-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Clinical Medicine and Diagnostics
p-ISSN: 2163-1433 e-ISSN: 2163-1441
2019; 9(4): 68-73
doi:10.5923/j.cmd.20190904.02

Role of 18F-FDG PET/CT in Detecting Extrahepatic Metastases in Cirrhotic Patients with Hepatocellular Carcinoma in Pre Liver Transplantation
Ekram Ali1, Mahasen Abou-Gabal2, Mohamed El-Hussieny3, Hosna Mustafa2
1Nuclear Medicine Department, Sohag Oncology Center, Sohag, Egypt
2Kasr Al-Ainy Center of Oncology and Nuclear Medicine, Cairo University, Cairo, Egypt
3Radiology Department, National Liver Institute, Minoufiya University, Minoufiya, Egypt
Correspondence to: Ekram Ali, Nuclear Medicine Department, Sohag Oncology Center, Sohag, Egypt.
| Email: |  |
Copyright © 2019 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Aim of the work: to evaluate the role of 18F-FDG PET/CT in the assessment of extrahepatic metastases in patients with liver cirrhosis and hepatocellular carcinoma pre liver transplantation. Patients and Methods: analysis of 80 patients retrospectively with cirrhotic liver and HCC. All patients are histopathologically proofed to have HCC. All patients were subjected to triphasic CT and whole-body 18F-FDG PET/CT. All of them were received chemotherapy and radio frequency ablation. Results: the studied patients were 75 males (93.8%) and 5 females (6.3%), the mean age of 54.45+10.19 years. Seventy patients with poorly differentiated hepatocellular carcinoma, 2 of them had moderately differentiated hepatocellular carcinoma, 8 of them had well-differentiated HCC, alpha-fetoprotein was (100-400 ng/ml) in 10 patients and >400 ng/ml in 70 patients, 68 patients had extrahepatic sites, bone metastases, and adrenal metastases.
Keywords: 18F-FDG PET/CT, Extrahepatic metastases in patients with liver cirrhosis with HCC
Cite this paper: Ekram Ali, Mahasen Abou-Gabal, Mohamed El-Hussieny, Hosna Mustafa, Role of 18F-FDG PET/CT in Detecting Extrahepatic Metastases in Cirrhotic Patients with Hepatocellular Carcinoma in Pre Liver Transplantation, Clinical Medicine and Diagnostics, Vol. 9 No. 4, 2019, pp. 68-73. doi: 10.5923/j.cmd.20190904.02.
Article Outline
1. Introduction
- Hepatocellular carcinoma is an epithelial cell tumor of the liver, similar to normal hepatocytes [1]. It is representing the fifth most common tumor in the world, increasing incidence, especially in Western nations [2]. 80% of cases of HCC developing in patients with liver cirrhosis [3]. The annual incidence of HCC ranges from 2% to 8% [4]. The highest incidence of HCC in those infected with hepatitis C virus or hepatitis B virus, the highest incidence of severity in those with decompensated cirrhosis, epidemiologically it is higher in Japan compared to Europe or the United States, and higher in the male gender.The risk is significantly higher in individuals with multiple risk factors as well as in those co-infected with human immunodeficiency virus [5]. Patients without cirrhosis also may develop HCC [6].The early detection of HCC is important for improving the survival of affected patients. Clinical guidelines advocate surveillance of patients at risk due to cirrhosis or chronic viral hepatitis [7]. Multiphasic CT and MR imaging with extracellular agents were endorsed by guideline as first-line modalities for this purpose [8].The present study is a retrospective study for assessment of the role of 18F-DG PET/CT in the evaluation of extrahepatic metastasis.A significant advantage of 18F-FDG PET/CT is the capability to perform full-body examinations, the potential to detect extrahepatic metastases in one single examination and in differentiating new active disease from scar or necrotic tissue [9].The whole-body nature of the 18F-FDG PET/CT study also contributes to the increased sensitivity through detection of distant metastatic lesions. Tumors with intense 18F-FDG uptake are metabolically active and biologically aggressive [10,11]. The aim of this work is to asses extrahepatic metastatic spread using 18F-FDG PET/CT.
2. Patients and Methods
- A retrospective analysis of all cases of Hepatocellular carcinoma referred to Alpha scans Center during the period November 2011 to December 2014. All patients received chemotherapy and/or radiotherapy. Almost all patients were subjected to Tri-phasic CT, and 18F-FDG PET/CT in 40 patients included in this study and proved the histopathological diagnosis.
2.1. Eligibility Criteria
- All patients were evaluated with alpha-fetoprotein measurement if available, histopathology confirmation, and the ethical committee of NEMROCK center has approved the study.Patients with previous chemotherapy or radiation, Presence of clinical suspicious of second cancer and patient with no available histopathological prove.
2.2. PET/CT Imaging
- Patients were fasting for 6 hours before the 18F-FDG PET/CT study. Diabetic patients can be fast for 4.5 h after taking their oral medication together with a meal with blood sugar should be controlled, and less than 160 mg/ml.PET images were done during normal breathing in the three-dimensional mode for 60 minutes 4 minutes per bed position after intravenous administration of 18F-FDG (5 MBq/kg of body mass) in an intravenous line connected to saline. 18F-FDG PET/CT imaging will be performed using a 64-detector row LYSO PET/CT (Gemini TF, Philips Medical Systems).Approximately 60 minutes after 18FDG was injected, a scout image will be initially obtained for subject localization. The whole body, from the skull base to med thighs, volumetric axial CT will be obtained using a low-dose protocol after the administration of oral manitol 5%. Subsequently, 3D PET data will be acquired using about 11 frames lasting 1.30 minutes each using sequential 50% overlapping scans; PET images are reconstructed by using standard reconstruction algorithm Ordered Subsets – Expectations Maximization (OSEM).Attenuation correction of 18FDG-PET/CT images is performed by using attenuation data from the low dose CT component of the examination; emission data are corrected for scatter, random events and dead-time by using the manufacturer’s software.
2.3. Excluding Criteria
- Uncontrolled diabetes.
2.4. Criteria for Tri-phasic CT and PET/CT Interpretation
2.4.1. PET/CT Interpretation
- Interpretation of PET/CT scans was done independently by 2 experienced physicians blinded to the clinical situation, and any disagreement was resolved by consensus.Qualitative evaluation: A visually abnormal focus of 18FDG-PET uptake was reported as a focal uptake relatively higher than that of surrounding tissue with no similar activity seen in the contralateral side of the body.Quantitative evaluation: The intensity of FDG uptake within the specific lesion is calculated by using SUV (a volume of interest over the lesion).
2.4.2. Tri-phasic CT Imaging
- The CT component of the study comprises a multi-detector CT examination, was done from the base of the skull to the upper thighs (120 mA, 140 kVp, table speed = 13.5 mm per rotation). The CT images were also used for attenuation correction of the PET images, based on the use of complementary Tri-phasic CT scan in all patients have hyperenhancement, iso/mixed enhancement and iso/mixed enhancing pattern in arterial, porto-venous and equilibrium phases were suggested to be malignant lesions respectively. Also, metastasis appears hypervascular enhancing on arterial phase with the mixed pattern on port venous and equilibrium phase. However, hypo-vascular metastasis appears hypo-enhancing on arterial phase and shows maximum enhancement on porto-venous phase.
2.4.3. Pathological Assessment
- Final assessment was done in correlation to pathological diagnosis for different grade of HCC.
2.5. Statistical Analysis
- The data were entered using the statistical package SPSS version 25. The data were summarized using descriptive statistics for qualitative and quantitative variables which are not normally distributed. Number and percentage were used for qualitative values. The Chi-square test was used for qualitative variables. Sensitivity, specificity, positive.
3. Results
- The current retrospective study included 80 with hepatic lesions referred to Alfa Scan Radiology Centre proved to have malignant lesions in 40 patients. The study included 75 males (93.8%) and 5 females (6.3%), the mean age of 54.45±10.19 (Figure 1).
 | Figure 1. Distribution of sex in patients of the studied group |
3.1. Alpha-fetoprotein Measurements
- Serum Alfa fetoprotein (AFP) level was documented in all patients. 10 (22.5%) patients had AFP level (100-400) ng/ml, while 70 (87.5%) patients had AFP > 400 ng/ml.Predictive value; NPV and total accuracy were used to test the validity of studied parameters. Kappa agreement measure was used to test agreement between studied parameters. P-Values less than or equal 0.05 were considered statistically significant. Comparison of results of PET/CT with CT and histopathology was done on patients-based analysis.
3.2. Diagnostic CT Scans
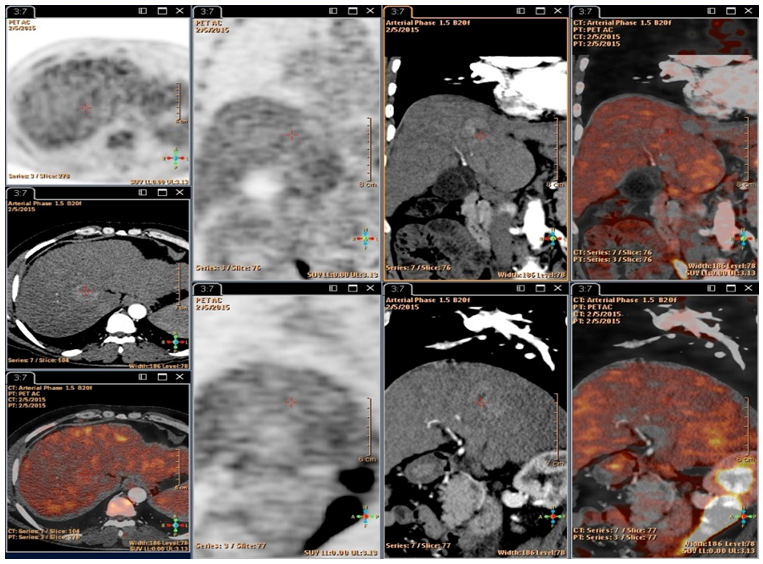
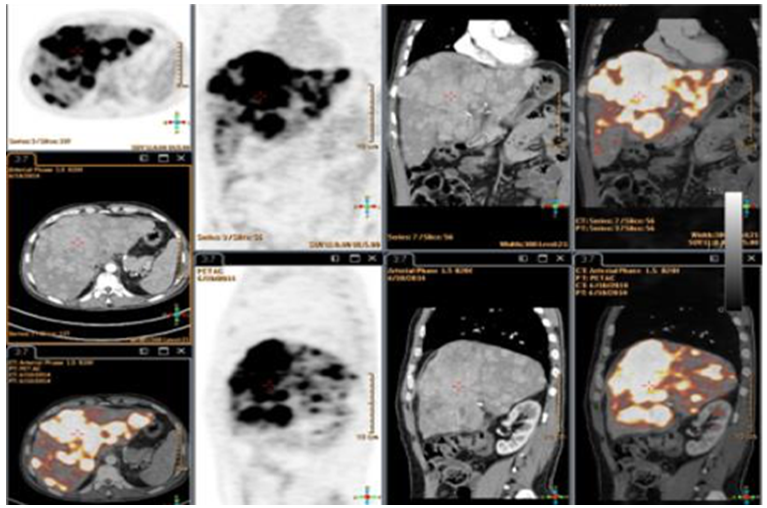
- All of the 80 patients had abdominal Tri-phasic CT. CT showed hepatic focal lesion with criteria of HCC with extrahepatic in 68 patients, and CT was negative in 12 patients.18FDG-PET/CT scans: 68 patients (85%) were positive for metabolically active extrahepatic metastases. While 12 (15%) patients had HCC with no extrahepatic metastases.
3.3. SUV Calculations
- Semi-quantitative analysis; SUV was obtained for liver parenchyma as well as HCC hepatic lesions, extra-hepatic LN involvement, and distant metastases. Mean SUV for liver lesions and normal liver parenchyma in the 80 patients was maximum ranging from 2.3-25, while the SUV liver parenchyma of 80 patients was maximum ranging from 1.4-3.6.
3.4. Liver Histopathology
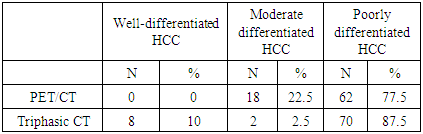
- Eighty patients performed a liver biopsy; all patients showed signs of cirrhosis. Histopathology of the Hepatocellular Carcinoma (HCC) showed well or moderately differentiated HCC in 18 patients (22.5%), while 62 patients had poorly differentiated HCC (77.5%).
3.5. Correlation of PET/CT, Tri-phasic CT with Histopathology
- 18FDG-PET/CT detected 62 patients with poorly differentiated HCC and was negative in 18 patients with well and moderately differentiated HCC, while Tri-phasic CT detected 79 patients with HCC and showed negative results in only one patient. 70 patients of 80 patients had poorly differentiated hepatocellular carcinoma, 8 patients had well-differentiated hepatocellular carcinoma, and 2 patients had moderately differentiated hepatocellular carcinoma (Table 1).
|
3.6. Correlation of Alpha-fetoprotein Level and Pathological Grade
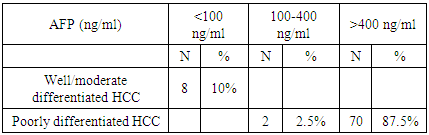
- Among the 80 patients, it was positive in 72/80 patients with poorly differentiated carcinoma with 70 patients of them had AFP level >400 ng/ml. In well-differentiated and moderately differentiated carcinoma, AFP was positive in 8 patients with 2 patients had AFP level < 400ng/ml (Table 2 and Figure 2).
|
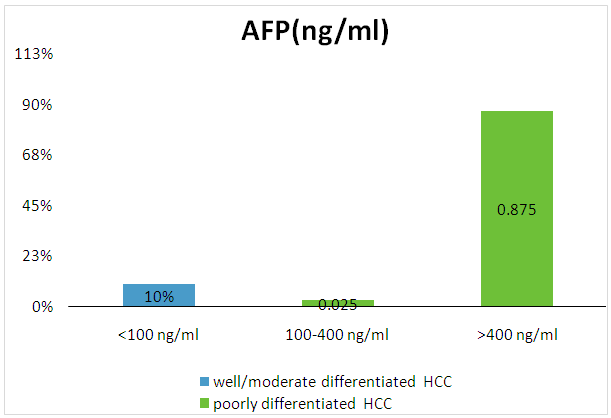 | Figure 2. Correlation between the level of AFP and grade of HCC |
3.7. Correlation between AFP and Mean SUV
- Correlation between AFP and mean SUV max revealed high level of mean SUV max in patients with AFP level >400 ng/ml of 6.2 ±2 as compared for 5.8±4.3 in patients with AFP ranging from 100-400 ng/ml, and lowest mean SUV max is seen in those with AFP <100 ng/ml.
3.8. Extrahepatic Involvement 18FDG-PET/CT and Diagnostic CT
3.8.1. Lymph Node Involvement
- Tri- phasic abdominal CT showed criteria of positive metastatic lymph nodes involvement in 26 patients (32.5%). Groups of lymph node involved were the gastric, mesenteric, porto-caval, porta-hepatislymph nodes.PET/CT positive for the metastatic lymph node involvement in the same groups detected by the Tri-phasic CT, also, it was able to detect additional lymph nodes in two patients in mediastinal.
3.8.2. Extrahepatic Metastasis
- Tri- phasic abdominal CT detected one patient with adrenal metastasis. PET/CT was able to identify 33 patient's extrahepatic metastases being done for the whole body; 17 patients had lung metastases, 15 patients had bone metastases in addition to three patient with adrenal metastases. Total extrahepatic metastases detected by 18FDG-PET/CT were 68 patients (85%).
4. Discussion
- The early detection of HCC is critical to improving affected patients the survival. Ultrasonography (US) is recommended as the primary imaging test for surveillance, and two guidelines advocate the use of serum biomarkers [8]. Currently, multiphasic CT and MR imaging with serum biomarkers are considered as first-line modalities for detection of HCC [8]. Wolfort et al. [12] revealed that in a retrospective study of 20 patients showed a correlation between FDG-PET positivity, alpha-fetoprotein level (AFP), tumor size, and histologic grade. Twenty patients on liver transplant service with biopsy-proven HCC that underwent 18FDG-PET, increased FDG uptake was noted in 14 patients (70%). 18F-FDG PET/CT detected tumors < 5 cm, only 2 of 8 was positive, while patients with tumors > 5 cm and/or multiple lesions, 12 patients were detected by FDG-PET. AFP levels < 100 ng/ml, 18F-FDG PET/CT was positive in 5 of 9 patients. In patients with AFP level > 100 ng/ml, 6 well-differentiated, 6 moderately differentiated, and 2 poorly differentiated HCC, 6 of 7 patients had positive scans. Histologically, were detected FDG- PET detected 4 of 6 patients for both well and moderately differentiated HCC and 2 Patients with poorly differentiated HCCs. The sensitivity of 18F-FDG PET in HCC detection is low in size < 5 cm than for larger tumors. There was a good correlation of sensitivity and FDG uptake intensity with larger tumor size and elevated AFP levels.In the current study, there is a good correlation with AFP with degree of liver differentiation as 6 out of the 8 patients had ALP < 400 ng/ml, while 21 out of the 32 patients had AFP > 400 ng/ml (P-value <0.01). So, there is a good correlation of sensitivity of 18F-FDG PET/CT uptake and AFP level (P < 0.001). Lee et al. [13] and Song et al. [14] found that the tumor SUV max and mean SUV ratio were a good prognostic factor. Also, Song et al. [14] confirmed that the TSUV max/L SUV mean (SUV ratio) is the good prognostic factor. In the current study, 18F-FDG PET/CT was positive for the detection of extrahepatic metastases in 68 of 80 patients (85%) with poorly differentiated HCC. Mean SUV max of 5.21±0.11 while the SUV liver parenchyma of patients was ranging from 1.4-3.6 with mean SUV max of 2.75 ±0.47. There was a strong increase in tumor to normal liver parenchyma ratio (T/N ratio), allowing for good lesion detection by visual inspection on the 18F-FDG PET/CT scan in poorly differentiated HCC. Mean SUV max ratio of parenchyma was 2.78±0.86.The pathology of the focal hepatic lesion in the current study was poorly differentiated and well/mod differentiated hepatocellular carcinoma the number of each group was 62 patients with pathology proved poorly differentiated HCC and 18 patients with moderately differentiated HCC. The correlation between PET/CT and Tri- phasic CT in detection of extrahepatic metastases revealed that PET/CT is superior in the detection of extrahepatic metastatic spread as whole-body scan in one session, correlation with aggressiveness of tumor, its superior detection of lytic lesion, adrenal metastases, lymph node detection, and lung nodules.Several studies have shown that the fusion of PET data with CT improves the sensitivity of PET and its specificity [15]. The advantage of PET/CT over PET alone, due to these causes of the low anatomic resolution of PET and the difficulty in lesion localization on PET only. Morphologic data from CT share in improving the diagnostic accuracy of nonspecific lesions with increased 18F-FDG uptake [16,17]. Although 18F-FDG PET can help to differentiate tumors and useful in the staging of HCC as a complementary modality to CT by detecting unsuspected regional and distant metastases [18]. In a study by Yoon et al. [19], pretreatment FDG PET examinations were performed on 40 patients with HCC who underwent MRI or CT studies, to detect extrahepatic metastases which was identified in 28 of 40 patients. In addition, 18F-FDG PET identified tumor lymph node metastases not seen in CT.18F-FDG PET/CT sensitivity in the detection of poorly differentiated HCC tumors is reflecting more aggressive, and thus associated with metastases for identification and staging of HCC metastases. Ho et al. [20] found dual-tracer PET /CT to have a sensitivity of 98%, a specificity of 86%. Positive predictive value (PPV) of 97% a negative predictive value (NPV) of 90%, and an accuracy of 96%.In the present study, Sensitivity of 18F-FDG PET/CT of 87% is related of reflection of tumor biology with higher detection of poorly differentiated HCC while high specificity 100% of 18FDG-PET/CT in detecting extrahepatic metastases may be explained in a part that most of the included lesions were suspicious for HCC. 18F-FDG PET/CT with dedicated contrast-enhanced CT raised the sensitivity to 98%. In the present study, 18FDG-PET/CT detected extrahepatic HCC metastases in 85%, with definite metastatic lesions in lungs, LNs, bone, and adrenals as adrenal,
5. Conclusions
- 18F-FDG PET/CT is important in evaluating extrahepatic spread of HCC. Whereas abdominal Tri-phasic CT detected only a single patient of extrahepatic metastasis in adrenals. Also, 4 patients with negative 18F-FDG PET/CT for primary hepatic HCC had extrahepatic metastases in the lung in 2 patients and 2 patients in mediastinal LN. In the setting of extrahepatic metastatic spread, 18F-FDG PET/CT seems to be an accurate method for HCC staging [12] seen in poorly differentiated HCC. Also, 18F-FDG PET/CT imaging is superior in detecting extra-hepatic metastases.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML