-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Food and Public Health
p-ISSN: 2162-9412 e-ISSN: 2162-8440
2022; 12(1): 7-13
doi:10.5923/j.fph.20221201.02
Received: Mar. 30, 2022; Accepted: Apr. 16, 2022; Published: Apr. 21, 2022

Health Risk Assessment of Heavy Metals Via Consumption of Cassava, Cultivated on Reclaimed Mining Land Sites in Prestea-Huni Valley District, Ghana
Victus Bobonkey Samlafo 1, Kofi Essel 2, Bright Ankudze 1
1Department of Chemistry Education, University of Education, Winneba, Ghana
2Archbishop Porter Girl’s Secondary School, Takoradi
Correspondence to: Victus Bobonkey Samlafo , Department of Chemistry Education, University of Education, Winneba, Ghana.
| Email: |  |
Copyright © 2022 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The soil determines the quality of food and hence the health of the individuals. However, most lands in the District are under mining or concessions to the detriment of livelihood. This situation resulted in the cultivation of food crops on reclaimed mining land sites contrary to the accepted norm. To assess the human health risk and the quality of cassava cultivated on reclaimed mining land sites in Prestea-Huni Valley District, soil and cassava samples were taken randomly in separate polyethylene bags in triplicates from four farms located on reclaimed mining land sites within the Prestea-Huni Valley District. Atomic Absorption Spectrophotometric (AAS) technique was employed in the determination of the metals after digestion. The precision and accuracy of the analytical methods were evaluated by analysing IAEA-soil-7 and 1547-Peach leaves, which are standard reference materials with recoveries ranging from 87% to 98%. The trend in metal tolerance for both the cassava tuber and its peel were similar and in the order Cd > Pb>Cr>Zn >As. Both soil Pollution Load Index (PLI) and Total Hazard Quotient (THQ) were below their respective values. Using the health index of 0.8017< 1.0 () as a criterion for human health assessment, the cassava cultivated on reclaimed mining land sites in the Prestea-Huni Valley District, are safe and of high quality, since the health index was below the threshold level of 1.0, a level below which the cassava is deemed fit for human consumption.
Keywords: Prestea-Huni Valley, Cassava, Peel, Heavy metals, Health index, Bioaccumulation
Cite this paper: Victus Bobonkey Samlafo , Kofi Essel , Bright Ankudze , Health Risk Assessment of Heavy Metals Via Consumption of Cassava, Cultivated on Reclaimed Mining Land Sites in Prestea-Huni Valley District, Ghana, Food and Public Health, Vol. 12 No. 1, 2022, pp. 7-13. doi: 10.5923/j.fph.20221201.02.
Article Outline
1. Introduction
- The increasing demand for food and food safety is a major public concern and has drawn the attention of both governments and scientists to the risks associated with the consumption of contaminated food (i.e pesticides, heavy metals, and toxins in food) [1]. Cassava (Manihot esculenta Crantz) belongs to the family Euphorbiaceae. It is cultivated as a monocrop or intercropped with other food crops, either as a dominant or subsidiary crop. In terms of the quantity produced, cassava is the most important root crop followed by yam and cocoyam, but ranked second to maize in terms of area planted [2]. It is an important starchy staple crop in Ghana with per capita consumption of 154.0 Kg/ year [2,3]. Besides being a staple food crop, cassava can be processed into fermented and unfermented products. Fermented products include cassava bread, fermented cassava flour, fermented starch, fufu, akyeke, agbelima, whereas unfermented products include tapioca, cassava chips, gari, and pellets. Fermentation is one of the oldest biotechnological approaches to solving food processing and preservation as well as beverages production [4]. Processing the root tuber into various products increases the shelf life and makes transportation to urban markets less expensive [5]. It is also a raw material for industrial starch and ethanol. The leaves of cassava are also edible, while the peel is either eaten by animals or used in the formulation of animal feed.Cassava peel is the epidermal layer of cassava root tuber and as such after harvesting and processing, constitutes 25% of the whole plant. Cassava peels obtained from the products of cassava are usually either discarded as waste in the environment or used as animal feeds if properly processed. The large volume of processed cassava is proportional to the volume of cassava peels wasted without proper use [4]. The history of mining in Africa, in particular, and the third world in general, is a history of land appropriation, displacement of people from their lands, environmental devastation, and further marginalization and oppression of people belonging to the lower economic sectors of the society [6]. In Ghana, the mining sector, especially the mining of gold for export, has received renewed emphasis with new mining codes, clearly defining the rights of the foreign investor [6]. In the Western Region of Ghana alone, it is estimated that over 70% of the land previously used for farming activities is under mine concessions [2] to the detriment of livelihood. As a result, farmlands were lost to mining activities and mine concessions, forcing farmers to cultivate food crops on reclaimed mining land sites for their livelihood.Reclamation is widely used to refer to the revegetation of degraded mining land sites. It aims to recover the productivity of a degraded site mostly using exotic tree species. The success criteria are the restoration of the disturbed site to acceptable physical appearance and acceptable after-use or the end-use objectives [7]. The issue of environmental contamination concerning heavy metals is the focus of research globally, especially in third world countries, as a result of the exploitation of mineral resources. Heavy metals are found in extremely small quantities in animals and plant tissues by consumption of food and water.Naturally, heavy metals may accumulate in the environment through the processes of weathering and dissolution. However, heavy metals may also be introduced to the environment (soil and water) by humans during mining, agricultural, and industrial activities [8]. Heavy metals may enter the food chain as a result of their uptake by the edible parts of the plant, thus, determination of heavy metals in food crops especially those cultivated on reclaimed mining land sites is very important. Heavy metals from soil enter plants primarily through the root system. Plant roots are the most important sites for the uptake of toxic substances from the soil [8,9]. At varying levels, dependent on species, plants can extract or remediate toxins from the soil through their root system [9]. Once the heavy metal is present in the soil and water, these metals are likely to be passed on to humans through the food chain by the process of bioaccumulation.Information on heavy metal uptake and concentration in edible tissues of food crops and their dietary intake are therefore very important for assessing human health risks [10]. Absorption of heavy metals in low doses by humans over a long period through food has been shown to have resulted in serious health consequences such as declining economic development in terms of low productivity as well as the direct cost of treating illnesses. Research has shown that high levels of Pb, Cd, Co, Cu, Ni, and Zn have cumulative effects since there is no homeostatic mechanism that regulates their toxicity. Some common health implications of heavy metals in humans are kidney diseases, damage to the nervous system, diminished intellectual capacity, heart diseases, bone fracture, and cancer [8]. Heavy metals in soil reduce the yield of vegetables by disrupting the metabolic processes of plants. Generally, the levels of heavy metals in soil are higher than the levels observed in food crops. This implies that only a small portion of soil heavy metals is transferred to plants (food crops) and the root acts as a barrier to the translocation of heavy metals within the plant [1]. It is also noticed that the application of phosphate fertilizers to food crops is the main source of soil heavy metal pollution. This is because of the presence of Cd, As, Pb, Cr, Hg, and Ni as impurities in phosphate rocks [1]. The Soil may also become contaminated through mining and related anthropogenic activities [11].The soil-to-plant transfer quotient which is termed bioaccumulation factor (BAF) is the main source of human exposure. The relative difference in bioavailability of metals to plants is the transfer quotient, which is a factor in determining metal tolerance by food crops. The transfer quotient or bioaccumulation factor threshold value of 1.0 is the benchmark above which the plant is said to be tolerant to a specified metal and indicates a high probability of food crop contamination by anthropogenic activities [1]. Information on heavy metal concentrations in edible tissues of food crops and their dietary intake is therefore very important for assessing human health risks [10]. Total Hazard Quotient (THQ) is the adverse health effect that a given metal is likely to produce if present in food and is the determining factor in Health Index (HI). A total hazard quotient value of 1.0 is the threshold value below which the food is safe for human consumption [5]. The sum of THQs is equal to HI.This study was therefore set out to find out the quality of food crop (cassava), a staple food, cultivated on reclaimed mining lands sites, and its associated health risks to residents in the study area.
2. Materials and Methods
2.1. Study Area
- Prestea-Huni Valley District is located between latitude 2° 06′ and longitude 5° 47′ (Figure 1). The District has a land area of approximately 1,809 km2. The land rises from about 240 m to about 300 m above sea level with the topography generally undulating with few scarps ranging between 150 m and 300 m above sea level. The district falls within the forest disserted plateau physiographic region. Precambrian rocks of Birimian underlie the forest disserted plateau. The Birimian rocks are regarded as the most important formations due to their mineral potentials and thus the existence of minerals deposits in the District [12].
 | Figure 1. Map of Prestea-Huni Valley District Showing the sampling points |
2.2. Sampling
- The cassava samples cultivated on reclaimed mining land sites in the study area were harvested in triplicates during three sampling campaign periods from four farms randomly, at each farm, the cassava was harvested at four locations and put together from which a representative sample was obtained and labelled. The corresponding soil samples were taken from the respective farms using the auger, to a depth of 30.0 cm to cover the plough zone. At each farm, the soil samples were taken from three different places, bulked together, from which a representative sample was taken from the bulk and labelled. Both samples were packed into separate polyethylene bags and transported to the laboratory.
2.3. Sample Preparation and Analysis
- The cassava tubers were washed with water to remove any surface deposit of soil particles that might act as contaminants. The washed cassava was then peeled using a stainless knife into an already washed plastic container. Both the cassava tuber and the peel (waste) were collected separately and labelled, put into an oven to dry at 105°C to a constant weight. The dried tuber and the peel were ground separately using cleaned ceramic mortar and pestle into powder and sieved using <40 mesh, homogenized, and packed in polyethylene bags. The samples were stored in the fridge for further analysis. Similarly, the soil samples were disaggregated, dried in an oven at 105°C to a constant weight, and taken through the same treatment as the cassava tuber and the peels.Triplicate 2.0 g of each powdered cassava tuber and the peel were weighed and digested separately using aqua regia (a mixture of HCl and HNO3 in the ratio 1:3) on a water bath at 80°C until a transparent solution appeared depicting the end of the digestion [13]. Similarly, triplicate 2.0 g of the powdered soil samples were also taken and digested in a 100.0 mL polytetrafluoroethylene teflon bombs. About 10.0 mL of concentrated HNO3 was added to each soil sample and allowed to stand for 10 minutes. About 30% H2O2 was also added drop by drop to the mixture until the mixture no longer effervesced on the addition of H2O2. To each mixture in the teflon bomb, 2.0 mL of concentrated H2SO4 were added, followed by the addition of 5.0 mL of concentrated HClO4 successively to each mixture. The resulting mixtures were digested for 25 minutes in a Milestone microwave oven (Ethos 900) using the following operating parameters: 250W for 2.0 min, 0W for 2.0 min, 250W for 6min, 400W for 5.0 min, 650W for 5.0 min and 5.0 min for venting [14]. The rotor was put into a bowl of water to cool the content of the tubes and also to reduce the associated pressure. The digested soil samples were then filtered using whatman (No 1) filter paper into 100.0 mL volumetric flask and made up to the mark using de-ionized distilled water. Calibration standards were prepared for the various analytes together with their respective blanks were used for the determinations, using Varian Fast Sequential Atomic Absorption Spectrophotometer at the respective wavelengths. Acetylene gas was used as the carrier gas in the determination, while argon was passed through the system to remove interfering gases between each reaction time [13,15].
2.4. Quality Control/ Assurance
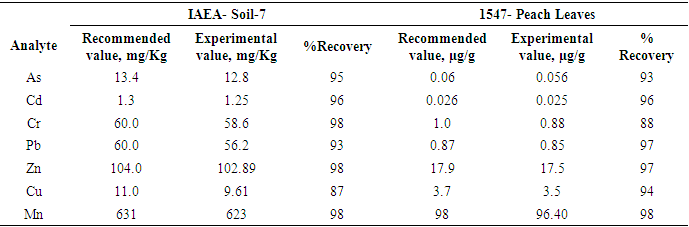
- The chemicals used were of analytical grades obtained from Sigma Aldrich. The reproducibility and precision of the analytical procedures were tested by carrying out triplicate analysis on standard reference materials IAEA-soil-7 and Peach leaves [16,17]. Triplicate results did not differ by more than 5% of the mean. Recoveries from the reference materials ranged between (87-98)%.
2.5. Pollution Load Index (PLI)
- Pollution Load Index is the degree of soil pollution in mined soil compared to a reference value. It was estimated using the pollution load index technique based on soil metal levels as shown in equation 1.
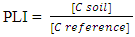 | (1) |
2.6. Bioaccumulation Factor (BAF)
- The heavy metal transfer factor which is termed the Bioaccumulation factor of heavy metals in food crops was estimated using the plant bioaccumulation factor (BAF), which is the ratio of heavy metal concentration in food crop [Cplant] to that of contaminated soil [Csoil] as shown in equation 2 [16].
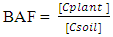 | (2) |
2.7. Estimation of Dietary Exposure and Health Risk
- Estimated Average Daily Intakes (EADIs mg/g/day) of metals of interest were determined based on their average concentrations in cassava samples and a consumption rate of 154 Kg/person/day of cassava was assumed for Ghana [3]. For each type of exposure, the EADI was obtained by the equation 3.
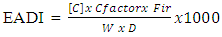 | (3) |
2.8. Reference Oral Dose (Rf Dose)
- Reference oral dose (Rf dose) is an estimated exposure of metals to the human body per day that has no negative effect during a lifetime. The values of the oral dose were obtained from the literature [18].
2.9. Target Hazard Quotient (THQ)
- The THQ is defined as the ratio of exposure (EADI) to toxic metals and the reference dose which is the highest level at which no adverse health effects are expected. The ratio is normally divided by 1000 as a unit conversion factor in equation 4
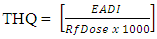 | (4) |
2.10. Health Index (HI)
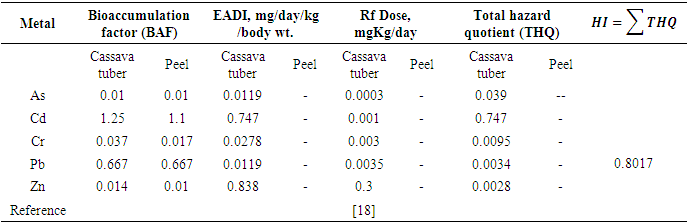
- The health or hazard index (HI) is the summation of the individual target hazard quotients of the metals assessed in cassava. The health index (HI) assumed that the consumption of a particular food type would result in simultaneous exposure to several potentially toxic metals. Even if individual THQs for the metals in the food item is lower than unity, the cumulative effects of consumption may result in adverse health effects. If HI is > 1, there is a potential adverse health effect. However, if the index is below the threshold level of 1.0, the food is considered safe for the consumer [18]. The equation for HI is given by equation 5 [19].
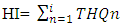 | (5) |
3. Results and Discussion
- The reproducibility of the analytical method was assessed by analysing standard reference materials IAEA soil-7 [16] and 1547-Peach leaves [17]. Table 1 shows the recommended values for As, Cr, Pb, Zn, and Cd in soil-7 and 1547-Peach leaves against the experimental values obtained. The percentage recoveries ranged from 87% to 98%.
|
|
|
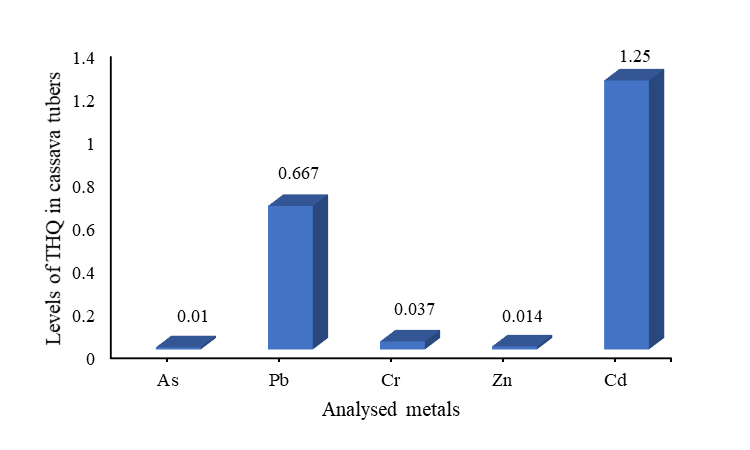 | Figure 2. Relative contributions of analysed metals to health index in cassava tubers |
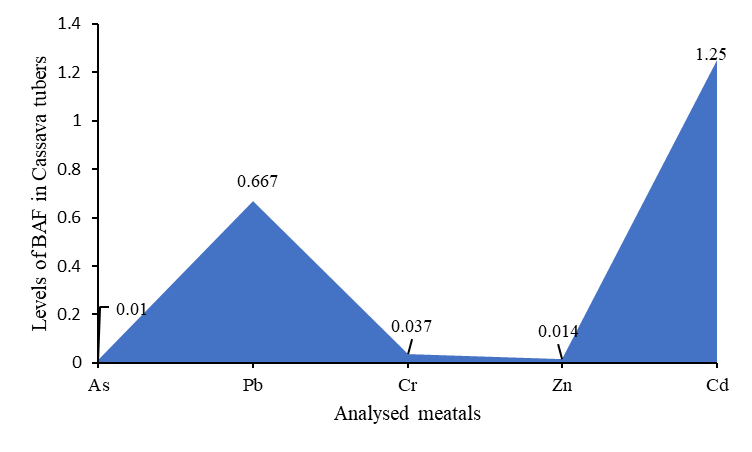 | Figure 3. Relative (bioaccumulation factor) tolerance of analysed metals in cassava tubers |
4. Conclusions
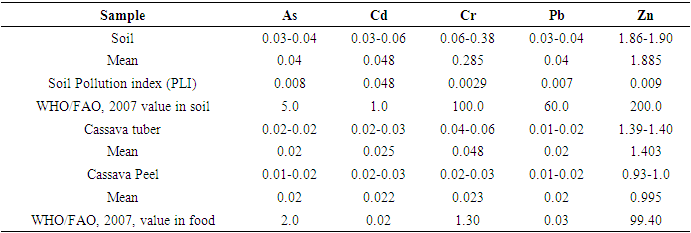
- Evaluation of the soil pollution load index (PLI), suggested that all the metals were below the threshold level of 1.0, an indication of good metal levels for agricultural soil. However, the soil-peel-cassava transfer which is the bioaccumulation factor for Cd in cassava tuber, and its peel were 1.25 and 1.1 respectively, and were all above the threshold of 1.0, indicating that cassava and its peel had a higher tolerance for Cd compared to the other metals. Cadmium also recorded the highest total hazard quotient of 0.747, a factor in determining the human health index, hence contributing positively to the health index.In general, the cumulative Health Index (HI) of all the analysed metals of 0.8017 < 1.0 (a threshold level below which the food is deemed to be safe) which is the sum of all the total hazard quotients (exposure to heavy metals) indicated that there is no potential health effect associated with the consumption of cassava tubers cultivated on reclaimed mining land sites in the Prestea-Huni Valley District.
ACKNOWLEDGMENTS
- The authors are grateful to the University of Cape Coast, for using their facility in analysing the samples and Lord Bobobee a Senior Lecturer at the Department of Chemistry Education for reading the manuscript and making important suggestions.
Conflict of Interest
- The authors have no conflict of interest to declare.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML