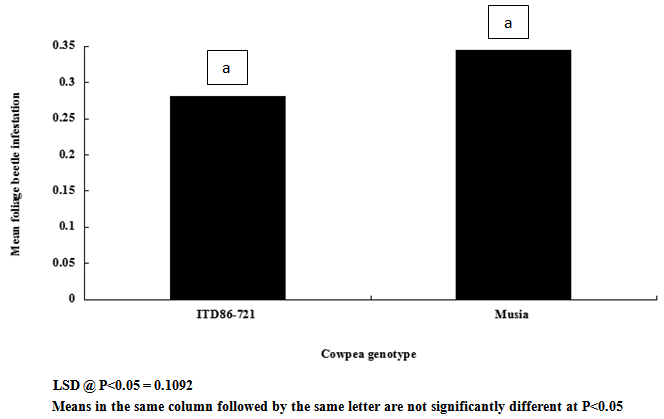 | Figure 8. Cowpea genotype response to foliage beetle infestation |
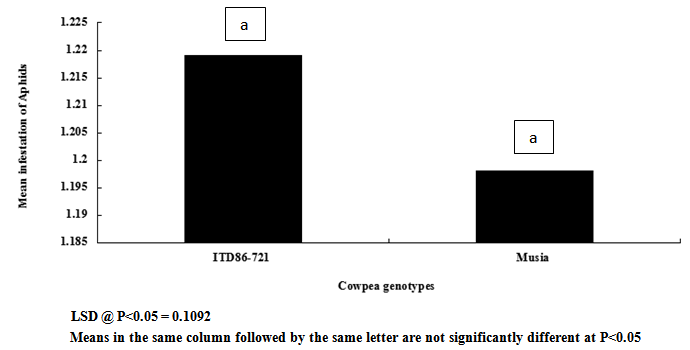 | Figure 9. Cowpea genotype response to aphids infestation |
4. Discussion
- Result of this study shows that weeding and planting density are effective cultural practices that can contribute reduction in the population and severity of key insect pests of cowpea. If weeding is delayed and plants are clustered together, it is without doubt that the spread and establishment of pests (including insects and diseases) is more pronounced, than in plots where weeding is not delayed and plants are not clustered together. In this study, weeds density was noted to be higher in un-weeded plots, but lower in plots weeded once at 3 WAS and twice at 3 and 6 WAS. Aphids and foliage beetles population were observed to be higher in un-weeded plots, and where plants were clustered together or with high plant density. From this result, it can be noted that increased in population density of aphids and foliage beetles are directly related to increase in weeds density. This result agrees with other studies that relate insect density and diversity with weed populations[43-45]. In general, weeds have been defined as higher plants in agro ecosystem, which are not sown, undesired, out of place or generally as plants which do more harm than good. Besides their impact on direct yield loss and deterioration of quality, weeds compete with crop plants for water, nutrients, light, space and/or carbon dioxide. Additionally, weeds create good habitat for a number of crop pests; notably insect pests and diseases. This might explain the reasons for increased in the population density and severity of Aphis craccivora and Ootheca mutabilis Sahl, as observed in this study. Weeding once at 3 WAS and twice at 3 and 6 WAS reduced the population and severity of the two major insect pests, compared with the control treatment (zero-weeding). On the whole, more aphids and foliage beetles were recorded in control (zero-weeded) plots than in plots weeded once and twice.[56] recorded 70.27% of A. craccivora, M. sjostedt and P. sabeaus in zero weeded plots. From result obtained in this study, it can thus be observed that weeding cowpea plots once-at 3 WAS or twice-at 3 and 6 WAS, has the potential to reduce the population of A. craccivora and (Ootheca mutabilis Sahl). It has been reported that the presence or absence of certain weeds may contribute to or reduce insect infestations in crops, and that management practices for weeds can affect insects population density. Refence[47-48] reported that reduced cowpea biomass, flowers, pods and grain yields were associated with cowpea plots where weeds and insect pests were not controlled. It has been reported that the activity of important insect pests of cowpea begin to colonize from 2 weeks after sowing (WAS), and population increases between 8 and 13 WAS, to coincide with flower, budding and pod formation stages, when there is abundant food resource availability[49]. In this study, it was observed that the population of Aphis craccivora increased progressively from 2, 4, 6 and 8 weeks after sowing (WAS), with the lowest recorded at 2 and peak population recorded at 8 weeks after sowing. Similar result was obtained for Ootheca mutabilis Sahl, whose population also increased progressively from 2, 4 and 6 WAS, but dropped at 8 WAS. From this result, it can be noted that the population of A. craccivora and O. mutabilis Sahl is lowest at the early growth stage of cowpea, but appears to increase as the plant progresses in growth. The significant increase in the population of these key insect pests of cowpea at 6 and 8 WAS, is an indication that the damage level or severity of infestations is expected to be very severe during these periods, compared to 2 and 4 WAS. Based on this finding, and as reported by[44] and[16], it is without doubt that A. craccivora and O. mutabilis Sahl are major threat to cowpea at 6-8 WAS. Reference[49] in his work in Uganda reported that at 2-3 weeks after cowpea emergence, more than 30% of the cowpea plants already had a high population density of 100-300 Aphis craccivora per plant. Though there was no statical difference between the mean population and severity of Aphis craccivora and Ootheca mutabilis Sahl on cowpea as influenced by the plant density levels, plots with higher plant density supported the highest population of these insects, while lower plant density supported the least population. This gives an indication that insect pest population density is directly related to planting density, and this implies that the more plants are clustered together, the more the spread and establishment of insect pests on cowpea. The observed trend, that increasing plant population density resulted in increased insect pest population, was consistent with the reports of[1]. They reported that Scaptomyza fly populations might build-up with increased hectares of meadowfoam (Limnanthes alba) cultivated.[52] also pointed out that multiple cropping (that leads to increased plant populations/ ha) could increase, or decrease the incidence of an insect or disease, or the population of its natural enemies, depending on the component species in the mixture, and the pest or disease concerned. Reference[53] however reported that in areas where shoot borer (Chilo infuscatellus Snellen) is a problem, narrow row spacing would tend to increase shoot borer damage in sugarcane crop than wider spacing.[54] evaluated the effect of row to row distance of onion plants on thrips (Thrips spp.) population; an inverse relation was found between increased line spacing and thrips population.
5. Conclusions and Recommendations
- Results of this study generally conclude that weeding twice-at 3 and 6 weeks after sowing (WAS), and planting at lower density (wider spacing) reduce the population and severity of Aphis craccivora and Ootheca mutabilis Sahl on cowpea. Aphids and foliage beetles population density increased in plots where plant density was high (i.e. clustered) compared to plots with low plant density (wider spacing). It can thus be concluded that planting cowpea at wider spacing (50 x 30 cm) has the potential to reduce Aphis craccivora and Ootheca mutabilis Sahl population density. It is recommended that for better control of aphids and foliage beetles on cowpea, with consequent increase in grain yields; lower plant population of 152,173.913 plants/ha----50.0 x 30.0 cm, and weeding cowpea plots twice-at 3 and 6 WAS, be adopted by smallholder farmers in Sierra Leone. More long-term studies should however be done to clearly identify the influence of plant population and weeding regime on the parameters investigated in this study. In addition, trials investigating the seasonal and ecological variation in the incidence and severity of infestations due to the insect pests investigated in this study should be considered in the future.
ACKNOWLEDGMENTS
- The authors wish to extend profound thanks and gratitude to Shekey Kanteh, Alex Sam, Abdul Seasy and Francis M. Kabba; for their assistance in field preparation and data collection. May God bless them.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML