-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Food and Public Health
p-ISSN: 2162-9412 e-ISSN: 2162-8440
2015; 5(3): 92-101
doi:10.5923/j.fph.20150503.05
Supercritical Water Gasification of Biomass for Hydrogen Production: Variable of the Process
Lachos-Perez D.1, Juliana M. Prado2, Torres-Mayanga P.1, Tânia Forster-Carneiro1, M. Angela A. Meireles1
1Department of Food Engineering, School of Food Eng., University of Campinas (UNICAMP), Rua Monteiro Lobato, Campinas, SP, Brazil
2CCN (Center for Natural Sciences) / Lagoa do Sino Campus – UFSCar (Federal University of São Carlos), Rod. Lauri Simões de Barros, Buri, SP, Brazil
Correspondence to: Tânia Forster-Carneiro, Department of Food Engineering, School of Food Eng., University of Campinas (UNICAMP), Rua Monteiro Lobato, Campinas, SP, Brazil.
| Email: |  |
Copyright © 2015 Scientific & Academic Publishing. All Rights Reserved.
Waste biomass has various origins, such as agricultural crops, food waste, animal waste, municipal solid waste, and has the potential to be converted into energy and applied in biorefineries, thus contributing with lignocellulosic material. The emerging technology Supercritical Water Gasification has a great potential for recycling biomass for the production of synthesis gas with a higher percentage of hydrogen. The supercritical water gasification (SCWG) does not require drying; thus, the problem of drying is largely avoided by the SCWG and can be used for biomass with high percentage of humidity. The conversion efficiency of the SCWG is generally higher when compared with conventional technologies. This paper reviews known and emerging key supercritical water properties that influence the SCWG of biomass (viscosity, density, dielectric constant and ionic product), the advantages of the SCWG with respect to conventional gasification, the economic viability of the process, and the kinetics of the biomass in the process, this review describes the factors that influence the process (temperature, pressure, residence time, concentration, effect of the catalyst, effect of the reactor geometry, reactor design, heating rate of the biomass particle and type of biomass). Finally, this article concludes that the technology supercritical water gasification has great potential for a cleaner biogas production, with a high percentage of hydrogen, by different types of biomass, thus reducing the pollution and CO2 emissions.
Keywords: Biomass, Supercritical Water, Gasification, Supercritical Water Gasification, Supercritical Technology
Cite this paper: Lachos-Perez D., Juliana M. Prado, Torres-Mayanga P., Tânia Forster-Carneiro, M. Angela A. Meireles, Supercritical Water Gasification of Biomass for Hydrogen Production: Variable of the Process, Food and Public Health, Vol. 5 No. 3, 2015, pp. 92-101. doi: 10.5923/j.fph.20150503.05.
Article Outline
1. Introduction
- The development of renewable energy sources is gaining increasing importance on account of several factors primarily, because of significant shortages of fossil fuels (oil, gas and coal) scheduled for the next few decades as a result of the growing world demand, driven by developing countries. Secondarily, it is important to stress that the centralized production of these energy sources in certain areas of the world is essentially governed by political factors, which results in high and volatileprices. And finally the issues of global warming, mostly related to the releases of CO2 in the atmosphere caused by thermal power plants and, in general, by combustion plants using fossil fuels, will demand many national governments to seek alternative and more environmentally friendly ways to produce energy [1].In this context, biomass can be regarded as an effective source of renewable energy. Biomass is renewable by definition, and it can be seen as an alternative way of storing solar energy using plant tissues. The carbon dioxide released by its combustion (or possibly partial oxidation) derives from the carbon stored in the tissues of biomass throughout its life. Unlike fossil fuels, biomass is distributed more evenly around the world, and therefore implies geopolitical advantages [2]. Residues generated from biomass worldwide represent a great potential resource for energy achievement [3]. These residues can become a major renewable resource for the production of new chemicals, fuels and energy [4].Among the various proposed technologies for converting biomass into energy, gasification is quite promising [5]. The technology of sub/supercritical from lignocellulosic residues is in development, because of the complexity of the cellulosic matrix and the different reaction mechanisms that are favored by water in the subcritical and supercritical state. However, results show that the gasification in subcritical and supercritical environment favors, the rapid fractionation of hemicellulose and cellulose, respectively, because of the change of the ionic product, polarity and electrical conductivity of the water at high pressure and temperature [6], [7]. The SCWG of biomass for the production of hydrogen is a recent technology. The treatment of biomass in supercritical water (SCW) converts biomass into biogas fuel (rich in hydrogen, H2), which is easily separated from water by cooling at room temperature. SCWG process works with temperature above its critical point, and usually around 600°C, which makes water a strong oxidizing agent, resulting in the complete disintegration of the structure of the biomass through the transfer of oxygen from water to the carbon atoms of the substrate [8], [9]. The SCWG technology also offers a good opportunity for the gasification of biomass with high moisture content. There are some recent studies on the use of supercritical gasification for the conversion of raw materials with different percentages of moisture: high content of moisture (70-95%) – such as bagasse, seaweed, sewage sludge, plant waste (vegetables and fruits), stillage, wastewater – and low moisture content (peanut shells, corn husks, sugar cane, etc.) [9].Additionally, in these studies, different types of biomass were gasified in supercritical conditions with success and the gas obtained was composed of hydrogen, carbon dioxide, methane, carbon monoxide and a small amount of ethane and ethylene. Thus, SCWG is a highly efficient method for the production of renewable hydrogen [10].Therefore, the conversion of biomass by the sub/supercritical water process is a technology with enormous potential, and hydrolysis and gasification studies must be conducted to evaluate the influence of the main operational parameters, such as pressure, temperature, residence time, reactor design and heating rate, for biomasses with higher production biohydrogen yield [11].
2. Biomass
- Biomass is the fourth largest source of energy in the world. Projections for the future indicate that the importance of biomass will greatly increase, going so far as to represent 10 to 20% of all energy used by humanityat the end of the 21st century [12]. Biomass refers to all organic materials that originate from green plants, as a result of photosynthesis. It is a source of solar energy stored in the form of chemical energy, which can be released when the bonds between adjacent carbon, hydrogen and oxygen molecules are broken by various biological and thermochemical processes [13]. Biomass includes a wide range of organic materials, which are usually composed of inorganic constituents, water, cellulose, hemicellulose, lignin, lipids, proteins, starches and simple sugars [14]. Among these compounds, cellulose, hemicellulose and lignin are the three main components. As for the elementary carbon compositions (51% of dry weight) and oxygen (42% of dry weight), together they contribute with more than 90% of the dry weight of a typical biomass [15].The waste from the food industry or agriculture is produced in large quantities and its exploitation is of great interest. These wastes are a source of compounds with high added value, which can be made available by extracting and/or separating the components. The current use of these residues as livestock feed, fertilizer or fuel has promoted the development of promising new technologies for the exploitation of biomass [16]. Based mainly on the moisture content of the biomass, the type of biomass selected subsequently determines the most likely form of energy conversion process [13].
2.1. Biomass to Energy
- Bioenergy is a renewable and clean energy source, which is derived from biomass. It has been attracting a lot of attention currently because of the decrease in fossil fuel reserves and increase in greenhouse gases produced through the use of fossil fuels [5]. In recent years, concerns have been growing around the world about the environmental consequences of the strong dependence on fossil fuels, particularly climate change [17]. Therefore, it is very reasonable to foresee, that in a finite number of years, the fossil resources available will be exhausted. To this end, it is worth recalling the Hubbert peak theory, which predicts that global production of energy from other fossil resources, petroleum and follows a bell curve, with a peak of maximum production [18].Lignocellulosic biomass may be used in biorefineries contributing with lignocellulosic material (leaves, roots, stems, bark, bagasse, straw waste, seeds, pieces of wood, etc.), produced by agri-food industries in their daily operations, providing billion tons per year of energy supply and other chemicals with high added value production [19]. The lignocellulosic materials that contribute to biorefinery and gasification products are shown in Figure 1.
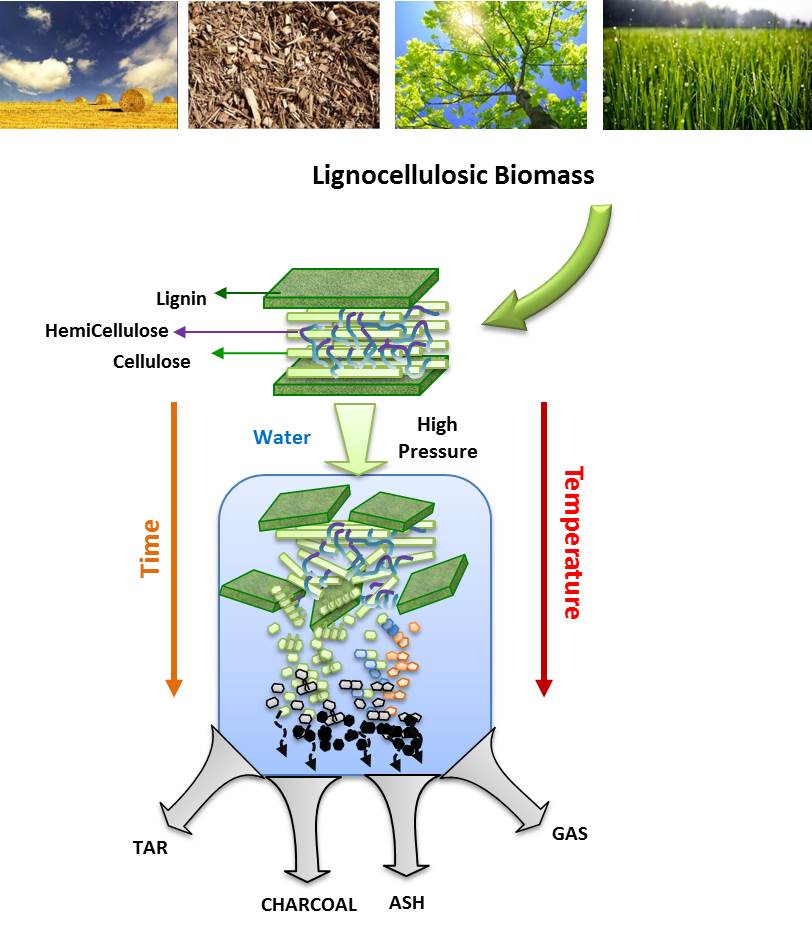 | Figure 1. The lignocellulosic materials contribute to biorefinery and gasification products. (Adapted from [20]) |
3. Supercritical Water Technology
- Water above its critical temperature, 374.1oC, and critical pressure, 22.1 MPa, is known as “Supercritical water” (SCW) [26]. The water in the supercritical state has similar properties of the viscosity as a gas and the density as a liquid (Table 1). When both the critical pressure and temperature are overcome, the water is not a liquid, as the liquid phase is not possible when the critical temperature is exceeded. However, the water is not yet able to be a gas, since it is not possible at pressures greater than the critical one. As a result, a completely new state is reached, which is called "supercritical state". Therefore, SCW is something halfway between a liquid and a gas. In fact, some of its characteristics are closer to those physical properties of a gas, while some other properties correspond to the properties of a liquid [27]. Consequently, during the last decades, there has been a great interest in the use of subcritical water as a solvent and reaction medium for converting biomass [28].
|
3.1. Properties of the Supercritical Water
- Density, dielectric constant and ionic product are three main properties that influence the gasification of biomass. SCW is well suited for gasification, on account of its favorable values for all these three properties, and as such these are briefly discussed below [23].
3.1.1. Density
- The density of the water varies continuously and, although its value decreases around the critical point, its values are much higher than in environmental conditions. For instance, at 400°C supercritical water (SCW) at 300 bar exhibits a density 200 times higher than atmospheric water vapor [31]. A low viscosity facilitates the transfer of mass and a density of a liquid promotes improved solvation [31].
3.1.2. Dielectricconstant
- Water at room temperature has a dielectric constant of approximately 80, which makes it a strong enough polar solvent. When it is heated and compressed to the supercritical state, the dielectric constant drops to the typical values of a non-polar solvent [18], [32]. A consequence of the change in density is that when the density decreases, the number of hydrogen bonds also decreases, and its strength is weakened, manifested by the decrease in dielectric constant [33].
3.1.3. Ionicproduct
- Finally, another important feature of SCW is its ionic product. At 25 MPa, the ionic product of water at room temperature is 10-14 and increases to a maximum of 10-11 at approximately 200-300°C, but when the temperature increases even further, 550°C, the ionic product drops below 10-23 (mol/kg)2 [33]. The ionic product affects the ability to solubilize the inorganic salts of the supercritical water technology. Most ionic substances such as inorganic salts are soluble in subcritical water, but are nearly insoluble in the typical conditions when SCW gasifies.
4. Supercritical Water Gasification (SCWG)
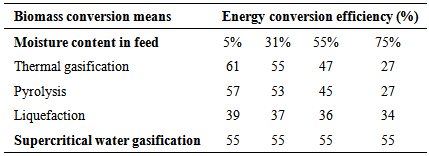
- SCWG is an excellent means for extracting energy from biomass. By adjusting the temperature or pressure, the characteristics of the supercritical technology can be modified, which in turn change the chemical reaction and the kinetics, thus producing compounds products in the desired concentrations. Because of the unique properties of the SCW, the thermodynamic equilibrium and high rates of chemical reactions are possible. The supercritical gasification lets of the high solid conversion (more than 99%) and a high concentration of hydrogen product gas (up 50%) with the suppression of formation of char and tar [34]. Table 2 compares the energy conversion efficiency and the moisture content present in the biomass. It is observed that as the moisture content increases, the energy conversion efficiency decreases to conventional technologies, but in the case of supercritical water gasification, efficiency remains constant. The gasification in supercritical water therefore offers the best option for high-moisture biomass gasification. In the case of biomass with high percentage of moisture (55 and 75%, respectively), the conversion efficiency of the SCWG is generally higher when compared with other technologies such as conventional thermal gasification (47-27%), pyrolysis (45-42%), anaerobic digestion (31-35%) and liquefaction (36-34%). However, when the biomass has low moisture content (5%), the conversion efficiency of the thermal gasification is higher (61%) than the SCWG (55%) [24].
|
4.1. Advantages of Supercritical Water Gasification with Respect to Conventional Gasification
- Most biomass organic materials can be dissolved in supercritical water for a relatively high dielectric constant of supercritical water (SCW), thus the biomass SCWG is a homogeneous reaction, which can decrease the mass transfer resistance between phases.● High pressure of the gaseous product enables the transportation, usage, carbon capture and further purification of the product gas through steam reforming or PSA (pressure swing adsorption).● Higher energy efficiency can be achieved in SCWG especially for biomass with high moisture content as the drying process is not required in the SCWG.● The reaction temperature is much lower than that in conventional gasification and pyrolysis. For example, the temperature of conventional steam gasification is always above 1000°C, while the complete gasification of glucose can be achieved at 650°C, 35.4MPa, in SCWG.● The gaseous product can be very clean, as almost no NOx and SOx were generated in SCWG, and the CO concentration is very low, especially with the catalyst to enhance the water-gas shift reaction [37].
4.2. Kinetics in the Process of Supercritical Water Gasification
- Rate laws and kinetic parameters are essential for reactor designs and the estimation of distribution of the product. The published articles of kinetic models for SCWG [40-44] focused solely on income or gasification feedstock conversion, without capturing the pathways that lead to the formation and conversion of gas species. As a result, little is known about the rates of different potential reaction paths. Among the three largest components of the biomass, cellulose, hemicellulose and lignin, the first has been studied more extensively. A simplified image of the liquefaction and/or gasification reaction pathways for the major components of the biomass, i.e. cellulose, is shown in Figure 2.Figure 2 shows two parallel paths for the conversion. The one on the left is via the free radical reaction, preferred by the supercritical condition (high temperature and/or low pressure), while the one on the right is the Ionic reaction, preferred by the subcritical condition (low temperature and/or increased pressure)[9], [35], [44]. As gases are typical products of free radicalreactions, the formation of gaseous products occurs at higher temperatures (above the critical temperature) [44].
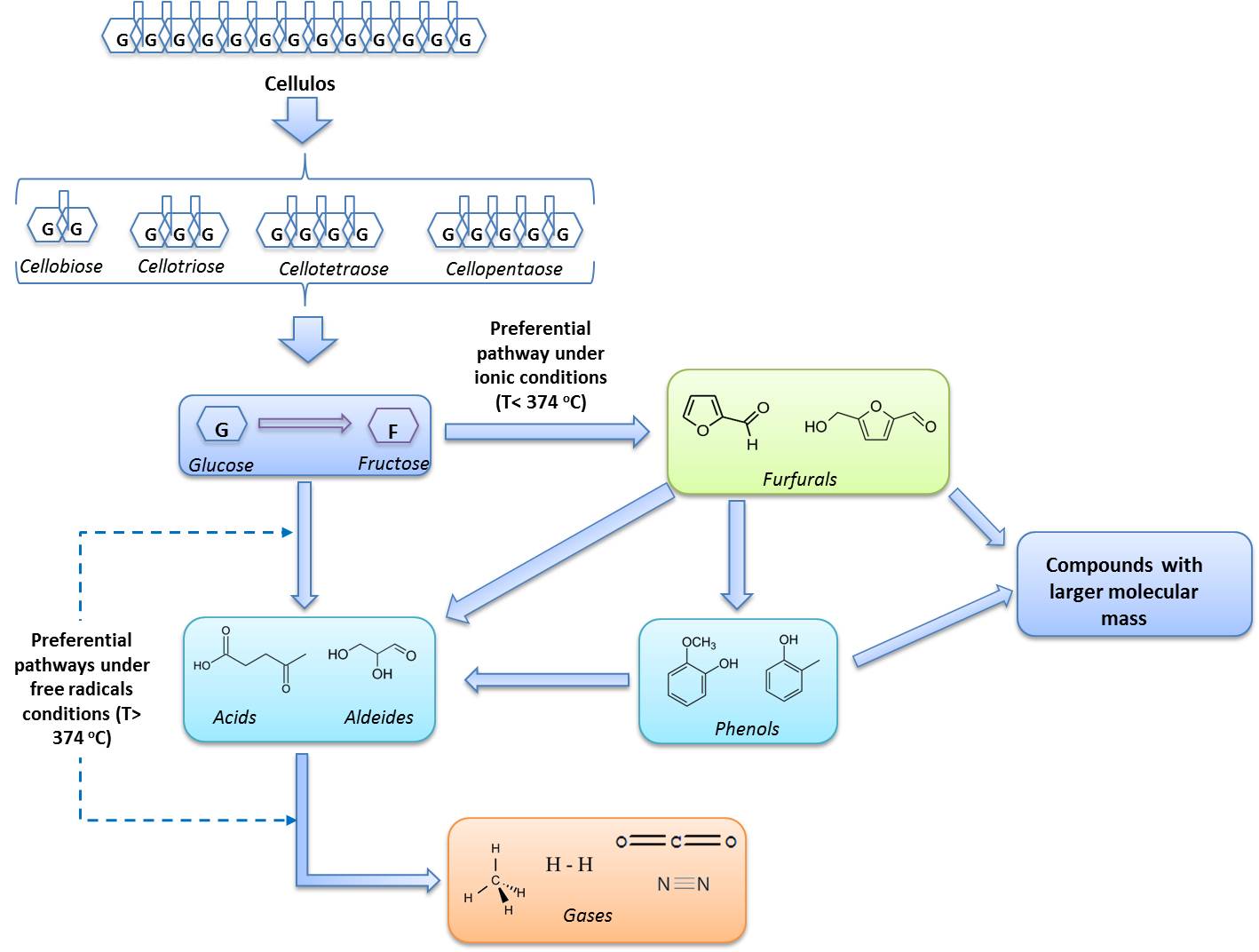 | Figure 2. Primary reaction routes for the liquefaction of pulp and/or gasification. Adapted from [43] |
4.3. Chemical Reactions
- The overall chemical reaction for the SCWG of biomass hydrogen can be represented by the following simplified reaction [45]:

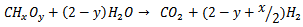
 (Reaction 1)Where x and y are the elemental molar ratios of H/C and O/C of the biomass, respectively. The product of the reaction is the synthesis gas, whose quality depends on x and y. Reaction 1 is endothermic. It is known from Reaction 1 is not only the water solvent, but a reagent and the hydrogen in the water are released by the gasification reaction. After Reaction 1, three intermediate competitive reactions may occur during the SCWG of biomass [45].● Steam reformation
(Reaction 1)Where x and y are the elemental molar ratios of H/C and O/C of the biomass, respectively. The product of the reaction is the synthesis gas, whose quality depends on x and y. Reaction 1 is endothermic. It is known from Reaction 1 is not only the water solvent, but a reagent and the hydrogen in the water are released by the gasification reaction. After Reaction 1, three intermediate competitive reactions may occur during the SCWG of biomass [45].● Steam reformation
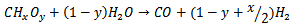
 (Reaction 2)● Steam shift reaction
(Reaction 2)● Steam shift reaction
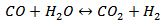
 (Reaction 3)● Methanation
(Reaction 3)● Methanation
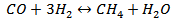
 (Reaction 4)
(Reaction 4)4.4. The Kinetics of the Supercritical Water Gasification of Biomass
- For the determination of the kinetics of the SCWG of biomass, various researchers, such as D'Jesús et al. [42] and D'Jesús et al.[46], used tubular flow reactors. It can be seen as an ideal piston flow reactor when it meets the criteria below:

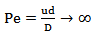
 (Equation 1)Where: Pe = Péclet numberu = Axial speed (m/s)d = Diameter of the ReactorD = Axial dispersion coefficient (m/s2)When the length/diameter ratio (L/d) is greater than 100, the axial dispersion coefficient tends to 0, as expressed in Equation 2.
(Equation 1)Where: Pe = Péclet numberu = Axial speed (m/s)d = Diameter of the ReactorD = Axial dispersion coefficient (m/s2)When the length/diameter ratio (L/d) is greater than 100, the axial dispersion coefficient tends to 0, as expressed in Equation 2.
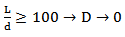
 (Equation 2)For an ideal piston flow reactor, the reaction is modeled according to D'Jesús et al. [42] and D'Jesús et al. [46]:
(Equation 2)For an ideal piston flow reactor, the reaction is modeled according to D'Jesús et al. [42] and D'Jesús et al. [46]:
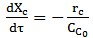
 (Equation 3)Where: XC = carbon conversion of biomass (mol C in gas/mol C in feedstock)rC = reaction rate of carbon, mol/L.s
(Equation 3)Where: XC = carbon conversion of biomass (mol C in gas/mol C in feedstock)rC = reaction rate of carbon, mol/L.s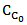 = initial concentration of carbon in the feed, mol/LDi Blasi et al. [47] and Lee et al. [39] used an expression for the Arrhenius type reaction rate, irreversible and of first order, assuming that the reactor operates at isothermal and isobaric conditions. This assumption gives
= initial concentration of carbon in the feed, mol/LDi Blasi et al. [47] and Lee et al. [39] used an expression for the Arrhenius type reaction rate, irreversible and of first order, assuming that the reactor operates at isothermal and isobaric conditions. This assumption gives 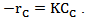 Equation 3 can be rearranged to:
Equation 3 can be rearranged to:
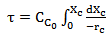
 (Equation 4)Where,
(Equation 4)Where, 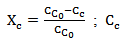 = cellulose content, wt%The residence time,
= cellulose content, wt%The residence time, 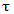 is given by experimental data and the reaction constant K can be estimated from:
is given by experimental data and the reaction constant K can be estimated from:
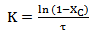
 (Equation 5)Assuming an Arrhenius type reaction, the reaction rate can be written as:
(Equation 5)Assuming an Arrhenius type reaction, the reaction rate can be written as:
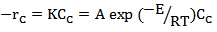
 (Equation 6)Where:A = pre-exponential factor, s-1E = activation energy, kJ/mol
(Equation 6)Where:A = pre-exponential factor, s-1E = activation energy, kJ/mol5. The influence factors in the Process of Supercritical Water Gasification
- SCWG is a relatively new field of research. It has not been explored fully. So far, the following parameters have been identified as the ones influencing the gasification of biomass in SCW [23]:
5.1. Temperature
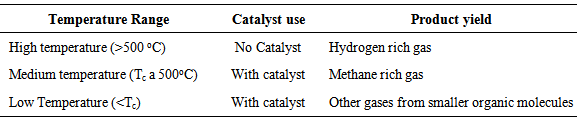
- The temperature shows a significant effect on the SCWG of biomass, particularly when no catalytic converter or less effective catalysts are present [45]. In fact, the process of pressurized water gasification can be divided into three groups (Table 3), depending on the primary product of the gasification [48]:
|
5.2. Pressure
- The effect of pressure on the SCWG of biomass is a complex thing. Bühler et al. [44] observed that the free radical reaction rate decreases with pressure because of the "cage effect", while the ionic reactions increase with the pressure because of ionic products in higher densities. The density dependence of these two types of reactions is opposite to each other [23]. In the experiment to evaluate the effect of pressure on the efficiency of the gasification (EG) and efficiency of the carbon (EC) of the SCWG of rice husk with the concentration of 2% of weight, 60 minutes and fixed residence time, it is noted that there is no significant difference in the pressure increase in EG and EC of 26 MPa for the temperatures of 500°C, 600°C and 700°C [23]. As a result, the ionic reaction rate increases, and the free radical reaction rate is contained with increased pressure. The hydrolysis reaction has a significant role in the SCWG, which requires the presence of H+ or OH-. With increased pressure, the ionic product increases, thus the hydrolysis rate also increases. In addition, high pressure promotes a change in the steam reaction, but it reduces the decomposition reaction rate [45].
5.3. Residence Time
- From the viewpoint of thermodynamics, biomass can be gasified completely in SCW with the formation of H2 and CO2. The gasification reaction was controlled by kinetics and few seconds were required to complete the gasification process. Antal et al. [49] gasified 0.1M glucose at 34.5MPa and 873 K with various residence times. They found that glucose can be gasified quickly and 100% EC was achieved with 28 s of residence time. Lee et al. [39] reported that the yields of all the gases remained almost constant at 973 K, being independent of the residence time, except for the shortest residence time of 10.4 s, when the 0.6 M glucose was gasified at 28 MPa. This way, one can speculate that the peak production for gases at a low temperature can occur in a greater residence time.
5.4. Concentration
- The efficiency of SCWG may vary depending on the concentration of the dry biomass. It is observed that higher concentrations in the feed are associated with lower yields of biomass converted into gas. In addition, feed with a low concentration of raw material is gasified completely. However, higher yields of methane (CH4) are obtained when employing high concentrations of raw material in the process [9]. In summary, to obtain a high yield of biogas with a high concentration of raw material in the feed, the conditions of high temperature, high heating rate and use of catalyst [49] are required.
5.5. Effect of the Catalyst
- To improve the economic efficiency of the SCWG, the improvement of the efficiency of the gasification, as well as a decrease in the operating temperature should be considered. To this end, the catalyst is a possible solution. However, the catalyst for SCWG must be different from the one used in conventional gasification under the particular conditions of operation, such as high pressure values, goal (hydrogen production, instead of synthesis gas) and specifics of the supercritical medium [34]. Generally, four types of catalysts are used for the SCWG in the literature: Alkaline, Activated charcoal, Metal, Metal oxide. A comparison study of the gas yield (a), for the SCWG of cellulose with various catalysts, was conducted by Lu et al. [9]. The results indicate that the maximum hydrogen production values were obtained using ruthenium/carbon (Ru/C) as catalyst, i.e. values rather than between 15 and 20 mol/kg (gas yield), when compared with other types of catalyst or without catalyst.
5.6. Effects of the Reactor Geometry
- Most of the experiments with the SCWG have been carried out in small reactors of few millimeters in diameter. The scale up of the results from such miniature reactors to reactors of several centimeters or even meters in diameter may be subjected to many uncertainties as such small reactors do not show the effect of the hydrodynamics, mass transfer and heat transfer inside the reactor [23]. A comparison study with a tubular reactor with different diameters, 6 and 9 mm, for supercritical gasification under the following operating conditions: temperature of 650ºC, 25 MPa of pressure, residence time of 15 s and biomass of rice straw with 2 wt% of CMC (Carboxymethyl cellulose) [9]. The results indicated greater yield of hydrogen gas of 21.21% in the reactor with smaller diameter (6 mm); on the other hand, in terms of efficiency of the gasification (EG), the highest values (90%) were obtained in the reactor with larger diameter (9 mm). Therefore, the effect of setting the reactor geometry on the gas yield function is an empirical parameter, in which the reactor with larger internal diameter seems to favor the efficiency of both the biomass gasification and carbon, as well as significantly reduce the concentrations of Total Organic Carbon (TOC), possibly by promoting the transfer of heat and the formation of gases in the process.
5.7. Effect of the Heating Rate
- A high heating rate generally favors the SCW gasification process of biomass [9], [50], while a slow heating up leads to the formation of coke/tar. One possible explanation for this is the formation of coke at low heating rates. At a low heating rate, the biomass-water mixture is gently heated from the subcritical temperature to the supercritical temperature. Thus, the feed spends a finite time at subcritical temperatures in the preheating section of the reactor. As explained in the previous section, the ionic reaction takes place at low temperatures forming intermediate products, furfurals or other unsaturated compounds. When the reactants reach supercritical temperature, the free radical reaction takes over [35], and the compounds polymerize forming coke/char. The formation of char in turn lowers the conversion of carbon. For the higher heating rate, the time spent in the subcritical region is short and the formation of coke/tar is reduced and a higher conversion is achieved. Matsumura et al. [51] examined the phenomenon and found that a lower amount of tar was produced at higher heating rates because of this.
5.8. Effect of the Particle of Biomass
- The particle size is an important issue in the design of any gas, because the smaller the size, the greater the energy consumption required for its suitability. Although the SCWG is still at the stage of introduction to the real scale, there are few studies that examine this issue. Conceptually, since the resistance to mass transfer in SCW is minimal, the particle size may not have a greater effect. Lu et al., [9] studied the gasification of rice straw and found that smaller particles of biomass (< 0.177 mm) result in higher hydrogen yield (17.00%), efficiency of gasification (EG) of 93.22% and efficiency of carbon (EC) of 77.28% during SCWG when compared with larger particles (0.177-0.4 mm). As the additional energy is required for the milling of biomass, an ideal particle size can be found considering the economic viability.
5.9. Effect of the Type of Biomass
- Cellulose, hemicellulose and lignin are the main components of biomass and they have different structures. Thus, the different components can have a different effect on the SCWG. Yoshida and Matsumura [52] investigated the interaction of cellulose, xylan (composite model for hemicellulose) and lignin, mixing them in different proportions in the SCWG. They found that the hydrogen yield for the SCWG of cellulose and hemicellulose are higher than lignin, and there was no apparent interaction between the hydrogen production from cellulose and hemicellulose in SCWG. In the mixture with lignin, the production of hydrogen from cellulose and hemicellulose SCWG was deleted. In a later study of Yoshida et al. [24], they showed that this effect depended on the species of lignin and the interaction between each component.
6. Conclusions
- The SCWG does not require drying; thus, the problem of drying is largely avoided by the supercritical water gasification and can be used for biomass with high percentage of moisture (55 and 75%, respectively). Studies suggest that greater energy efficiency can be achieved in the SCWG of biomass with high moisture content; they also show that the temperature of the reaction is much smaller than the conventional pyrolysis and traditional gasification, with almost no NOx and SOx, and that the concentration of CO is very low. The conversion efficiency of the supercritical water gasification is generally higher when compared with other technologies. Supercritical water gasification is still in a very early stage of development and more studies should be done to improve its efficiency. The emerging technologies "Supercritical Water Gasification" has a great potential for the recycling of biomass for the production of new products for hydrogen production.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML