-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Journal of Health Science
p-ISSN: 2166-5966 e-ISSN: 2166-5990
2026; 14(1): 1-5
doi:10.5923/j.health.20261401.01
Received: Apr. 20, 2026; Accepted: May 5, 2026; Published: May 12, 2026

From Monitoring to Quality: How Surveillance Systems Shape Indigenous Medicine in Kajiado and Tharaka Nithi Counties
Julius Kaluai1, Wanja Tenambergen2, Carol Kawila Kyalo1
1Kenya Methodist University, Department of Health Systems Management, Kenya
2Riara University, Kenya
Correspondence to: Julius Kaluai, Kenya Methodist University, Department of Health Systems Management, Kenya.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

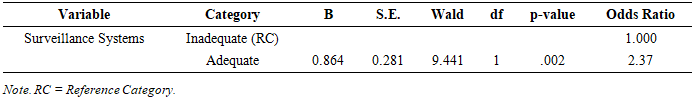
The growing reliance on indigenous medicine in Kenya has intensified concerns regarding its safety, consistency, and quality. Pharmacovigilance surveillance systems are central to monitoring adverse drug reactions and ensuring quality assurance; however, their effectiveness within indigenous medicine systems remains inadequately understood. This study examined the influence of surveillance systems on the quality of indigenous medicine in Kajiado and Tharaka Nithi Counties, Kenya. Anchored on Regulatory Compliance Theory and the Health Belief Model, the study adopted a mixed-methods research design. Quantitative data were collected from 334 respondents across regulators, community health promoters, practitioners, and manufacturers/suppliers, while qualitative data were obtained through key informant interviews. Descriptive statistics and binary logistic regression were used for quantitative analysis, while qualitative data were analysed thematically. The findings reveal that functional surveillance systems significantly increase the likelihood of good-quality indigenous medicine (OR = 2.37, p = .002). However, descriptive and qualitative evidence indicates that these systems are constrained by weak reporting mechanisms, unclear reporting channels, limited feedback loops, and poor stakeholder integration. The study concludes that while surveillance systems have strong potential to improve medicine quality, their operational effectiveness remains limited. Strengthening reporting infrastructure, enhancing feedback systems, and improving system integration are essential for improving the safety and quality of indigenous medicine in Kenya.
Keywords: Pharmacovigilance, Surveillance Systems, Indigenous Medicine, Quality, Adverse Drug Reactions, Kenya, Health Systems Strengthening
Cite this paper: Julius Kaluai, Wanja Tenambergen, Carol Kawila Kyalo, From Monitoring to Quality: How Surveillance Systems Shape Indigenous Medicine in Kajiado and Tharaka Nithi Counties, Journal of Health Science, Vol. 14 No. 1, 2026, pp. 1-5. doi: 10.5923/j.health.20261401.01.
1. Introduction
- Indigenous medicine remains a cornerstone of healthcare delivery in many low- and middle-income countries, particularly in sub-Saharan Africa, where it serves as a primary source of treatment for a large proportion of the population. The World Health Organization estimates that nearly 80% of people in developing countries rely on traditional or indigenous medicine for their primary healthcare needs [1,2]. This reliance is driven by a combination of structural and socio-cultural factors, including limited access to formal healthcare services, affordability constraints, and deeply rooted cultural beliefs that shape health-seeking behaviour. In Kenya, the use of indigenous medicine is similarly widespread, with over 70% of the population utilizing these services due to their accessibility, perceived effectiveness, and alignment with local cultural practices [1,3]. As such, indigenous medicine constitutes a critical but largely informal component of the national health system.Despite its widespread use, concerns about the safety, consistency, and quality of indigenous medicine persist. Unlike conventional pharmaceutical products, which undergo rigorous regulatory approval, standardized manufacturing, and clinical validation, indigenous medicines are often prepared using informal and highly variable processes. This variability introduces risks related to inconsistent dosing, contamination, and uncertain efficacy. These concerns are widely documented in the literature on herbal and traditional medicines [4]. Empirical studies highlight recurring challenges, including variability in active ingredients, lack of quality control, and undocumented adverse drug reactions [5,6]. Weak regulatory oversight and limited integration into formal health systems further compound these risks, creating gaps in accountability and quality assurance [3].Pharmacovigilance surveillance systems provide a critical mechanism for addressing these challenges. These systems enable the systematic detection, reporting, analysis, and prevention of adverse drug reactions, thereby generating evidence necessary for improving medicine safety and quality [1,7]. Their relevance is particularly pronounced in indigenous medicine contexts, where variability in preparation and use is high. By linking patient experiences, practitioner practices, and regulatory oversight, surveillance systems support more responsive, data-driven decision-making and continuous quality improvement [5].However, in Kenya and similar low-resource settings, pharmacovigilance systems for indigenous medicine remain underdeveloped and fragmented. Reporting pathways are often unclear or poorly understood, and many practitioners and community-level actors lack both awareness and incentives to report adverse events. This aligns with broader evidence showing that weak reporting culture and lack of incentives significantly undermine pharmacovigilance systems [8]. Feedback mechanisms, which are essential for reinforcing reporting behaviour and enabling system learning, are frequently weak or absent. In addition, coordination among key stakeholders, including regulators, healthcare providers, indigenous practitioners, and community health actors, remains limited. These structural and operational constraints contribute to significant underreporting and hinder the generation of reliable safety data needed for timely regulatory action [3,5].These challenges are particularly evident in Kajiado and Tharaka Nithi Counties, where indigenous medicine is widely practiced and deeply embedded in community health systems. The largely informal nature of practice, combined with limited regulatory oversight and weak surveillance infrastructure, results in fragmented and inconsistent safety monitoring. From a systems perspective, such fragmentation reflects broader weaknesses in health system integration and governance [9]. Adverse drug reactions are often managed at the household or practitioner level without formal documentation or escalation to regulatory authorities, raising concerns about the effectiveness of existing systems in safeguarding public health.Although pharmacovigilance has been extensively studied in relation to conventional pharmaceutical systems, its application within indigenous medicine contexts remains insufficiently understood, particularly in Kenya. Existing frameworks are often not adequately adapted to the unique characteristics of indigenous medicine, resulting in gaps in monitoring, reporting, and quality assurance. This lack of context-specific evidence limits the ability of policymakers and regulators to design effective interventions for strengthening surveillance systems in indigenous medicine settings.This study therefore examines the influence of pharmacovigilance surveillance systems on the quality of indigenous medicine in Kajiado and Tharaka Nithi Counties, Kenya. By integrating quantitative and qualitative evidence, the study provides a comprehensive assessment of the operational effectiveness of these systems and their role in shaping medicine quality outcomes. The findings aim to inform policy, strengthen regulatory frameworks, and support the integration of indigenous medicine into broader health system quality assurance efforts.
2. Methodology
- This study employed a convergent mixed-methods design, allowing for the simultaneous collection and integration of quantitative and qualitative data to provide a more nuanced understanding of how surveillance systems influence the quality of indigenous medicine. This approach enabled triangulation of findings and strengthened the validity and interpretability of results.Study settingThe study was conducted in Kajiado and Tharaka Nithi Counties, Kenya, regions where indigenous medicine remains widely utilized alongside formal healthcare systems. These counties were purposively selected due to their active engagement in indigenous medicine practices, diversity of stakeholders, and ongoing policy interest in strengthening regulatory and surveillance frameworks.Study population and samplingThe study targeted key stakeholders involved in the indigenous medicine ecosystem, including regulators, community health promoters, traditional medicine practitioners, and manufacturers/suppliers. A stratified random sampling approach was used to ensure proportional representation across these stakeholder groups. Within each stratum, respondents were randomly selected from available stakeholder lists provided by county health departments and relevant professional associations. The final sample comprised 334 respondents for the quantitative component. For the qualitative component, key informant interviews (KIIs) were conducted with purposively selected participants based on their expertise and role in surveillance and regulatory processes, ensuring depth of insight.Data collectionQuantitative data were collected using a structured questionnaire administered through trained research assistants. The tool captured key dimensions of surveillance systems, including: reporting mechanisms for adverse events, feedback and response systems, and stakeholder coordination and integration. Qualitative data were obtained through semi-structured key informant interviews, guided by an interview protocol designed to explore experiences, perceptions, and systemic challenges related to pharmacovigilance and quality assurance in indigenous medicine.Measurement of variablesThe dependent variable was the quality of indigenous medicine, operationalized as a binary outcome (good vs. poor quality) based on predefined criteria including safety, consistency, and adherence to recommended practices. The main independent variable, surveillance system adequacy, was similarly categorized (adequate vs. inadequate) based on composite indicators derived from reporting efficiency, feedback mechanisms, and stakeholder engagement. Data quality assuranceTo enhance reliability and validity, the data collection instruments underwent pre-testing in a comparable setting. Feedback from the pre-test informed refinement of the tools. Internal consistency of scales was assessed using Cronbach’s alpha, while content and face validity were ensured through expert review by specialists in public health, pharmacovigilance, and health systems research. Research assistants received standardized training to ensure consistency in data collection procedures.Data analysisQuantitative data were analysed using SPSS version 26. Descriptive statistics (frequencies, proportions, means) were used to summarize respondent characteristics and key variables. A binary logistic regression model was employed to estimate the association between surveillance system adequacy and the likelihood of achieving good-quality indigenous medicine. Adjusted odds ratios (AORs) with 95% confidence intervals (CIs) were reported, and statistical significance was set at p < 0.05. Model diagnostics, including goodness-of-fit tests and multicollinearity checks, were performed to ensure robustness.Qualitative data were transcribed verbatim and analysed using thematic analysis. An inductive coding approach was applied, involving open coding, development of categories, and identification of emerging themes. Findings were then integrated with quantitative results during interpretation to provide a comprehensive explanation of observed patterns.Ethical considerationsEthical approval was obtained from a recognized Kenya Methodist University Science, Ethics and Research Committee (SERC) and a research permit was obtained from the National Commission for Science, Technology and Innovation (NACOSTI), Kenya. Permission to conduct the study was also obtained from relevant county authorities. All participants provided written informed consent prior to participation. Confidentiality was maintained through anonymization of data, and participation was entirely voluntary, with the right to withdraw at any stage without penalty.
3. Results
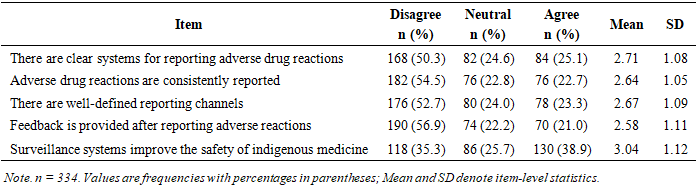
- The descriptive findings in Table 1 show a fairly weak and underperforming pharmacovigilance surveillance systems, particularly in relation to the reporting and follow-up of adverse drug reactions (ADRs).
|
|
4. Conclusions
- The findings demonstrate that pharmacovigilance surveillance systems play a significant and measurable role in improving the quality of indigenous medicine. The logistic regression results show that adequate surveillance systems more than double the likelihood of achieving good-quality indigenous medicine (OR = 2.37, p = .002), confirming their critical importance as a health system function. The findings show that the effectiveness of the pharmacovigilance surveillance systems is weak because its full operational performance remains constrained by unclear reporting mechanisms, inconsistent reporting of adverse drug reactions (ADRs), absence of effective feedback loops, and limited coordination among stakeholders. These weaknesses undermine trust, discourage reporting behavior, and prevent the generation of actionable safety data. Importantly, stakeholders still recognize the value of surveillance systems, suggesting that the issue is not conceptual acceptance but system design and implementation failure. Overall, the study concludes that while pharmacovigilance systems have strong potential to enhance safety and quality, their current effectiveness is constrained by systemic and operational deficiencies, limiting their contribution to health system strengthening.
5. Recommendations
- To bridge the gap between potential and performance, the following actions are recommended:1. Strengthening Reporting Systems and Infrastructurea. The national government regulator should develop clear, standardized, and user-friendly adverse drug reaction (ADR) reporting mechanisms tailored to indigenous medicine contexts.b. Establish functional feedback mechanisms by creating structured feedback loops to ensure that reported cases lead to visible action.2. Enhancing Stakeholder Coordination and System Integrationa. Promote collaboration among regulators, traditional practitioners, community health promoters, and manufacturers to enhance coordination and information sharing.b. Develop integrated systems to improve information flow and facilitate comprehensive safety monitoring across stakeholders.3. Capacity Building and Community Engagementa. Train practitioners and community-level actors on pharmacovigilance principles, reporting procedures, and the importance of surveillance systems in improving medicine safety.b. Promote community engagement by educating patients and communities on the importance of reporting adverse effects to encourage bottom-up participation in surveillance systems.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML