-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Food and Public Health
p-ISSN: 2162-9412 e-ISSN: 2162-8440
2013; 3(3): 142-146
doi:10.5923/j.fph.20130303.05
Immunomodulatory Effect of Instant Tea Powder from Black Cincau (Mesona palustris BL) in the Treatment of Salmonella typhimurium-Induced Infection in Balb Mice
Tri Dewanti Widyaningsih1, Jaya Mahar1, Novita Wijayanti1, Rahmani2, Mukhammad Najmuddin2
1Food Science and Technology Department , Faculty of Agricultural Technology, Brawijaya University, Malang 65145, East Java-Indonesia
2Master Degree Student of, Food Science and Technology Department , Faculty of Agricultural Technology, Brawijaya University, Malang 65145, East Java-Indonesia
Correspondence to: Tri Dewanti Widyaningsih, Food Science and Technology Department , Faculty of Agricultural Technology, Brawijaya University, Malang 65145, East Java-Indonesia.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
Black cincau or black grass jelly (Mesona palustris BL) is a traditional beverage, believed to have a medicinal function in Indonesia. Black cincau is used to reduce hypertension, diabetes and liver disease. In China and Taiwan it is called Hsian-tsao (Mesona procumbens Hemsl). Previous research reports that Hsia-tsao extract contains bioactive compounds that have antioxidant, antibacterial, antimutagenic, hepatoprotecive, antihypertensive, antidiabetic, immunomodulatory and cancer preventive properties. The purpose of the present research was to examine the immunomodulatory property of a black cincau-based functional instant tea powder, using BALB mice infected by Salmonella thypimurium. The functional beverage with added cinnamon and pandan leaves had an IC50 of 141,25 ppm and total phenolic content of 4,49%. This functional beverage has the ability to increase interferon gamma (IFN-γ) and immunoglobulin G (IgG) in mice infected with the bacteria Salmonella thypimurium at dose of 18,2 mg/20 g body weight and 36,4 mg/20 g body weight respectively.
Keywords:
Black Cincau (Mesona palustris BL), Instant Tea Powder, Immunomodulators, Interferon Gamma (IFN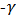 ), Immunoglobulin G (IgG)
), Immunoglobulin G (IgG)
Cite this paper: Tri Dewanti Widyaningsih, Jaya Mahar, Novita Wijayanti, Rahmani, Mukhammad Najmuddin, Immunomodulatory Effect of Instant Tea Powder from Black Cincau (Mesona palustris BL) in the Treatment of Salmonella typhimurium-Induced Infection in Balb Mice, Food and Public Health, Vol. 3 No. 3, 2013, pp. 142-146. doi: 10.5923/j.fph.20130303.05.
Article Outline
1. Introduction
- Prevention of disease by increasing endurance is improving the effectiveness of the immune system so that immune cells can continue to fight the cause of the disease and the body can be protected from various diseases. Since human beings are born with specific and non specific immune systems, the body's defense system is expected to offer protection from a variety of bacteria, viruses, fungi, and other foreign substances that can cause various diseases[1].The immune response is required for three functions, such as: (1) the body's defense against infectious microorganisms, (2) the homeostasis of the body to eliminate old components, and (3) the destruction control of the mutated cells, especially malignant cells. In other words, the immune response can be defined as a system for the body to maintain a balance between the outside and inside environment of the body. Immunomodulator is a substance or drug that can restore imbalanced immune system by stimulating and improving the function of the immune system[2]. Black cincau (Mesona palustris BL) is a traditional food used as a mixture of dessert. Black cincau is also believed to have benefit as a medicine. Black cincau can be used as medicine for fever, abdominal pain, diarrhea, cough, mouth sores, and high blood pressure[3]. In China and Taiwan, black cincau called Hsian-tsao (Mesona procumbens Hemsl) has been shown to contain a variety of bioactive compounds that have antioxidants, antibacterial, antimutagenic, hepatoprotective, antihypertensive and antidiabetic properties[4,5, 6, 7]. The water extract of black cincau is a proven immunomodulator with ability to increase interferon gamma (IFN-γ) and immune surveillance components (NK cells, CD8+ and macrophages) in both healthy and benzo(a)pyrine-induced mice[8].Black cincau or grass jelly (Mesona palustris BL) is also a plant belonging to the Labiatae family. In Indonesia it called cincau hitam because the color of this jelly is translucent black. Black cincau is easy to identified thanks to the gel content similar to gelatin especially in the leaves. To test these plants, simply soaked them into water for about 5 minutes and then squeeze them. They will stick together like glues, the more sticky they are means the better quality. Uniquely, the gel content in black cincau is affected by the altitude. To get highest content of the gel must plant it minimum at 2000 meters above sea levels[8]. Many bioactive compounds, such as sterol compounds, stigmasterol, ß-sitosterol, triterpene compounds, oleanolic acid, and ursolic acid, have been isolated from Hsian-tsao [4,5,6].Considering the potential benefits of black cincau, it can be used as a functional food that is effective, stable and palatable. Present day consumers prefer instant drinks. Therefore, black cincau can be developed into a multifunctional beverage that can increase the activity of the immune system as well.In the present study instant tea powder beverage made up of black cincau, cinnamon and pandan leaves (6: 1: 1) was tasted for the ability to improve levels of interferon gamma (IFN-Υ) and immunoglobulin G (Ig G) in Balb mice infected with Salmonella typhimurium.
2. Materials and Methods
2.1. Materials
- (1) Instant tea powder made up of black cincau, cinnamon and pandan leaves (6:1:1) was found to the best combination based on previous research. (2) 2 months old male Balb mice (20 – 25 gram), mice diets. (3) Mouse IFN-γ ELISA Kit and Mouse Ig G ELISA Kit (Koma Biotech Inc.), chloroform, Salmonella thypimurium.
2.2. Equipments
- Equipment used in this experiment were: (1) equipment for total phenol analysis and antioxidant activity, such as: glassware, spectrophotometer UV 2100 Unico, water bath and vortex. (2) mice cage, analytical balance, surgery kit. (3) equipment for IFN-γ dan Ig G analysis, such as: disposable centrifuge tube 15 ml (Nune), micropippet, tip, vortex, and Micro-plate Reader Spectrophotometer (Bio-rad model 550), ELISA reader, eppendorf tube, autoclave and sterile glassware.
2.3. Total Poliphenol Content Assay
- Concentration of total polyphenol was determined by Follin Ciocalteu method with modifications[9], blank solution was prepared by adding 2 ml of 96% ehanol in a 10 ml test tube. Caffeic acid 1000 ppm was used as stock solution. 50 mg tannic acid was diluted with 50 ml of 96% ethanol. The sample was weighed approximately 1 mg and diluted with 2 ml of 96% ethanol in a 10 ml test tube.The standard solution and sample were added to 5 ml deionized water, 0.5 ml of Follin Ciocalteu reagent (50% v/v) and incubated for 5 minutes. Then it was homogenized with 1 ml of sodium carbonate solution (5% v/v), and incubated at room temperature and in the dark for 1 hour. After incubation, the solution was homogenized again. Then the total phenol content was measured with a spectrophotometer at a wavelength of 725 nm. The value of total phenolic content is interpreted by milligram equivalents of caffeic acid per gram extract (mg/g extract).
2.4. Determination of Antioxidant Activity by IC50 Method
- Antioxidant activity was determined according to the method with slight modification[10]. Sample was diluted with distilled water to concentration of 20, 40, 60 and 80 ppm, respectively. Each sample solution (4 ml) was added to 1 ml DPPH solution (0,2 mM). The reduction of DPPH was measured at 517 nm against a blank assay for 30 minutes. The percentage of radical inhibition in medium was calculated as the different of absorbance of the blank and absorbance of the sample divided by that of DPPH control at the same time multiplied by 100. It can be calculated by this formula:
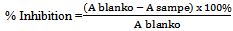 The value of sample concentration and inhibition percentage was graphically plotted for equation of linear regresion (y=ax+b). The equation was used for calculating the IC50 value (inhibitor concentration 50%) from each sample. The IC50 value is defined as the concentration of antioxidant (sample or control) necessary to decrease the initial DPPH concentration (50%) and is expressed as mg/mL.Determination of Immunomodulator by Interferon gamma (IFN-γ) and Immunoglobulin G (IgG) as indicators on mice
The value of sample concentration and inhibition percentage was graphically plotted for equation of linear regresion (y=ax+b). The equation was used for calculating the IC50 value (inhibitor concentration 50%) from each sample. The IC50 value is defined as the concentration of antioxidant (sample or control) necessary to decrease the initial DPPH concentration (50%) and is expressed as mg/mL.Determination of Immunomodulator by Interferon gamma (IFN-γ) and Immunoglobulin G (IgG) as indicators on mice2.5. Animals Treatment
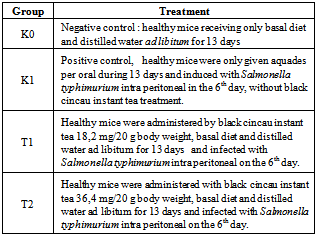
- Healthy mice were infected by Salmonella thyphimurium and there were four groups. First group was treated through the study by basal diet and water ad libitum without Salmonella thyphimurium induction as negative control. Second group was treated by basal diet and water ad libitum with Salmonella thyphimurium induction as positive control. Third and fourth groups were provided with black cincau instant tea at different dosage orally and induced by Salmonella thyphimurium on sixth day. All experiment procedures were approved by the Ethical Clearance Committe, Brawijaya University, Malang, Indonesia.
2.6. Dose Calculation
- Dose calculation was based on conversion of human equivalent dose to mice dose. Every mice received an equal volume of black grass jelly drink solution (0,2 ml/20 gram body weight) every day for 13 days orally by gastric gavage. Mice were weighed every 5 days.● The human consumtion of instant tea powder = 7 gram à for mice multiply by 0,0026● Dose 1 : instant tea dose for mice = 7 x 0,0026 = 0,0182 gram = 18,2 mg/20 gram mice● Dose 2 = 2 x Dose 1 = 36,4 mg/20 gram miceAfter 13 days of treatment, all mice were anesthetized by chloroform inhalation and sacrificed to collect the blood and remove the lymph for further analysis. ELISA method was used for IFN-γ and IgG analysis.
2.7. Blood and Lymphocytes Isolation from Mice Lymph
- Blood was collected from heart and placed in vac tube 5 ml that contain EDTA K3. The sample were centrifuged 4000 rpm for 7 minutes to precipitate the blood cell. Supernatant was taken and placed to eppendorf tube 1 ml for determination IFN-γ by ELISA kit.Lymph was removed with pippete and placed onto sterilized petridish. Steril phosphate buffer saline (sPBS) pH 7 was added to petridish. Lymph was pressed by using sterile spatula and filtered to remove debris. The sample suspension was placed onto sterile tube 15 ml and centrifuged at 1600 rpm for 5 minutes. 1 ml supernatant was centrifuged again at 5000 rpm for 10 minutes at 4°C. Supernatant was removed and added with 400 µL sPBS pH 7,2. Sample was ready for analysis.
|
2.8. Determination of IFN- γ Contain by ELISA Method
- ELISA procedur in this experiment was used with ELISA kit according to the method with slight modification. ELISA microplate (96 well) was washed with 300 µL wash buffer. 100 µL sample diluent was placed on the well (standard A1-H1 and A2-H2, H1 and H2 for blank) to maked standard curve. Then, the samples (A1 and A2) were added with 100 µL ml IFN-γ standard. Afer mixed, the sample from A1 and A2 was taken and placed in B1 and B2. This procedur was repeated until G1 and G2 because H1 and H2 was used as blank. Thus, there were seven IFN-γ standard solutions with different concentrations (1000, 500, 250, 125, 63, 32, and 16 pg/mL). Every standar solution was taken (50 µL), placed onto sample well and added with 50 µL sample. Then, every sample well was added with 50 µL biotin conjugate and incubated for 2 hours, 200 rpm at room temperature. Microplate well was washed 3 times by using 300 µL wash buffer, then all well was added with 100 µL TMB substrate solution and incubated for 10 minutes at room temperature. The reaction was stopped by adding 100 µL stop solution. After 30 minutes incubation, the sample was measured by ELISA reader at 450 nm to determine IFN-γ content[11].
2.9. Determination of Immunnoglobulin G (IgG) by ELISA Kit Method
- All wells of microplate were washed three times by 200 µL washing solution and added with 100 µL sample solution (duplo). Microplate was covered by seal and incubated for an hour. The samples were subsequently washed again four times, added with 100 µL staining solution and incubated for 3-30 minutes. The reaction was stopped by adding 100 µL stopper solution in every well. The samples were evaluated by ELISA reader 450 nm. Standar solution was also prepared for making standard curve[12] .
3. Result and Discussion
3.1. IC50 and Total Phenol Content of Instant Tea Powder from Black Cincau
- Instant tea powder from black cincau was made with addition of cinnamon extract and pandan leaves extract (6:1:1). Total phenol content was 4,49% and IC50 was 141,25 ppm. Instant tea powder-based black cincau was found to be the best treatment based on previous research.
3.2. Interferon Gama (IFN-γ)
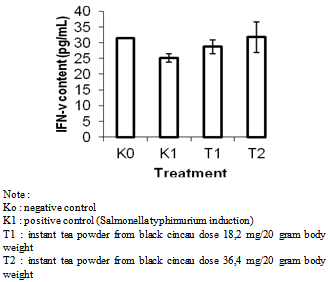
- Determination of IFN-γ content by Brown Forsythe method and followed by Games Howell method showed that the instant tea powder from blacj grass jelly treatment affected significantly to increase IFN-γ content. The increasing of IFN-γ content of treatment was showed in Figure 1.
3.3. Immunoglobulin G ( IgG)
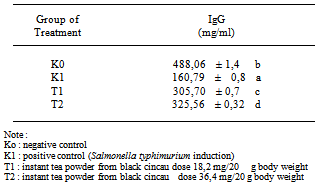
- Infection of mice by Salmonella typhimurium causes illness and triggers the production of immunoglobulins. The purpose of bacterial infection was to generate an effective immune response resultin in the production of immunoglobulins and memory cells. Humoural immune response plays a role in defense against extracellular microbes, mediated by antibody, while the defense against intracellular microbes such as viruses requires a cellular immune response[2].Determination of IgG content by Brown Forsythe and Games Howell methods showed that instant tea powder treatment significantly increase IgG content (see Table 2). Instant tea powder from black cincau T1, dose 18,2 mg/20 g body weigh dan T2 dose 36,4 mg/20 g body weight was increased IgG content in mice. This indicates that extract instant tea powder from black cincau also possess some mitogenic activity, which can enhances the IgG secretion. Some of the components bioactiv from black cincau: phenol, flavonoid, ß-sitosterol, stigmasterol, and caffeic acid[4], these components might add to the mitogenic effect which in turn might stimulate the secretion of IgG[15].
|
4. Conclusions
- Instant tea powder from black cincau at dose of 18,2 mg/20 g body weigh and dose 36,4 mg/20 g body weigh can increase IFN-γ and IgG content in Salmonella typhimurium infected mice. Therefore, this fungsional beverage can be used as an immunomodulator.
ACKNOWLEDGEMENTS
- The authors gratefully acknowledge the financial support for this research by “Penelitian Hibah Kompetensi” Project of Directorate General Higher Education, Ministry of National Education, Indonesia.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML