-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Food and Public Health
p-ISSN: 2162-9412 e-ISSN: 2162-8440
2016; 6(2): 33-37
doi:10.5923/j.fph.20160602.01

Determination of Additives in Cigarettes Utilizing Evolved Gas Analysis (EGA)
Karissa Ferguson, Michael J. Samide, Cierra Schmicker, Tyler Schenk, Anne M. Wilson
Clowes Department of Chemistry, Butler University, Indianapolis, USA
Correspondence to: Anne M. Wilson, Clowes Department of Chemistry, Butler University, Indianapolis, USA.
| Email: |  |
Copyright © 2016 Scientific & Academic Publishing. All Rights Reserved.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

While additives to consumer products must be listed in order by amount, it is not required to report the actual amounts of these additives. Given that impacts on human health are dose dependent, knowledge of additive amounts would aid discussions of the safety of these compounds for inhalation. Additives for cigarettes include flavors, such as menthol, and propylene glycol, a humectant. Rapid determination of the amounts of menthol and propylene glycol additives in cigarettes was accomplished utilizing evolved gas analysis (EGA) – gas chromatography (GC) – flame ionization detection (FID). Careful preparation of samples and appropriate sample storage are critical components to this study. Menthol amounts in cigarettes were consistent with previous studies and this represents the first report of propylene glycol amounts.
Keywords: Additives, Menthol, Propylene glycol, Evolved gas analysis
Cite this paper: Karissa Ferguson, Michael J. Samide, Cierra Schmicker, Tyler Schenk, Anne M. Wilson, Determination of Additives in Cigarettes Utilizing Evolved Gas Analysis (EGA), Food and Public Health, Vol. 6 No. 2, 2016, pp. 33-37. doi: 10.5923/j.fph.20160602.01.
Article Outline
1. Introduction
- With the publication of Celebuki’s study in 2005 [1], it seemed that the amount of menthol in mentholated cigarettes was confirmed. These experimentally determined amounts were consistent with industry documents [2]. Interest in mentholated cigarettes has continued amid questions such as whether or not mentholated cigarettes are more addicting, promoted to minority groups and children, and that the menthol itself may provide an additional health risk [3-10]. In 2015, another study reported slightly higher amounts of menthol in mentholated cigarettes, and that menthol was found in measurable amounts in non-mentholated cigarettes [11]. An additional sampling technique has been reported that showed large amounts of menthol in Southeastern Asian cigarettes, even though they were not marketed as containing menthol [12]. Understanding menthol levels in cigarettes remain an important factor related to public health.Propylene glycol is added to tobacco as a humectant [2, 11]. Propylene glycol is also used as a major ingredient in electronic cigarettes [20], as a solvent for flavoring compounds in food products [21], and as a solvent for pharmaceuticals which may lower the toxicity threshold of the drug [22]. The FDA has classified propylene glycol as a food additive that is “generally recognized as safe” [23]. However, the European Union has set limits on the amount of propylene glycol allowed in food products [24]. For inhaled propylene glycol, there are conflicting studies that show low toxicology of propylene glycol in cigarettes [25] and toxicology concerns about electronic cigarettes [26]. Given the controversy around propylene glycol as an additive to ingested and inhaled products, determination of the amount of propylene glycol in common products would be useful.Given the continued interest in menthol levels in cigarettes and a growing interest in propylene glycol amounts, a rapid method for quantitatively determining amounts of volatile additives would be useful. Analysis of flavor and odor compounds in products is well documented. Methods of extraction of volatile organic compounds (VOCs) are well known and have been described at length in the literature [13-15]. These methods can be broadly categorized as (a) solvent extraction and/or distillation [13-15, 18], (b) headspace sampling [16-18], (c) thermal desorption and trapping [16-18], (d) solid-phase microextraction [16-18] and (e) evolved gas analysis [19]. The methods above, with the exception of (e), all utilize organic solvents [1, 11, 12-18]. Methods (a) through (d) can be time consuming and require large sample size (grams). Method (e) has been shown as a rapid method for accelerated emission and subsequent quantitative analysis of VOCs in milligram quantities of solid samples without any need for extraction [19].The current work describes the quantitative analysis of menthol and propylene glycol additives emitted from 8-13 mg of cigarette material using evolved-gas analysis, gas chromatography, flame ionization detection (EGA-GC-FID).
2. Methods
2.1. Samples
- Cigarette samples were obtained from a local convenience store and include Newport 100s, Kool 100s, and Marlboro Menthol 100s, Marlboro 100s (red), and Marlboro Special Blend (gold). All packages were stored at 4°C inside two nested zip top bags. Prior to analysis, samples were allowed to warm to room temperature, 22°C.Menthol, propylene glycol, and nicotine were purchased from Aldrich Chemical Company and used without further purification. Menthol and propylene glycol were utilized as quantitation standards in acetone, and nicotine was utilized as a comparative standard. Acetone (HPLC grade) was purchased from Aldrich Chemical Company.
2.2. Experimental Equipment
- Analysis of VOCs from solid samples was performed using an EGA oven similar to that reported in the literature [19, 27]. Herein, an oven for evolved-gas analysis was built in house using Swagelok fittings and steel and PEEK tubing. Sample introduction occurs through the top opening of a Swagelok tee. The sample (typically 5 to 20 mg) is contained in a cup (Frontier Laboratories, PY1-EC80F) and suspended from a 150 mm long wire hook formed from a 0.020” steel wire. The seal on the top fitting of the tee is made using a 9 mm green rubber septum and a ¼” Swagelok nut. Helium carrier gas from the gas chromatograph is introduced to the oven in the side port of the Swagelok tee, using a reducing union and connector tubing. The body of the EGA oven is constructed using a ¼” O.D. and 5/32” I.D. steel tube and is attached to the tee using an appropriate reducing union. A second reducing union is used to connect the heating tube to the injection needle (22 gauge, Hamilton 7780-04). The oven is operated at 180°C (± 2°C) and heating is accomplished using a heating mantle jacket connected to a Variac. The sample was introduced into the oven for 60 or 120 seconds and VOCs emitted at the elevated temperature were concentrated on the front end of the cool (40°C) column. This introduced the volatile organic compounds present in the sample to the GC column as a tight band. As the volatile compounds of interest have reasonably high boiling points (menthol: 212°C, propylene glycol: 188°C, nicotine: 247°C), these are effectively trapped onto our column held at 40°C without the need of cryotrapping.All chromatograms were obtained using an EC-Wax column (Alltech, 30 m 0.25 mm 0.25 µm) installed in an Agilent 6890 GC equipped with an FID operating with a constant He carrier gas flow rate of 1.1 mL min-1. The oven temperature program was 40°C for 2 min; an 8°C min-1 ramp to 150°C; a 15°C min-1 ramp to 200°C with an isothermal hold for 15 min.
2.3. Procedures
- Prior to analysis, tobacco leaves were pulled from a cigarette, cooled under liquid nitrogen, and ground to a fine powder with a mortar and pestle, and stored as a powder in a sealed container. In triplicate, the ground tobacco samples (6-13 mg) were added to the sample cup and were heated at 180°C for 60 or 120 seconds. Analysis of the VOCs by GC/FID followed. Strands of the filter material from one of the cigarettes (Kool 100s) were placed directly into the sample cup and analyzed in a similar fashion.
2.3.1. Calibration Procedure
- A standard stock solution was prepared by dilution of a known mass of menthol or propylene glycol with acetone to a volume of 100 mL. This stock solution was further diluted to prepare several calibration standards.
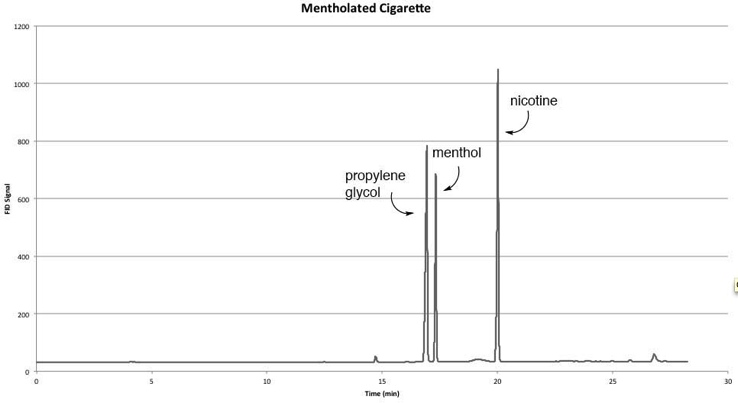 | Figure 1. Sample Chromatogram of a Mentholated Cigarette |
2.3.2. Multi-Injection Procedure
- For multi-injection studies, the same sample, prepared as above, was inserted and removed as described. After the initial injection had eluted from the column, second, through fourth injections were performed on the same sample. Individual chromatograms were recorded for each injection. Both 60 second and 120 second injections were performed on mentholated cigarette samples.
2.3.3. Method Validation
- An acetone solution with a known amount of menthol was added to unmentholated cigarette tobacco (Marlboro 100s) and shaken in a sealed container for 10 minutes. This tobacco was then treated as above (leaves were sampled, cooled under liquid nitrogen, and ground to a fine powder). A spike recovery of 81% was obtained for menthol.
3. Results
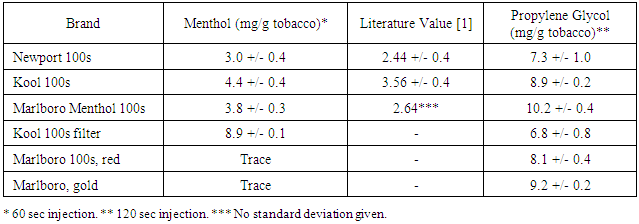
- Figure 1 depicts a chromatogram showing the separation of three major VOCs emitted from mentholated tobacco leaves: propylene glycol (16.8 min), menthol (17.2 min), and nicotine (19.9 min). For the three brands of mentholated cigarette studied, the amounts of menthol ranged from 3.0 to 4.5 mg menthol per g tobacco (Table 1). This amount is significantly higher than previously reported [1, 2]. Furthermore, the amount of menthol in the filter was nearly double that found in the tobacco. This is consistent with the practice that menthol is often added to the filter material as opposed to the tobacco leaves [11]. As controls, two additional cigarette sub-brands (Marboro 100s red and gold) showed trace amounts of menthol.
|
 | Figure 2. Multi-Injection of Mentholated Cigarette, Menthol (60 seconds) |
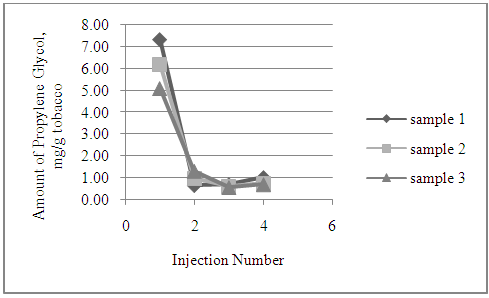 | Figure 3. Multi-Injection of Mentholated Cigarette, Propylene Glycol (120 seconds) |
4. Discussion
- By our method, menthol amounts in our sample cigarettes were above reported amounts (see Table 1). We found that menthol amounts in our sample significantly decreased over time if not kept in a sealed container. This suggests that careful sample preparation and storage are vital to quantitation of flavoring agents in cigarettes and that a rapid method of analysis is optimal.Menthol amounts were considerably higher in the filter. It is common practice for menthol to be introduced to mentholated cigarettes via the filter [11]. Previous literature reports utilized the entire cigarette (filter, paper, and tobacco) for their testing. Given that our tobacco samples were also higher than the literature values, it is likely that previous researchers lost some menthol in sample preparation. It is our recommendation that steps be taken to reduce menthol loss (sealed containers, care not to heat the sample, etc.). It is also possible that the amount of menthol added to mentholated cigarettes has increased since 2005.To our knowledge, this is the first report of quantitation of propylene glycol in cigarettes. As propylene glycol is found in a wide variety of food products, the EGA-GC-FID method represents a way to obtain quantitative information about propylene glycol using very small amounts of sample without needing to perform an extraction. As the European Union has placed limits on the amount of propylene glycol in food items, this may prove to be a useful tool in this arena.The increased usage of propylene glycol by the electronic cigarette industry [12, 20, 26] highlights the importance of a rapid method for the determination the amount of propylene glycol in small samples. Quantitation is especially important given the conflicting information about toxicity of inhaled propylene glycol [25, 26, 28]. While nicotine was observed in our tobacco samples, quantitation has not yet been realized due to what we believe is a diffusion issue. After an initial release, the remaining naturally occurring nicotine diffuses through cell walls slower than the injection time for the EGA method. The fourth injection of a four-injection study showed significant amounts of nicotine still present in the cigarette material. Sample preparation and fine grinding (in a cryomill) may improve quantitation of nicotine by reducing particle size so that diffusion is no longer a limiting factor.Utilizing our EGA method, we were able to perform a rapid analysis of additive VOCs in cigarettes with small amount of sample, 10 mg or less. With this method, we obtain quantitative data for menthol and propylene glycol. The data collected indicate that significantly more menthol is present in our samples than had been previously reported in the literature. This may be due to our very rapid method, which may lead to less VOC loss. Furthermore, we have provided the first quantitation of propylene glycol in tobacco, with an average of about 9 mg of propylene glycol per gram of tobacco.
5. Conclusions
- Utilizing our EGA method, we were able to perform a rapid analysis of additives in cigarettes with small amount of sample, 6-13 mg, without the need for extraction with organic solvents. With this method, we obtain quantitative data for menthol and propylene glycol. Knowledge of the amount of menthol and propylene glycol in ingested and inhaled items will allow for recommendations to be made based on the amount of exposure, not just the presence or absence of these compounds from common items.
ACKNOWLEDGEMENTS
- The authors would like to thank Dean Jay Howard of the College of Liberal Arts and Sciences and the Holcomb Undergraduate Research Fund at Butler University for support of undergraduate research.
References
| [1] | C. C. Celebuki, G. Ferris Wayne, G. N. Connolly, J. F. Pankow, E. I Chang. 2005. Characterization of measured menthol in 48 U. S. cigarette sub-brands. Nic. Tob Res. 7(4), 523-531. doi:10.1080/14622200500186270. |
| [2] | G. Ferris Wayne, G. N. Connolly. 2004. Application, function, and effects of menthol in cigarettes: A survey of tobacco industry documents. Nic. Tob. Res. 6 (Supp 1), S42-S54. doi:10.1080/14622203310001649513. |
| [3] | Preliminary scientific evaluation of the possible public health effects of menthol versus nonmenthol cigarettes. FDA report, 2012. http://www.fda.gov/downloads/UCM361598.pdf. Accessed January 20, 2016. |
| [4] | Tobacco Products Scientific Advisory Committee. FDA report, 2011. UCM269697.pdf. Accessed January 20, 2016. |
| [5] | Public health effects on menthol cigarettes. American Legacy Foundation, 2015. http://www.legacyforhealth.org/. Accessed August 20, 2015. |
| [6] | Y. O. Lee, S. A. Glantz. 2011. Menthol: putting the pieces together. Tob. Control 20, ii1-ii7. doi:10.1136/tc.2011.043604. |
| [7] | K. Klausner. 2011. Menthol cigarettes and smoking initiation: a tobacco industry perspective. Tob. Control, 20 (supp 2): ii12-ii19. doi:10.1136/tc.2010.041954. |
| [8] | J. D. Heck. 2010. A review and assessment of menthol employed as a cigarette flavor ingredient. Food Chem. Tox., 48, S1-S38. doi:10.1016/j.fct.2009.11.002. |
| [9] | G. A. Giovino, A. C. Villanti, P. D. Mowery, et. al. 2013. Differential trends in cigarette smoking in the USA: is menthol slowing progress? Tob. Control, 24, 28-37. doi:10.1136/tobaccocontrol-2013-051159. |
| [10] | Altria Client Services on behalf of Phillip Morris USA background information to: Tobacco Products Scientific Advisory Committee menthol discussion, July 15-16, 2010. UCM246043.pdf. Accessed January 20, 2016. |
| [11] | J. A. Ai, K. M. Taylo, J. G. Lisko, H. Tran, C. H. Watson, M. R. Holman. 2015. Nic. Tob. Res. doi:10.1093/ntr/ntv162. |
| [12] | J. G. Lisko, S. B. Stanfill, C. H. Watson. 2014. Quantitation of ten flavor compounds in unburned tobacco products. Anal. Methods, 6, 4698-4704. doi:10.1039/C4AY00271G. |
| [13] | E. S. Ong. 2004. Extraction methods and chemical standardization of botanicals and herbal preparations. J. Chrom. B, 812, 23-33. doi:10.1016/j.jchromb.2004.07.041. |
| [14] | X. Wang, V. Kapoor, G. A. Smythe. 2003 Extraction and chromatography-mass spectrometric analysis of the active principles from selected Chinese herbs and other medicinal plants. Am. J. Chin. Med., 31, 927 – 944. doi:10.1142/S0192415X0300165X. |
| [15] | K. Vilkhu, R. Mawson, L. Simons, D. Bates. 2008. Applications and opportunities for ultrasound assisted extraction in the food industry – A review. Innov. Food Sci. Emerg. Technol., 9, 161-169.doi:10.1016/j.ifset.2007.04.014. |
| [16] | M. L. Ruiz del Castillo, G. Santa-María, M. Herraiz, G. Blanch. 2003. A comparative study of the ability of different techniques to extract menthol from Mentha piperita. J. Chromatogr. Sci., 41, 385 – 389.doi:10.1093/chromsci/41.7.385. |
| [17] | F. Augusto, A. Leite e Lopes, C. Alcaraz Zini. 2003. Sampling and sample trends for analysis of aromas and fragrances. Trends Anal. Chem.. 22(3), 160-169. doi:10.1016/S0165-9936(03)00304-2. |
| [18] | J. G. Wilkes, E. D. Conte, Y. Kim, M. Holcomb, J. B. Sutherland, D. W. Miller. 2000. Sample preparation for the analysis of flavors and off-flavors in foods. J. Chrom. A, 880, 3-33. doi:10.1016/S0021-9673(00)00318-6. |
| [19] | M. J. Samide, G. D. Smith. 2015. Analysis and quantitation of volatile organic compounds emitted from plastics used in museum construction by evolved gas analysis–gas chromatography–mass spectrometry. J. Chrom. A., 1426, 201-208.doi:http://dx.doi.org/10.1016/j.chroma.2015.11.066. |
| [20] | D. F. Maron. 2014. Smoke Screen: Are E-Cigarettes Safe? Scientific American, 310, 31-32. http://www.scientificamerican.com/article/smoke-screen-are-e-cigarettes-safe/, accessed 2-22-2016. |
| [21] | I. F. Gaunt, F. M. B. Carpanini, P. Grasso, A. B. G. Lansdown. 1972. Long-term toxicity of propylene glycol in rats. Food and Cosmetics Toxicity, 10 (2), 151-162. |
| [22] | “Propylene Glycol” in Handbook of Pharmaceutical Excipients 5th edition (2006). R. C. Rowe, P. J. Sheskey, S. C. Owen, Eds.. Pharmaceutical Press; 521-523. |
| [23] | Propylene Glycol. “Agency for Toxic Substances Portal”, Agency for Toxic Substances and Disease Registry. http://www.atsdr.cdc.gov/substances/toxsubstance.asp?toxid=240 accessed 2/20/16. |
| [24] | “Directive 2003/114/EC of the European Parliament and of the Council Amending Directive 95/2/EC on Food Additives other than Colours and Sweeteners.” http://www.eumonitor.eu. Accessed 2-22-20016. |
| [25] | C. L. Gaworski, M. J. Oldham, C. R. E. Coggins. 2010. Toxicological considerations on the use of propylene glycol as a humectant in cigarettes. Toxicology, 269, 54-66. |
| [26] | R. Grana, N. Benowitz, S. A. Glantz. 2014. E-Cigarettes; A Scientific Review. Circulation. 129, 1972-1986. |
| [27] | M. J. Samide, A. M. Wilson, D. Shinholt, J. Smith. 2016. Development of a Low-Cost Evolved-Gas Analysis (EGA) Device for the Rapid Detection of Volatile Organic Compounds (VOCs) from Common Household Items. J. Lab. Chem. Educ., 4(1), 19-24 (doi:10.5923/j.jlce.20160401.04). |
| [28] | Propylene Glycol, Safety Data Sheet. http://www.sigmaaldrich.com |
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML