-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Food Science and Nutrition Engineering
p-ISSN: 2166-5168 e-ISSN: 2166-5192
2012; 2(6): 113-118
doi:10.5923/j.food.20120206.03
Effect of Ultrasonic Treatment on Buffalo Milk Homogenization and Numbers of Bacteria
Asaad Rehman Saeed Al-Hilphy, Alaa Kareem Niamah, Ammar B. Al-Temimi
Department of Food Science, College of Agriculture, University of Basrah, Basrah, Iraq
Correspondence to: Alaa Kareem Niamah, Department of Food Science, College of Agriculture, University of Basrah, Basrah, Iraq.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
The effect of ultrasonic treatment on milk homogenization and numbers of bacterial were studied. The results showed that ultrasound treatments with high power had an important effect on milk homogenization. The lowest homogenization indices (high efficiency) were 2 and 3% at 430 and 338W respectivily. The homogenization index was reduced with increasing time of homogenization with ultrasonic treatments. The mean D-values were 6.47 and 5.49 min. for ultrasonic treatments of 338 and 430 W respectively.
Keywords: Buffalo Milk, Bacteria, Homogenization, Ultrasonic
Cite this paper: Asaad Rehman Saeed Al-Hilphy, Alaa Kareem Niamah, Ammar B. Al-Temimi, Effect of Ultrasonic Treatment on Buffalo Milk Homogenization and Numbers of Bacteria, International Journal of Food Science and Nutrition Engineering, Vol. 2 No. 6, 2012, pp. 113-118. doi: 10.5923/j.food.20120206.03.
Article Outline
1. Introduction
- Utilization of high intensity ultrasound in food technology and biotechnology is in the sense of this progressive technology usage on most usable processes in food industry. Those include mixing, drying, homogenization, crystallization, grinding and many other operations. High intensity ultrasound is capable to change physical and chemical characteristics of treated materials[1].Ultrasound is of great investment in food processing for many reasons[2]. This technique can be generally used in systems which are liquids and can be defined as waves with high frequency[3]. High amplitude of ultra sound and longer exposure time ensure greatest effect on degree of homogenization. The mechanisms which are contributing to fat globules disruption is known as the capillary wave's mechanisms. Decreasing of fat globules is possible only if the diameter of fat globules is significantly larger than the oscillation wave length and for oil – water systems is approximately 10 µm. Cavitation is the most accepted mechanism for ultrasound emulsification. This mechanism is based on the implosion bubbles which produce powered shock waves in the milk surrounding the ultrasonic probe and jets of high velocity. This micro jet effectively causes fat globules disruption[4]. Ultrasound has various applications in the food industry, including killing or inhibiting bacteria. Historically, the effectiveness of low intensity ultrasound in inactivating bacterial cells has been limited by the protection afforded to the organisms by the food environment. Recently, however, systems with high output of ultrasonic energy at low frequency have greatly increased the lethal effect on bacteria[5]. In general, inactivation of the cells is more pronounced at an elevated power level and as the processing time increases. One of non traditional types of milk processing is ultrasonic processing. At present time, ultrasonic devices for homogenization are used at milk processing[7]. Ultrasound treatment with high amplitude (power) hands an impotent effect on milk homogenization compared to conventional homogenization. As exposure time and power levels increase homogenization efficiency also rise. The highest homogenization efficiency and the smallest fat globule diameter are 3.22 and 0.725 µm at power level 100 (450 W) for 10 min. respectively. The fat globule diameters at power level of 40 (180 W) for 10 min are similar to those of conventional homogenization[8].Increase of the power (100Watt) lead to increase of degree of stated homogenization, and decrease in size of fat globules in soybean milk[9]. Homogenization must always be sufficiently efficient to prevent creaming. The result can be checked by determining the homogenization index. The index for homogenized milk should be in the range of 1 to 10. The effect of homogenization on the physical structure of milk has many advantages: Smaller fat globules leading to no cream-line formation, whiter and more appetizing color, reduced sensitivity to fat oxidation, more full-bodied flavor, better mouth feel and better stability of cultured milk products[10]. Ultrasound treatment with high amplitude (power) has an important effect on milk homogenization compared with conventional homogenization and the best homogenization and the smallest fat globule diameter (0.725µm) are obtained at a power level of 10 minutes[8]. The objective of our study was to investigate the effect of different of ultrasound treatments on buffalo milk homogenization and numbers of bacterial cells during different times to compare these with conventional homogenization
2. Materails and Methods
2.1. Materials
- Buffalo milk was obtained from the dairy farm at Karmt Ali –Basrah city, Iraq.
2.1.1. Used Cultural Media
- Commercially media were used for total bacterial count, coliform bacteria and staphylococci. The media used for bacteriological analysis were nutrient agar , MacConkey agar and mannitol Salt agar (Hi-Media, India).
2.2. Methods
2.2.1. Conventional Homogenization
- The conventional homogenizer (Kirchfeld-Germany) was used to homogenize the milk under a high pressure of 200 bar at 55 oC .
2.2.2. Ultrasonic Homogenization
- An Ultrasonic homogenizer (Son prep 150 MSE, Korea) equipped with 0.5 cm diameter tip and working at a constant frequency (28 kHz) was used to homogenize 150 ml of buffalo milk for each treatment. The ultrasound probe was immersed in to the milk at depth of approximately 4 cm. The experiment was carried out using different power levels. Exposure times were 5, 10 and 15 min for each trail. The samples were kept at 20oC. The power trail of 99, and 77% corresponding to approximately 430 and 338 W, respectively was used.
2.2.3. Homogenization Index
- The homogenized milk was placed in to a graduated cylinder of 150 ml and kept in a refrigerator for 48 hr. then the fat content of sample for the upper part, i.e. 1/10 (a) and from the bottom, i.e. 9/10 (b) of the graduated cylinder was determined by using the Gerber method[9]. The following equation was use to calculate the homogenization index (refer to reverse homogenization efficiency) ηh of the sample[8, 10].
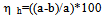 | (1) |
2.2.4. D-value
- D-value was calculated from the following equation[11]:
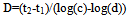 | (2) |
2.2.5. Microscopic Test
- The homogenized milk and control sample were showed by Bright-Field light microscope and effects for fat milk were noted.
2.2.6. Total Bacterial Count
- The Standard Plate Count (SPC) gives an indication of the total number of aerobic bacteria present in the samples[12]. The original sample is diluted five times to decrease the bacteria numbers sufficiently. One ml from last two dilutions is then dispensed into a petri plate and incubated at 35°C for 24 hours and the pour plate method was carried out. All counts were expressed as (CFU/ml) of milk [12].
2.3. Statistical Analysis
- One way ANOVA and post – hoc comparison statistical analyses which performed by using SPSS 11.0 statistical packages are applied [13].
3. Results and Discussion
3.1. Homogenization Index
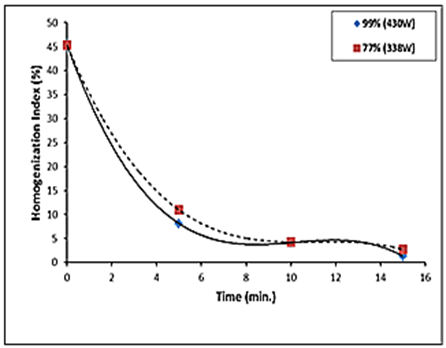 | Figure 1. Homogenization index vs. time for different ultrasonic treatments |
 reduced with increasing time at the power of 430 and 338 W (Fig. 1). The maximum homogenization index was in the control. Homogenization index was significantly
reduced with increasing time at the power of 430 and 338 W (Fig. 1). The maximum homogenization index was in the control. Homogenization index was significantly  reduced with increasing time in the ultrsonic treatments. Reducing homogenization index that means high homogenization efficiency. A minimum homogenization index was 2 and 3% at 430 W and 338W respectivily at 15 min.These results agree with Teknatext [10] who stated that homogenization must always be sufficiently efficient to prevent creaming. The result can be checked by determining the homogenization index. The index for homogenized milk should be in the range of 1 to 10%. The impircal equations for calculating homogenization index (%) at 338W and 430W were:
reduced with increasing time in the ultrsonic treatments. Reducing homogenization index that means high homogenization efficiency. A minimum homogenization index was 2 and 3% at 430 W and 338W respectivily at 15 min.These results agree with Teknatext [10] who stated that homogenization must always be sufficiently efficient to prevent creaming. The result can be checked by determining the homogenization index. The index for homogenized milk should be in the range of 1 to 10%. The impircal equations for calculating homogenization index (%) at 338W and 430W were: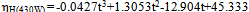 | (3) |
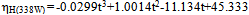 | (4) |
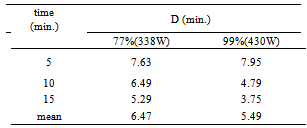
3.2. D-value
- The microbial destruction rate is generally defined in terms of a decimal reduction time (D-value) which is the heating time in minutes at a given temperature required to result in one decimal reduction in the surviving microbial population. In other words, D value represents a heating time that results in 90% destruction of the existing microbial population[11].The mean D-values were 6.47 and 5.49 min. for ultrasonic treatments at 338 and 430 W, respectively (Table 1). D-value was significantly (p<0.05) reduced with increasing time and power.
|
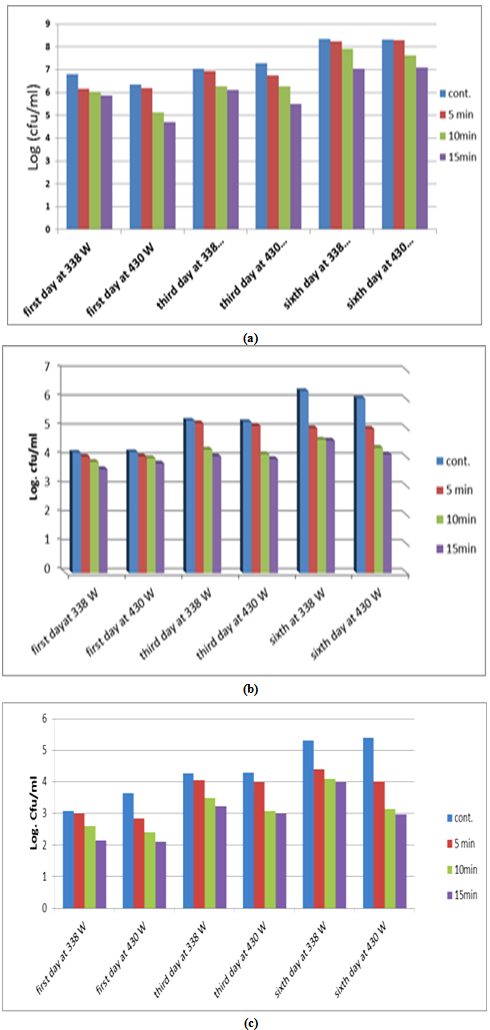
3.3. Effect of Ultrasonic on Bacterial Numbers
- Figure (2) illustrates the effect of ultrasonic on the numbers of bacteria in samples of buffalo milk and shows that the logarithms of numbers of bacteria were significantly (p<0.05) decreased with increasing power and increasing time of exposure compared with the control sample. The logarithms of the total numbers of aerobic bacteria after treatment directly were (5.87and 4.71 CFU/ml) when the powers were 388 and 430w respectively for a period of 15 minutes, and got increased by for storage where the logarithms of the total numbers of bacteria on the sixth day became (7.04 and 7.08 logCFU/ml) at 388 and 430w respectively for a period of 15 minutes and the logarithms of the numbers of bacteria were (8.34 and 8.32 logCFU/ml) at 388 and 430w respectively (Fig. 2-1). The effect of ultrasonic treatment was evident in reduction of the logarithms of numbers of the coliform bacteria (Fig. 2-2) as the logarithms of numbers after treatment directly were (2.11 and 2.14 logCFU/ml) at 388 and 430w respectively for a period of 15 minutes compared with samples of control (3.64 and 3.08), respectively. Also, the staphylococci numbers were decreased compared with samples of control and the logarithms of numbers of bacteria were (3.6 and 3.8 logCFU/ml) at 388 and 430w respectively for a period of 15 minutes while the control samples had (4.19 and 4.17) at 388 and 430w respectively (Fig. 2-3). Ultrasound causes cavitation's in aqueous solutions, which is an effective factor in damaging the cell wall of the micro-organisms, when a bubble collapses, a strong shear rate is generated in the environment that breaks the chemical bounds in the cells’ wall and membranes[14]. The survival of Lactic acid bacteria was very low in very long exposures of ultrasound[15].
3.4. Effect of Ultrasound on the Fat Globules
|
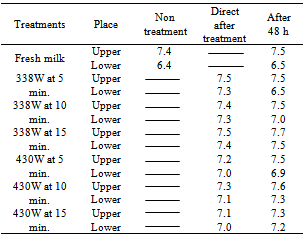
3.5. Effect of Ultrasound on the Fat Globules
- Figure 3 shows pictures of fat globules before and after ultrasound treatment and noted that the significantly (p<0.05) increase in frequency and duration produces fat globules smaller Compared with non-homogenization sample (sample No. 1) and a sample at device homogenizer (sample No.8) , The percentage of fat in the upper layer of the milk samples of no more than 10% for the bottom layer after 48 hours in all samples. Except for a sample of control and treatment 338w at 5min (table-2). It can be concluded that ultrasonic treatment of milk produced buffalo milk and homogenized fat globules were distributed uniformly in all parts of the sample . The percentage of fat in the upper layer of non –treatment sample is 7.4 and 6.5 in low layer but after 48 h is 7.9 , 4.9 respectively.
4. Conclusions
- The obtained data from using ultrasonic treatments in the buffalo milk homogenization showed that the homogenization index was reduced with increasing time, and power. D-value was decreased with increasing time and power. The logarithms of numbers of bacteria were decreased with increasing power and increasing exposure to ultrasonic compared with sample of control.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML