-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Journal of Health Science
p-ISSN: 2166-5966 e-ISSN: 2166-5990
2014; 4(3): 64-71
doi:10.5923/j.health.20140403.03
Protective Effect of Exendin-4 (Glp-1 Analogue) in Acute Kidney Injury in Experimental Animals
Eslam K. Fahmy1, 2, Husam M. Edrees1, 3
1Department of Physiology, Faculty of Medicine, Zagazig University, Zagazig, Egypt
2College of Medicine, Sulaiman Alrajhi Colleges (SRC), Alqassim, Saudi Arabia
3College of Public Health and Health Informatics, Qassim University, Saudi Arabia
Correspondence to: Eslam K. Fahmy, Department of Physiology, Faculty of Medicine, Zagazig University, Zagazig, Egypt.
| Email: |  |
Copyright © 2014 Scientific & Academic Publishing. All Rights Reserved.
A prophylactic concept should be applied in cases of drug induced and conditions causing acute kidney injury which is a serious and common danger. Epidemiological studies concluded that smoking is an important remediable risk factor for development of acute and chronic kidney disease to end-stage renal disease. High-dose therapy with cisplatin which is a potent antitumor drug is limited by its cumulative nephrotoxicity and neurotoxicity. Exhaustive exercise or overload training causes histological changes in kidney cell manifesting as hematuria, proteinuria and acute renal failure. Exendin-4 (GLP-1 analogue) treatment effectively protected the kidney from injury in a rodent model of renal ischemia reperfusion injury through the suppression of inflammatory reaction, apoptosis and oxidative stress. The aim of this research is to clarify the protective effects of Exendin-4 on different causes of acute kidney injury of experimental animals and compare the injurious effect of nicotine, cisplatin and exhaustive exercise. Design: 70 healthy adult male albino rats were used to study the protective effect of Exendin-4 (administered at a dose of 25 nmol/kg in saline for 11 days) against injurious effect of nicotine bitartrate (at a dose of 200 μg/ml in a 2% saccharine solution as drinking source for 4 weeks), cisplatin (a single intraperitoneal injection at a dose of 7 mg/kg body weight) and exhaustive exercise for 4 weeks. Kidney injury was assessed by analysis of creatinine, BUN, serum KIM and urinary KIM. Results: The results of this study showed that in group IIb (Nicotine/Exendin-4 treated group) and group IVb (Exhaustive exercise/Exendin-4 treated group), the mean values were found to be significantly decreased when compared with non exendin treated groups. On comparing group IIIa (Cisplatin treated group) with that of both group IIa (Nicotine treated group) and group IVa (Exhaustive exercise group), it was found that the mean values were found to be significantly increased for all parameters. However, in group IIIb (Cisplatin/Exendin-4 treated group), the mean values were found to be non-significant for all test parameters when compared with that of group IIIa (Cisplatin treated group). Conclusion:It was observed that exendin-4 has a protective effect against kidney injury induced by nicotine and exhaustive exercise but not against cisplatin.
Keywords: Acute kidney injury, Exendin-4, Nicotine, Cisplatin, Exhaustive exercise, Kidney injury molecule (KIM-1)
Cite this paper: Eslam K. Fahmy, Husam M. Edrees, Protective Effect of Exendin-4 (Glp-1 Analogue) in Acute Kidney Injury in Experimental Animals, Journal of Health Science, Vol. 4 No. 3, 2014, pp. 64-71. doi: 10.5923/j.health.20140403.03.
Article Outline
1. Introduction
- Acute kidney injury (AKI) is a common complication in hospitalized patients and significantly contributes to morbidity and mortality. Recent studies have further demonstrated that AKI was evident in around 20% of patients who died in hospitals and up to 50% of patients in the intensive care unit (ICU) [1].Exendin-4 (GLP-1 analogue), has been reported to have multiple cellular protective effects, including the protection of endothelial cells against senescence mainly through anti-oxidative and anti-inflammatory processes [2, 3]. It is rational to hypothesize that the inflammatory reaction and oxidative stress from acute renal ischemia reperfusion (IR) injury may be alleviated by Exendin-4 treatment through the induction of GLP-1 receptor (GLP-1R) expression [4, 5].Kidney injury molecule-1 (KIM-1) is a type 1 membrane protein that is not expressed in normal kidney but is markedly upregulated in the injured proximal tubular epithelial cells of the human and rodent kidney in ischemic and toxic AKI. KIM-1 is also expressed in other conditions where proximal tubules are dedifferentiated e.g. renal cell carcinoma [6, 7].Smoking/tobacco use is a serious health issue worldwide with a recently recognized impact on kidney health [8]. Nicotine (NIC), a major alkaloid of tobacco smoke, links smoking/tobacco use to renal injury [9]. Epidemiologic and experimental data show that smoking/NIC augments progression of chronic kidney disease [8-10] and also exacerbates acute renal ischemia/reperfusion injury (IRAKI) [11]. Importantly, NIC replacement therapy causes abnormalities in renal hemodynamics similar to smoking and is associated with increased patient mortality in intensive care units, perhaps in part via enhanced renal toxicity [12-14].Cisplatin, a widely used anti-neoplastic agent, is primarily used in the treatment of a variety of solid tumors [15]. However, the clinical usefulness of cisplatin has been seriously restricted because of its nephrotoxic side effects [16]. The major site of renal injury is the proximal tubules in the outer medulla of the kidney. The toxicity in epithelial cells is morphologically characterized by tubular necrosis, loss of microvilli, alterations in number and size of lysosomes, and mitochondrial vacuolization. These structural alterations are accompanied by functional disturbance of various cell organelles [17]. Several investigators have suggested different mechanisms by which cisplatin selectively kills the proximal tubule cells. It was hypothesized that cisplatin is activated in the kidney to toxic metabolite through a platinum-glutathione conjugate, and then to a cysteinyl-glycine-platinum-conjugate, which is further processed to a cysteine conjugate which is a metabolically reactive thiol [18]. In addition, two distinct patho-physiological mechanisms have been recognized as promoters of cellular damage, i.e. inhibition of protein synthesis and glutathione depletion [19, 20]. Moreover, many evidences have been accumulated that this side effect is closely related to reactive oxygen species (ROS) which cause mitochondrial damage, inhibition of membraneous transport proteins and lipid peroxidation [17].Long term high-intensity exercise or overload especially in high-intensity work, military training and competitive sports will inevitably cause overload or hypoxia that exceeds the endurance of the body. This will cause qualitative change from physiologic stimuli to pathologic stimuli [21]. Eventually, the systemic function will be affected, such as rhabdomyolysis and injuries of heart, liver and neural system. Kidney is one of the vulnerable organs suffered from the high- intensity exercise or overload training, manifesting as cystorrhexis, hematuria, proteinuria, acute renal failure [22, 23]. A considerable damage in kidney cells was presented in histological and ultrastructural examinations. Normally, the blood flow in the kidney is about 25% of the cardiac output. During exercise, the metabolic rate of muscle is increased and more blood supply is required [24]. In exhaustive situation, blood flow redistribution among the organs occurs due to the emergency response, which is activated by increasing in secretion of adrenaline and rennin angiotensin. Consequently, the renal artery contracted and the renal blood flow decreased, eventually the renal injury aggravated, especially when the exercise lasts and the intensity increases further on [25, 26]. The mechanisms were mainly related to the effect of reactive oxygen species, intracellular calcium overload, inflammatory corpuscle, inflammatory cascade reaction, nitric oxide, endothelin, free radical changes and cell apoptosis in renal tubular epithelial cells [27].Therefore, the aim of this study is to clarify the protective effects of Exendin-4 on different causes of acute kidney injury of experimental animals and compare the injurious effect of nicotine, cisplatin and exhaustive exercise.
2. Material and Methods
2.1. Experimental Animal
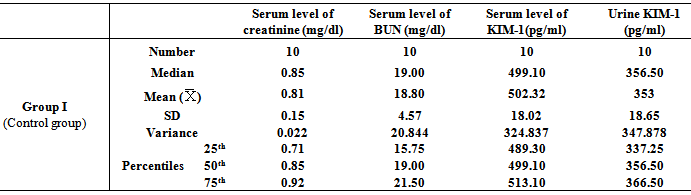
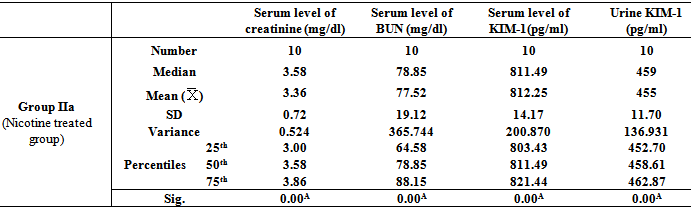
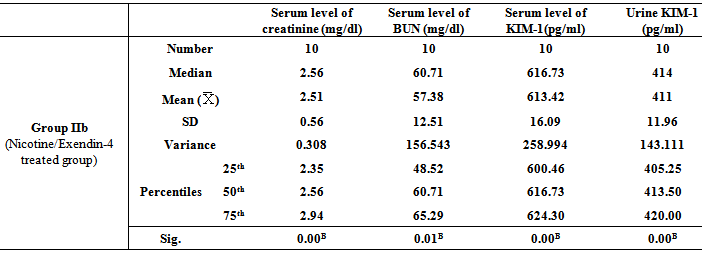
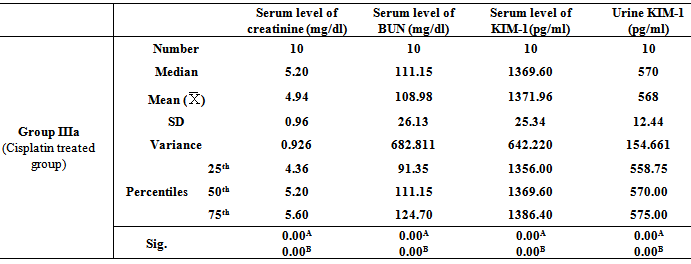
- The current study was carried on 70 male adult albino rats 12 – 15 weeks old with body weight 200-250 gm, were obtained from the animal house from faculty of veterinary medicine of Zagazig University. Rats were kept in steel wire cages (10/cage) in the physiology research laboratory in faculty of medicine of Zagazig University under hygienic conditions, rats were kept on the normal diet, which consisted of mixed commercial rat laboratory chow and supplied in separate clean containers. Animals had free access to water, kept at room temperature and were maintained on a 12 hr light/dark cycle. The rats were accommodated to laboratory conditions for two weeks before the experiments were going on. Rats were numbered and divided into the following groups:Group I: (Control group- n = 10): rats were served as a control group; they were fed on normal diet. Rats received a 2% saccharine solution for 4 weeks.Group IIa: (Nicotine treated group- n = 10): rats were fed on normal diet. They received nicotine bitartrate in a 2% saccharine solution at 200 μg/ml concentration as their drinking source for 4 weeks [28].Group IIb: (Nicotine / Exendin -4 treated group- n = 10): rats were fed on normal diet. They received nicotine bitartrate in a 2% saccharine solution at 200 μg/ml concentration as their drinking source for 4 weeks [28]. They received Exendin -4 (25 nmol/kg in saline) over the last 11 days of nicotine treatment [29].Group IIIa: (Cisplatin treated group- n = 10): rats were fed on normal diet. Kidney injury was induced by a single intraperitoneal (i.p.) injection of cisplatin in a dose of 7 mg/kg body weight [30].Group IIIb: (Cisplatin / Exendin-4 treated group- n = 10): rats were fed on normal diet. They received Exendin -4 (25 nmol/kg in saline) over 11 days starting one week before cisplatin injection [29]. Kidney injury was induced by a single intraperitoneal (i.p.) injection of cisplatin in a dose of 7 mg/kg body weight [30].Group IVa: (Exhaustive exercise group- n = 10): rats were fed on normal diet. Rats were subjected to exhaustive exercise for 4 weeks [21].Group IVb: (Exhaustive exercise / Exendin -4 treated group- n = 10): rats were fed on normal diet. Rats were subjected to exhaustive exercise for 4 weeks [21]. They received Exendin -4 (25 nmol/kg in saline) over the last 11 days of exhaustive exercise [29].
2.2. Drugs and Chemicals
2.2.1. Nicotine Hydrogen Tartrate
- (Sigma Aldrich, St. Louis, Mo, USA, 36733-1G (99% nicotine).
2.2.2. Cisplatin
- (Sigma Aldrich, St. Louis, Mo, USA).
2.2.3. Exendin-4
- (Sigma Aldrich, St. Louis, Mo, USA, E7144).
2.3. Methods
2.3.1. Exhaustive Swimming Exercise
- All the rats of group IV performed an exhaustive swimming exercise for 4 weeks in an acrylic plastic pool (90x45x45 cm) filled with water maintained at a temperature of 36±2℃. The water depth, 35 cm, was set so that the rats could not rest by supporting their tails on the bottom of the pool. Each rat had a weight attached (5% body weight) to its tail for the duration of the swim-to-exhaustion exercise. The animals were assessed as being exhausted when they failed to rise to the surface of the water to breathe within 7 sec. The exhaustive swimming time was recorded in min for each rat. [21].
2.3.2. Method of Urine Collection
- Method of urine collection was done using multiple animal method 24 hours before rats were sacrificed, involved partitioning rat cage into ten compartments (three X-shaped structures fabricated with cardboard) [31]. Each compartment consists of an upper chamber with a collection funnel, collecting tubes, and a simple stand. All parts of the cage can be disassembled and cleaned with warm soapy water or detergent solution [32]. Aseptically-collected urine samples were centrifuged, and an aliquot was immediately stored at −20℃ until KIM-1 measurement.
2.3.3. Collection of Blood Samples
- At the end of the experimental period and after overnight fasting, at 8:00a.m, the animals were sacrificed to get a blood sample for centrifugation and analysis of serum creatinine, blood urea nitrogen (BUN) and kidney injury molecule (KIM-1). Blood samples were allowed to clot for 2 hours at room temperature before centrifuging for 20 minutes at approximately 500 rpm [33]. The separated serum was stored at -20℃. Repeated freezing and thawing was avoided [34].
2.4. Laboratory Analysis
2.4.1. Estimation of Serum Creatinine Level
- According to Murray et al. [35] using creatinine kits supplied by SPINREACT, S.A.U. Ctra.Santa Coloma, 7E-17176 SANT ESTEVE DE BAS (GI) SPAIN.
2.4.2. Estimation of Serum Blood Urea Nitrogen (BUN) Level
- According to Murray et al. [35] using urea kits supplied by SPINREACT, S.A.U. Ctra.Santa Coloma, 7E-17176 SANT ESTEVE DE BAS (GI) SPAIN.
2.4.3. Estimation of Serum Kidney Injury Molecule (KIM-1) Level
- Using rat kidney injury molecule-1, KIM-1/ TIM-1/ HAVCR1 ELISA kit which is an in vitro enzyme-linked Immunosorbent assay for the quantitative measurement of samples in serum and urine, supplied by Putus Macromolecular Sci.&Tech. Ltd. Huaguang Road 18, East Lake New Technology Development Zone 430074 Wuhan (Hubei province), China [36].
2.4.4. Estimation of Urine Kidney Injury Molecule (KIM-1)
- Using rat kidney injury molecule-1, KIM-1/ TIM-1/ HAVCR1 ELISA kit [36].
2.5. Statistical Analysis
- Results are presented as mean ± standard error of the mean. Statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS), version 17.0 (SPSS Inc., Chicago, IL, United States). Repeated measures of analysis of variance (ANOVA) statistical analysis were applied followed by the Student-Newman-Keuls post hoc test. P value <0.05 was considered to be statistically significant.
3. Results
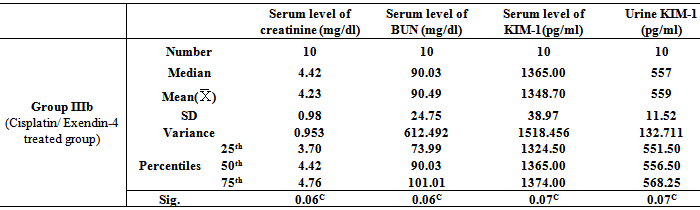
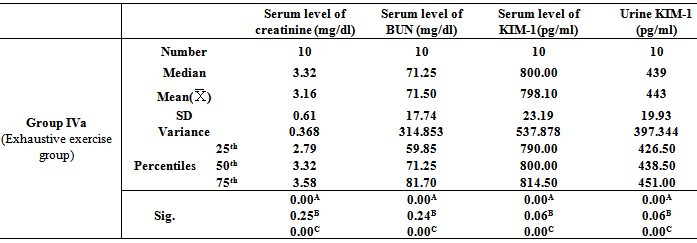
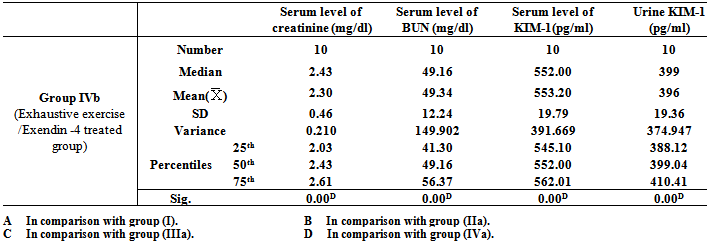
- Table 1, 2, 3, 4, 5, 6 and 7 show serum level of creatinine (mg/dl), serum level of blood urea nitrogen (BUN) (mg/dl), serum level of kidney injury molecule-1 (KIM-1) (pg/ml)and urine level of kidney injury molecule-1 (KIM-1) (pg/ml) in all studied groups expressed as mean ± standard deviation.In all groups, it was found that the mean values were found to be significantly increased (P< 0.001) for all parameters when compared with that of group I (Control group).In group IIb (Nicotine/Exendin-4 treated group), the mean values were found to be significantly decreased (P< 0.001, P< 0.01, P< 0.001 and P< 0.001 respectively) when compared with that of group IIa (Nicotine treated group).On comparing group IIIa (Cisplatin treated group) with that of both group IIa (Nicotine treated group) and group IVa (Exhaustive exercise group), it was found that the mean values were found to be higher (P< 0.001) for all parameters. However, in group IIIb (Cisplatin/Exendin-4 treated group), the mean values were found to be non-significant for all test parameters when compared with that of group IIIa (Cisplatin treated group).Moreover, the mean values were found to be non-significant for all test parameters on comparing group IVa(Exhaustive exercise group) with group IIa (Nicotine treated group). However, in Group IVb (Exhaustive exercise / Exendin -4 treated group), it was found that the mean values were found to be significantly decreased (P< 0.001) for all parameters when compared with that of Group Iva (Exhaustive exercise group).
|
|
|
|
|
|
|
4. Discussion
- Acute kidney injury (AKI) is due to a variety of conditions and has serious consequences. It is widely recognized that AKI leads to high morbidity and mortality in hospitalized patients. Therefore, there is an urgent need for effective therapy. Many animal and clinical studies have demonstrated conclusively the benefit of pharmacologic prophylaxis for AKI [37]. Exendin-4 (glucagon-like peptide-1 receptor (GLP-1R) activation exerts protective effects against reactive oxygen species [5].Several potential mediators of smoking-induced renal damage have been discussed, which can be subdivided into non-haemodynamic and haemodynamic mechanisms including smoking-induced activation of the renin–angiotensin system, oxidative stress-dependent generation of reactive oxygen species (ROSs) causing mitochondrial depolarization and consequent injury in cultured proximal tubule cells [38]. The anticancer drug cisplatin (CP) causes nephrotoxicity through different mechanisms, including the production of nephrotoxic metabolites, vascular injury, inflammation, generation of free radicals and apoptotic pathways [39]. Exhaustive exercise in the rat also decreased renal cortical NOS and SOD activity and caused significant renal tubular damage. Exercise-induced acute kidney injury (AKI) reflects renal ischemia caused by rhabdomyolysis (the breakdown of muscle fibers and/or severe volume depletion [40].In the light of previous data, the present study was designed to clarify the protective effects of Exendin-4 (GLP-1 analogue) against nephrotoxicity induced by nicotine, cisplatin and exhaustive exercise.The results of the present study revealed that the mean values in all groups were found to be significantly increased (P< 0.001) for all parameters when compared with that of group I (Control group). Our results regarding renal effect of nicotine studied in group IIa (Nicotine treated group) are in agreement with previous clinical and experimental studies which indicated that nicotine is at least in part responsible for the deleterious effects of cigarette smoking in the progression of CKD. The renal effects of nicotine are also linked to increased generation of reactive oxygen species and activation of pro-fibrotic pathways [41], increased fibronectin and NADPH oxidase 4 [42]. However, an interesting contradictory study claimed that nicotine pretreatment protected mice from renal dysfunction through the alpha7nAChR, as attested by the absence of protection in alpha7nAChR-deficient mice. Additionally, nicotine significantly reduced tubular damages (associated with a decrease in tubular cell apoptosis and proliferative response), prevented neutrophil infiltration and decreased productions of TNF-alpha and other proinflammatory cytokines [43].Additionally, our results regarding renal effect of cisplatin studied in group IIIa (Cisplatin treated group) are in line with Kilic et al. [30] who revealed that cisplatin causes damage of the proximal tubular cells as approved by histological analysis by enhancing production of ROS and inducing apoptosis. Another study done by AL-Kharusi et al. [39] showed that histopathological examination revealed acute tubular necrosis and many apoptotic cells in 60% of the examined sections of the cortical area of the kidney in rats treated with CP.Moreover, our results regarding renal effect of exhaustive exercise studied in group IVa (exhaustive exercise group) are in agreement with Lin et al. [44] who revealed that kidney function and tissue integrity were strongly affected by exhaustive exercise, as manifested by increased urine protein, serum urea nitrogen and serum creatinine, decreased renal SOD, NO and NOS activities, and marked renal cells apoptosis. Another study revealed that exercise rendered the kidney more vulnerable to acute kidney injury. The vulnerable rats exhibited exercise-dependent loss of the protective renal proteins critical for proper blood vessel health [40].In group IIb (Nicotine/Exendin-4 treated group), the mean values were found to be significantly decreased (P< 0.001, P< 0.01, P< 0.001 and P< 0.001 respectively) when compared with that of group IIa (Nicotine treated group). Moreover, in group IVb (Exhaustive exercise /Exendin -4 treated group), it was found that the mean values were found to be significantly decreased (P< 0.001) for all parameters when compared with that of group IVa (exhaustive exercise group). These results are in agreement with Xu et al. [45] who stated that exendin-4 (GLP-1 analogue) modulates renal function by inhibiting cell proliferation and fibronectin secretion. It also causes phosphorylation of AMPK which resulted in the degradation of fibronectin. Mima et al. [46] revealed that the renal protective effects of GLP-1 were mediated via protein kinase C (PKC) β2 activation and the increased degradation of GLP-1R in the glomerular endothelial cells.However, in Group IIIb (Cisplatin / Exendin-4 treated group), the mean values were found to be decreased but non-significant for all test parameters when compared with that of group IIIa (Cisplatin treated group). Our result is contradictory to the result of Katagiri et al. [47] who revealed that GLP-1 receptor agonist exendin-4 reduced cisplatin-induced renal injury and apoptosis, and suppression of renal GLP-1 receptor expression in vivo. Another contradictory result revealed by Yang et al. [48] who demonstrated that preconditional activation of the GLP-1 receptors with exendin-4 in the kidney significantly protected against ischemia-reperfusion injury in rats by increasing HO-1 expression. This contradiction may be due to pretreatment with Exendin-4, this resulted in prophylactic effect against cisplatin-induced renal injury.Interesting finding of our study was exposed on comparing group IIIa (Cisplatin treated group) with that of both group IIa (Nicotine treated group) and Group Iva (Exhaustive exercise group), it was found that the mean values were found to be higher (P< 0.001) for all parameters. However, the mean values were found to be non-significant for all test parameters on comparing Group IVa (exhaustive exercise group) with group IIa (Nicotine treated group). This finding indicates that the nephrotoxic effect of cisplatin is more prominent than both nicotine and exhaustive exercise.
5. Conclusions
- The present study suggests that treatment with exendin-4 (glucagon-like peptide-1 analogue) has protective effect against acute kidney injury induced by nicotine and exhaustive exercise; however, it has no significant effect against acute kidney injury induced by cisplatin with more observable nephrotoxic effect for cisplatin more than nicotine and exhaustive exercise. Thus, exendin-4 may represent a prophylactic option against acute kidney injury induced by smoking (nicotine) and exhaustive exercise.
ACKNOWLEDGMENTS
- The authors would like to acknowledge the supporting team of technicians in the Department of Physiology and Medical Biochemistry, Zagazig University for their assistance in the experimental work and laboratory analysis. This research received no specific grant from any funding agency in the public commercial or not-for-profit sectors.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML