Maghdouri N.1, Khaldi A.2, Bouras S.3, Moussa K.1, Benine A.2, Abbouni B.2, Tir Touil A.1, Meddah B.1, Benali M.4
1Laboratoire de Recherche sur le Système Biologique et Géomatique Faculté SNV, Université Mustapha Stambouli Mascara, Algérie
2Laboratoire de Microbiologie Moléculaire, Protéomics et Santéuniversité de Sidi Bel Abbes, Algérie
3Faculté de Pharmacie, Université de Sidi Bel Abbes, Algérie
4Laboratoire de Biotoxicologie Faculté de SNV, Université de Sidi Bel Abbes, Algérie
Correspondence to: Khaldi A., Laboratoire de Microbiologie Moléculaire, Protéomics et Santéuniversité de Sidi Bel Abbes, Algérie.
| Email: |  |
Copyright © 2018 Scientific & Academic Publishing. All Rights Reserved.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
The objective of this study is the evaluation of the contamination of the nourishment of the infants by the residues of antibiotics. This is a qualitative research that is divided into two parts, the first was a descriptive study in the form of a questionnaire for the caregiver and a second part on a qualitative analytical study by a microbiological media diffusion method. Agar (antibiogram) Twenty-two samples were taken into account covering four types of foodstuffs consumed by infants. The prevalence of contamination of the analyzed foods is 32%, this can be explained by the misuse of antibiotics in their production, associated with a lack of application of regulation and rigorous control. This high contamination rate can be corrected by the application of hygiene and control measures as well as by the involvement of health professionals in raising awareness among the population.
Keywords:
Residues, Antibiotics, Feeding, Infant, Susceptibility testing
Cite this paper: Maghdouri N., Khaldi A., Bouras S., Moussa K., Benine A., Abbouni B., Tir Touil A., Meddah B., Benali M., Investigation into the Search for Antibiotic Residues in Infant Feeding in Western Region of Algeria, Food and Public Health, Vol. 8 No. 1, 2018, pp. 8-12. doi: 10.5923/j.fph.20180801.02.
1. Introduction
Intensification of animal production over the last decades has been favored by the use of veterinary drugs, especially anti-infective drugs in modern breeding. These drugs are used either as a curative treatment applied individually or collectively to animals suffering from microbial diseases, or as a preventive treatment to prevent the appearance of certain pathologies or, in some extreme cases, to overcome inadequacies in hygiene in livestock farming. The use of anti-infectives as medicines is recent in contemporary history; it is considered to be one of the major advances in medicine because it has dramatically reduced the morbidity and mortality caused by many infectious diseases of bacterial etiology. However, it changes the ecology of bacteria and contributes to the selection of resistant bacteria. After treatment with animals, these treatments give rise to the presence of residues in the tissues and foods produced by these animals. The presence of antibiotic residues in foods of animal origin related to non-compliance with the conditions of use (dosage and waiting time) or errors in the conduct of the breeding may have serious consequences on the consumer health. As part of the regulation on veterinary pharmacy, a risk assessment approach has been put in place with the evaluation of active ingredients and the setting of maximum residue limits (MRLs). Then, during the evaluation for the marketing authorization of each drug formulation, a waiting time between the last administration of the product and the marketing of the products from the treated animals is defined for the authorized dosage. Finally, when they are used, management measures (good veterinary practices, animal identification, veterinary prescription and waiting time, breeding register) are recommended to reduce this risk.
2. Materials and Methods
Sampling:The sampling operation is based on the results obtained from the survey of caregivers of infants concerning foods (marketed and / or natural foods) most commonly used in infant feeding, while brands tested was supported by our pharmacy internship during the month of November 2016. 22 samples were taken into account covering four types of food consumed by our infants: milk, flours, dairy products and compotes. For ethical reasons we will name them with a capital letter followed by the number.Samples were collected in sterile, sealed and labeled plastic vials. Powdered forms are stored at room temperature are reconstituted according to manufacturers' recommendations for standardized consumption, aqueous or suspended samples are brought to freezing for conservation.Microbiological strainsChoice of reference strains:Two strains were tested during this research, it is Staphylococcus aureus isolated from a nosocomial infection. Staphylococcus aureus is the most pathogenic species of the genus Staphylococcus, it is responsible for food poisoning, localized suppurative infections and, in extreme cases, life-threatening infections.Escherichia coli Gram-negative bacilli isolated from a urinary tract infection, is an intestinal bacterium of mammals, very common in humans. Indeed, it makes up about 80% of our aerobic intestinal flora. Discovered in 1885 by Theodor Escherich, in goats' saddles, it is a faecal coliform generally commensal. However, some strains of E. Coli can be pathogenic, resulting in gastroenteritis, urinary tract infections, meningitis, or sepsis.Both strains were isolated, identified by biochemical tests to confirm their taxonomy whose results are provided by the microbiology laboratory of the central laboratory of Sidi Bel Abbés.A. Antimicrobial activity assayDiffusion methods or standard antibiogram are the most used by clinical laboratories [1]. They consist of depositing impregnated blotting paper disks of determined concentration of antibiotics on the surface of an agar medium, which has previously been inoculated uniformly with a standardized pure culture (105 g / ml) of the test microorganism. As soon as the discs are applied, the antibiotic diffuses from the disc evenly into the agar.Research of antibiotic residues in infant feeding by diffusion method on agar medium:We used the microbiological diffusion method in agar medium by observing the presence or absence of a zone of inhibition of the bacterial growth of the reference strains; Escherichia. Coli and Staphylococcus aureus, previously isolated and identified.The reading is done by observing on a black background the presence or absence of the inhibition zones and accurately measuring the different diameters of the inhibition zones using a rule. We made wells with a diameter of 6 mm. All samples giving inhibition zones of at least 8 mm in diameter were considered positive. That is, a 2 mm inhibition zone was taken into account.
3. Results and Discussion
Microbiological and biochemical identification of isolatesMacroscopic observation of colonies on selective media and a microscopic observation which give the starting point for the identification was confirmed by a biochemical study.Susceptibility pattern of strains to antibioticsIn conclusion, it follows that:The S aureus reference strain (ATCC25923) is sensitive to SPN, PT, AMX, DOX, GMN, AKN.The E coli reference strain (ATCC25922) is sensitive to GMN, CZN, AMP, CTX, IMP, AMX, NAL, CST, SXT, AKN.Search for antibiotic residues by diffusion method:32% of the foods tested were found to be positive, that is to say that they have zones of inhibition of bacterial growth with E coli and / or S aureus with a diameter that varies between 09mm and 35mm, which can be due to the antibacterial activity of ATB residues probably contained in these foods.This considerable percentage is worrying because of the importance of these foods in the feeding of infants, which can cause the inefficiency of antibiotic therapy and the increase in the number of resistant strains, beside the self-medication and non-targeted therapy.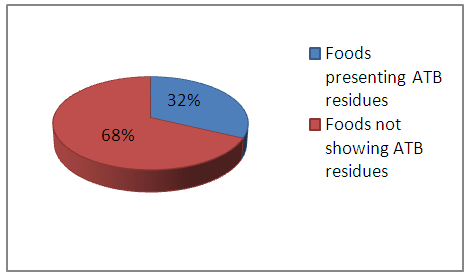 | Figure 1. Frequency of Food Contamination Analyzed by Antibiotic Residues (%) |
Milk and milk products rank first and then compotes and flours with regard to the presence of ATB residues, this can be explained by their animal origin unlike flours and compotes which are of vegetable origin. 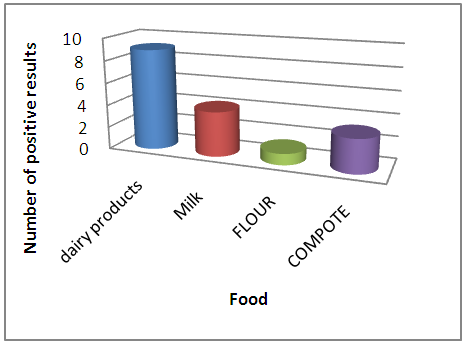 | Figure 2. Prevalence of the presence of antibiotic residues in each type of food |
Behavior of the reference strains with the tested foods:The milk samples (L1, L4, L6) develop zones of inhibition vis-à-vis E coli range from 09mm to 13mm, which means that they probably contain residues of ATB, with total absence of zones of In no case does it confirm the absence of ATB residues whose reference strain can be resistant. Regarding the L8 sample which is breast milk and despite which is taken from a woman under antibiotic therapy (Amoxicillin) the result was revealed negative this can confirm either the absence of this antibiotic in its milk or its presence in low undetectable quantity and this is quoted in the bibliographic part.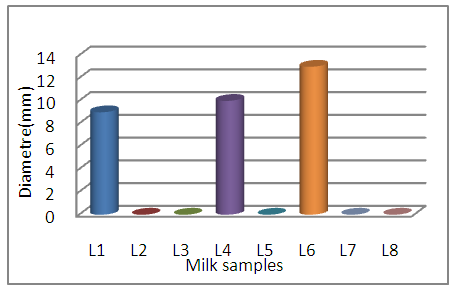 | Figure 3. Behavior of E. coli with respect to milk samples |
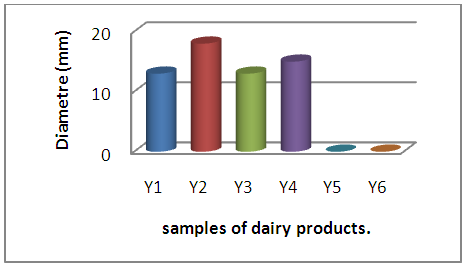 | Figure 4. Behavior of E. coli with respect to samples of dairy products |
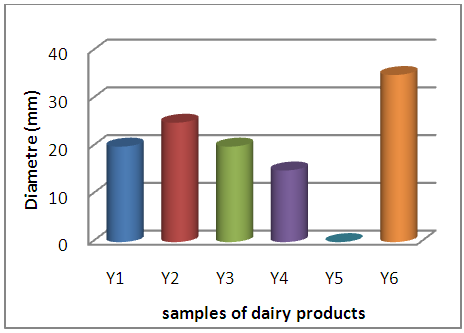 | Figure 5. Behavior of S.aureus with respect to dairy samples |
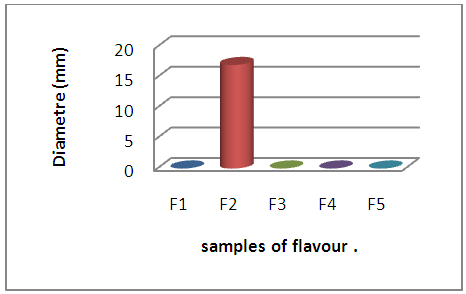 | Figure 6. Behavior of E. coli with flour samples |
A large proportion of the samples of dairy products have an inhibitory action on the two reference strains tested, whose diameters vary from 13mm to 15mm for E. coli and from 15mm to 35mm for S aureus, this action results from the probable presence of residues. of ATB to which the strains are sensitive.A single sample (F2) has developed a 17mm diameter inhibition zone with E coli with complete absence of inhibitory action of the other samples whether with E coli and / or S aureus.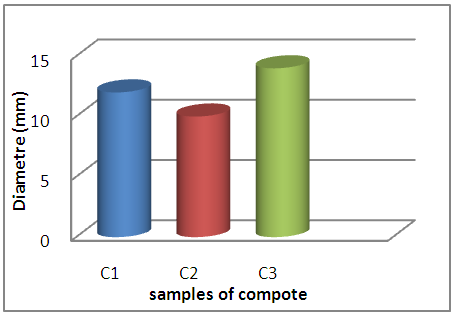 | Figure 7. Behavior of E. coli with respect to flour samples |
The compote samples show the presence of inhibition halos vis-à-vis E coli ranging from 10mm to 14mm, which means that they probably contain ATB residues, with bacterial growth on the whole of the whole of the agar surface of S aureus.Our present work has focused on the research of antibiotic residues in the diet of infants which is essentially milky whose main source is cow's milk which may contain residues of antibiotics resulting from their improper and irregular use which has not been without consequence because their presence is marked in the food chain, the co of this situation is the appearance of a large number of resistant bacterial strains on the one hand and treatment failures in the infant as a consumer on the other hand [2] Qualitative research on nutrient agar to highlight the presence or absence in: milk, infant formula, milk powders, dairy products, flours and compotes of the inhibitory substances (antibiotics) of microbial germs ie S. aureus and E. coli revealed the formation of the inhibitory zones around certain wells containing the analyzed samples, testifies the presence of ATB residues, since these have created an inhibitory action on S. aureus and E. coli; While the proliferation of S aureus and E coli has spread in the agar of well remnants, which proves the absence of the inhibitory action that is to say the absence of antibiotic residue.As a result, we can confirm the presence of ATB residues in our samples. The results show that milk powder, infant formulas and dairy products whosce raw material is cow's milk, and which are used as staple foods in infants' diets, were developed from milk containing residues of antibiotic substances harmful to the health of infants, contrary to the regulations in force and this is confirmed by the positive results marked by Ben-Mahdi in 2009 with a percentage of 7.87% of the contamination of cow's milk samples by antibiotic residues produced in Algiers and the results of AGGAD et al (2009) in western Algeria This can be explained by the fact that milk was obtained from animals treated with ATB-based drugs in the barns, with non-compliance with the waiting period (ABIDI 2004, BROUILLET 1994; GEDILAGHINE, 2005), or due to errors made by breeders according to ABIDI (2004), such as: an erroneous milking of a dried cow, recently treated with ATB, an accidental mixing of the milk of a cow treated with that other cows, or contamination by milking equipment due to lack of cleaning after milking treated cows, lack of identification of animals, poor hygiene during milking such as contamination of milk with faecal soils containing ATB excreted digestive (LABIE, 1981), the voluntary addition of ATB in milk in order to inhibit the development of microflora and improve the bacteriological quality of the product.Despite the thermal and enzymatic treatments that milk undergoes during its transformation into: milk powder, infantile preparations and various dairy products, the presence of ATB residues in these finished products is signaled.In the dairy industry, particularly in the manufacture of yoghurt, they use lactic ferments that must remain alive to ferment the milk and obtain yoghurt. Lactic acid bacteria such as: Streptococcus thermophilus, Lactobacillus bulgaricus, Lactococcus lactis ... (Mietton et al., 1994; Bories 1993, Mourot 1981) play an essential role as ferment in acidifying milk, these enzymes have the property of digesting lactose and producing lactic acid (Robb 2006, Mietton et al. Lactics are sensitive to very low doses of antibiotics (Brouillet 2002, Fabre and Joyes 2000), the presence of ATB residues in the milk will prevent its fermentation and cause great financial losses to the dairy industries, but their presence in the finished product (yoghurt) means that the lactic ferment is not susceptible to antibiotics in the same way that is why we have marked their presence in our samples of dairy products without building an obstacle to their production this is compared with the results of KHALDI et al (2012) who found that 42.5% of yogurts made in the region of orania are contaminated by antibiotic residues this can to be explained either by the contamination of the trenches (poor hygiene) or by the contamination of the raw material (cow's milk). Food contamination may manifest during manufacturing processes that is the most reasonable explanation for the detection of substances with antibacterial activity in compotes and flours, whose origin is vegetable, when the use of ATB in the field of agriculture (excluding breeding) remains limited as mentioned in the second chapter (antibiotics).Or may result in illegal acts such as the voluntary addition of ATBs by industries during manufacturing to improve bacteriological properties and extend the shelf life of products in the absence of strict control by the state.Regarding breastfeeding, health professionals who prescribe or dispense drugs are not well informed about prescribing for breastfeeding women. Mothers are afraid to poison their baby. In many cases, health professionals recommend that women wean their infants or stop breastfeeding. However, this measure is rarely necessary, as the risks associated with stopping breastfeeding are in the majority of cases greater than the risks associated with taking the drug by the mother, because this judgment uses artificial breastfeeding. presents a greater danger, Thus, despite the almost constant passage of the drugs consumed by the mother in the milk, the breastfed infant will receive a maximum of 1% of the total maternal dose. This is consistent with the negative result obtained in our work.Comparing our results with others, we see a diversification of data: For cow's milk that was revealed negative in our study, other studies more specify and which devotes the bulk to the search for ATB residues in cow's milk using the Delvot test as a detection method carried out here in Algeria as AGGAD et al (2009) where 28% of milk samples are positive and he is close to other authors such as: GUETARNI (2006) with 26.38% and Marroc SRAIRI et al (2004) with 26%.In other African countries the percentages of contamination are average as in Kenya. SHITANDI states that 14.90% are contaminated, and ABEBEW SYIT (2011) reports that 8.5% of samples are contaminated in Ethiopia.In the countries of the European Union, the percentages of the positive results are very low, they are lower than 0.5% in the countries of the West (HILLERTON and BERRY, 2004) where in Belgium and Denmark they are of 0.1% (FSA, 2006) in Spain they are 0.18% (GARCIA et al, 2001) and in Sweden the percentages are between 0.08% and 0.26% (STRENESJO, 1998).The work done on milk powder, infant formula, dairy products as finished product, as well as flours and compotes are limited by contribution to those of cow's milk, our results show positive for most of our samples, such as a study carried out on milk powder, carried out in Mexico by TOLENTINO et al in 2005, the latter carried out a study on four brands (A, B, C, D) of milks and the percentages of contamination were respectively: 47.20%, 58.30%, 44.70% and 50%.
4. Conclusions
From the results obtained after analysis of antibiotic residues by the microbiological diffusion method on agar medium, we can conclude that the degree of contamination of the samples remains quite high. However, it is much higher in infant formulas and dairy products (yoghurt) in addition to flour and compotes.However, microbiological detection methods do not give an indication of the identity of the inhibitory substance. The evaluation of the potential health or technological risks associated with their presence requires a prior qualitative and quantitative knowledge of them. Reason why, the work must be pursued by a semi quantitative research by ELISA method and in the end a quantification by HPLC.
References
| [1] | Schwarz. S, Kehrenberg. C. (2001). Use of antimicrobial agents in veterinary medicine and food animal production. International Journal of Antimicrobial Agents: p431-437. |
| [2] | Regeringskanliet. (1997). Pouvons-nous utiliser moins d’antibiotiques? Une brochure sur les antibiotiques dans l'alimentation animale et leurs effets sur l'homme et l'animal. Ministère de l'Agriculture, de l'Alimentation et de la Pêche de Suède, Stockholm, p 31. |
| [3] | Jeon. M, kim. J, Paeng. K.j, Park. S.W, Paeng. I.R. (2008). Biotin-avidin mediated competitive enzyme-linked immunosorbent assay to detect residues of tetracyclines in milk. Microchemical Journal. 2008. p 26-31. |
| [4] | Fiscus-Mougel F. (1993). Les résidus d’antibiotiques à usage vétérinaire dans le lait et la viande. Thèse de Doctorat en Pharmacie. Lyon: Université Claude Bernard. p 84. |
| [5] | Afssa. (2006). Agence Française de Sécurité Sanitaire des Aliments, Rapport: usages vétérinaires des antibiotiques, résistance bactérienne et conséquences pour la santé humaine. Janvier 2006. 214 p. |
| [6] | Cerniglia C.E. & Kotarski S. (2005). Approaches in the safety evaluations of veterinary antimicrobial agents in food to determine the effects on the human intestinal microflora. Journal of veterinary Pharmacology and Therapeutics. [en ligne]. Vol 28. p3-20. Disponible sur: http://onlinelibrary.wiley.com/doi/10.1111/j.1365-2885.2004.00595.x/abstract (consulté le 14/01/2017). |
| [7] | Commission nationale AnGR. (2003). Rapport national sur les ressources génétiques: Algérie. Ministère de l’agriculture et développement rural. Octobre 2003. 46p. |
| [8] | Lazak. M, Mâamar. G (1978). Contrôle de la qualité du lait commercialisé par les crémiers de Constantine: étude physico-chimique et pollution chimique. Mémoire d’ingénieur d’état en industrie alimentaire, université de Constantine, p 2-3. |
| [9] | Pougheon. S, (2001). Contribution a l’étude des variations de la composition du lait et ses conséquences en technologie laitière. Thèse Pour l’obtention du grade de docteur en médecine vétérinaire, Ecole Nationale Vétérinaire de Toulouse, p 14-15. |
| [10] | Courtet Leymarios. F (2010). Qualité nutritionnelle du lait de vache et de ses acides gras. Voies d’amélioration par l’alimentation. Thèse pour l’obtention du doctorat en médecine vétérinaire, faculté de médecine de Créteil, p 18-37. |
| [11] | Pujol-Dupuy. C (2004). Accidents alimentaires d’origine bactérienne lies à la consommation de laits et produits laitiers. Thèse doctorat (médecine-pharmacie) université Claude-Bernard - Lyon I. p 20-25. |
| [12] | Wehrmuller. K; Ryffel. S (2007). Produits au lait de chèvre et alimentation, 2007, ALP actuel, N° 28. Réussir Lait Élevage Avril 2007. p 2-3. |
| [13] | FAO (1998). Le lait et les produits laitiers dans la nutrition humaine, Collection FAO [en ligne]: alimentation et nutrition n° 28, ISBN 92-5-20534-6, disponible sur: http://www.fao.org/docrep/t4280f/T4280F00.htm, (consulté le 20/12/2016). |
| [14] | France AgiMer. (2011). établissement national des produits de l’agriculture et de mer. La consommation mondiale de viande: état des lieux, dynamique, défis et perspectives / édition février 2011. Numéro 5. 7p. |
| [15] | Amellal. R (1995). La filière lait en Algérie : entre objectif de la sécurité alimentaire et la réalité de la dépendance, Revue : Options Méditerranéennes, Série B, n°14, p 229-230. |
| [16] | Benelkadi. K (2007). Industrie du lait en Algérie, Journal el Watan, édition du 7 mai 2007. |
| [17] | Griffoul. B (2007). Une production laitière fortement dépendante des importations. Réussir Lait Élevage Avril 2007. p 2-3. |
| [18] | Mamart. M (2007). Filière lait en Algérie: Pour quand l’autosuffisance ? Journal el Watan. Édition du 7 mai 2007. |
| [19] | Ghazi. K et Niar1. A (2011). Qualité hygiénique du lait cru de vache dans les différents élevages de la Wilaya de Tiaret (Algérie), TROPICULTURA, 29/04/2011. p 193-196. |
| [20] | OIE, (2010). Code sanitaire pour les animaux terrestres. Volume 1- Dix-neuvième édition. ISBN 978-92-9044-772-6. © Copyright OMSA. |
| [21] | Coibon. L, (2008). Acquisition des qualités organoleptiques de la viande bovine: adaptation à la demande du consommateur. Thèse de doctorat vétérinaire, Université de Toulouse. Thèse: 03 – Tou 3 – 4018. |
| [22] | Rosset. R, Roussel. N, Ciquard, (1984). Composition chimique du muscle. Les viandes- Informations Techniques des Services Vétérinaires, P 97-102. |
| [23] | Belhadj. M-T (2008). Contribution à l’étude de la qualité bactériologique des viandes blanches commercialisées dans la Wilaya de Bordj Bou Areridj, page 7. Mémoire de magistère, école nationale vétérinaire El Harrach, Alger. |









 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML