-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
International Journal of Composite Materials
p-ISSN: 2166-479X e-ISSN: 2166-4919
2016; 6(2): 43-47
doi:10.5923/j.cmaterials.20160602.01

Synthesis and Electrical Property of Polythiophene/Sol-gel Silver Nanoparticle-based Polyethylene Composite
Ayesha Kausar
Nanosciences Division, National Centre For Physics, Quaid-i-Azam University Campus, Islamabad, Pakistan
Correspondence to: Ayesha Kausar, Nanosciences Division, National Centre For Physics, Quaid-i-Azam University Campus, Islamabad, Pakistan.
| Email: |  |
Copyright © 2016 Scientific & Academic Publishing. All Rights Reserved.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In this attempt, silver nanoparticles were prepared and surface-modified with polythiophene. Nano-sized polythiophene nanoparticles were synthesized using H2O2 and CuSO4·5H2O, whereas silver nanoparticles were prepared using sol-gel route. Later composite nanoparticles of PTh and Ag (PThNP/s-AgNP) were prepared and loaded in polyethylene matrix. Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM) and conductivity measurement were used to investigate the influence of filler content on the physical properties of polyethylene-based composites. SEM microphotographs establish that the PThNP/s-AgNP nanoparticles were effectively dispersed in PE matrix. The d.c. electrical conductivity of composite films was measured by four-probe method. It was found that the electrical properties of the composite films were affected by the nanofiller content as well as the temperature. The conductivity of the composites increased from 10-2 to 1 Scm-1 at room temperature, while from 0.05 to 1.1 Scm-1 at 80°C with filler loading.
Keywords: Polythiophene, Polyethylene, Silver Nanoparticle, SEM, d.c. Electrical Conductivity
Cite this paper: Ayesha Kausar, Synthesis and Electrical Property of Polythiophene/Sol-gel Silver Nanoparticle-based Polyethylene Composite, International Journal of Composite Materials, Vol. 6 No. 2, 2016, pp. 43-47. doi: 10.5923/j.cmaterials.20160602.01.
Article Outline
1. Introduction
- During the current years in the field of conducting polymers, there has been an explosive growth of research owing to their remarkable electrical properties and their prospective applications in a variety of fields like electronic devices, electrochromic displays, chemical and bio-sensors and modified electrodes. Since the invention of the conducting polyacetylene, many conducting polymers have been reported comprising polypyrrole (PPy), polythiophene (PTh), polyaniline (PANI) and so on [1-3]. Amongst these, PTh is presented to be a most superior conducting polymer for numerous electrorheological, electrochemical and electronic applications because of its high electrical potential and easy handling, easy conductivity control, and thermal stability [4-6]. Polythiophenes comprises a chain of irregular single and double bonds. Each first and onwards carbon atom are linked by a sulfur atom by generation of a thionyl ring. Therefore, in comparison to other C−C bonds, the bond between the second and the third carbon atom own more single bond character and accordingly the bonds linking the thionyl rings have single bond character (Fig. 1). In addition to the structure significant function, the sulfur atoms will also affect the optical and electronic features of polythiophene. The conjugation of C atoms leads the spectroscopic features of the polythiophenes. PTh demonstrate superior potentials such as ease of fabrication and low density in comparison to other polymers. Furthermore from conducting to the insulating range, different polymerization routes have been employed for PTh. Most of the research attempts have been focused on three main classes of polymeric materials i.e. polyacetylene and its derivatives, polyphenylenes and its derivatives and polyheterocyclics such as polypyrrole, polythiophene, etc. These polymeric materials can be achieved in various forms such as thin films and powders. In order to render a material appropriate for applications in numerous technological fields, one has to prevail over definite limitations such as processibility, poor mechanical properties and instability in ambient conditions. Several investigations have been carried out progress these features [7, 8]. The development of conducting composites is one of the best ways. The distinctive layered structure of the polymer solar cell requires conducting polymer including PTh to be used in the form of thin films [9]. For the creation of soluble PTh, one of the alternatives derivatives is to organize dispersible PTh nanostructures by heterogeneous oxidative polymerization. In addition to the intrinsic features of PTh exhibited by these nanostructures, other bonus rewards may also be introduced such as vast definite surface area particularly valuable for gas sensor application [10-12].
 | Figure 1. Chemical structure of polythiophene |
2. Experimental
2.1. Materials
- Polyethylene (average Mw ~ 4,000, average Mn ~ 1,700), sodium dodecyl sulfonate (SDS), hydroxyl peroxide (H2O2, 30% solution), cupric sulfate pentahydrate (CuSO4·5H2O), sodium chloride, tetramethyl orthosilicate (TMOS), dimethyl foramide (DMF), and thiophene, all were analytical grade reagents purchased from Aldrich.
2.2. Instrumentation
- Infrared spectroscopy (IR) spectra were taken at room temperature with a resolution of 4 cm-1 using Excalibur Series FTIR Spectrometer, Model No. FTSW 300 MX manufactured by BIO-RAD. The scanning electron microscopic (SEM) images were obtained by Scanning Electron Microscope S-4700 (Japan Hitachi Co. Ltd.). The size distribution was measured by the Brunauer, Emmet and Teller (BET) using Autosorb-1C Quantachrome. The electrical conductivity of thin films was measured at room temperature and ambient atmosphere using the four-point method (Keithley 2401).
2.3. Synthesis of Polythiophene Nanoparticle (PThNP)
- To a 250 mL round bottomed flask equipped with a mechanical stirrer, 2 g thiophene, 0.1 g SDS and 50 mL deionized water was added. The mixture was refluxed for 20 min at 60°C. Then 30% H2O2 (5g) solution was added to the above mixture. Later 0.01 mol CuSO4·5H2O dissolved in 10 mL water was added and the reaction was allowed to proceed for 7 h at 60°C. The polymer was precipitated by adding sodium chloride followed by centrifugation. The precipitate was washed several times with deionized water and then dried under vacuum at 60°C.
2.4. Sol-gel Ag nanoparticle (s-AgNP)
- 4 mL TMOS was mixed with 20 mL of ethanol. The mixture was hydrolyzed using 1M silver nitrate (5 mL) for 0.5 h. After that the solution was refluxed with 5 mL DMF at 100°C [21].
2.5. Synthesis of Polythiophene/Ag Nanoparticle (PThNP/s-AgNP)
- To the s-AgNP solution prepared in Section 2.4, 0.5 g of PThNP was added. The mixture was refluxed at 70°C for 6 h. The particles were filtered and washed several times with deionized water and then dried under vacuum at 60°C (Fig. 2).
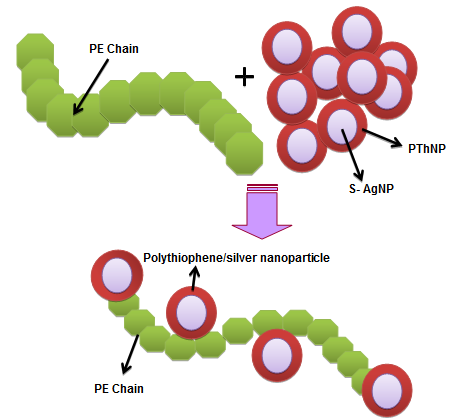 | Figure 2. Preparation of polyethylene composite with nanoparticles |
2.6. Preparation of Polyethylene and Polythiophene/Ag Nanoparticle Composite (PE/PThNP/s-AgNP)
- 1g polyethylene was dissolved in 20 mL of xylene via refluxing at 100°C for 4 h. Then as prepared nanoparticles (desired) were added to the above mixture and again refluxed for 2 h. Films were caste in glass Petri dishes.
3. Results and Discussion
3.1. FTIR Studies
- The nanostructure of polythiophene was studied by FTIR spectroscopic technique (Fig. 3). The FTIR spectrum exhibited characteristic peak at 3399 cm-1 due to water content. Major peak for C−H stretching vibration appeared at 2911 and 2844 cm-1. The peaks at 1201 cm-1 and 1039 cm-1 were due to C–H bending and C–H in-plane deformation. The peaks at 1669 and 1400 cm-1 were attributed to C=C asymmetric and symmetric stretching vibrations of thiophene ring respectively. The characteristic peak of polythiophene was found at 755 cm-1 (C–S bending) [22].
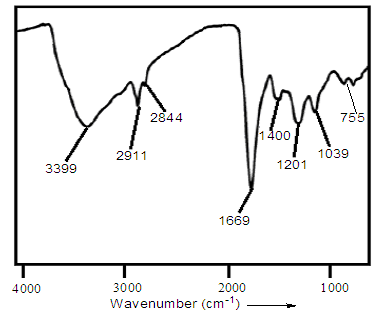 | Figure 3. FTIR spectrum of polythiophene nanoparticles |
3.2. SEM Analysis
- Fig. 4 shows SEM micrographs of the PE/PThNP/s-AgNP composite. The nanoparticles were well-dispersed in matrix and were of spherical shape. The morphology of PE/PThNP/s-AgNP 1 and PE/PThNP/s-AgNP composites are given in Fig. 4A and B, in which the composites were incorporated with 1 wt.% and 3 wt. % nanoparticles respectively. Whereas Fig. 4C shows the morphology of PE/PThNP/s-AgNP 5 with 5 wt. % nanoparticle loading. Some well-dispersed spherical dots of PThNP/s-AgNP were observed in the case of PE/PThNP/s-AgNP 1. In the PE/PThNP/s-AgNP 3, the nanoparticles were also finely distributed in the matrix. However in the case of PE/PThNP/s-AgNP 5, well-dispersed nanoparticles as well as some of the aggregated PThNP/s-AgNP were observed. The aggregation was in fact due to the higher nanoparticle content. This recognizes that an upper limit exists for an effective nanoparticle loading level. Particle size and surface area of PE/PThNP/s-AgNP 1 composite was characterized using BET analysis. The average size of nanoparticle dispersed in matrix was 70-90 nm estimated from the BET surface area (800 m2/g). Good dispersion of nanoparticles may form a conducting network that enhances the electrical conductivity of the composites. As a result, the composites with optimum PThNP/s-AgNP content have better performance in electrical properties.
 | Figure 4. SEM micrographs of (A) PE/PThNP/s-AgNP 1; (B) PE/PThNP/s-AgNP 3; and (C) PE/PThNP/s-AgNP 5 |
3.3. Electrical Characterization
- Room-temperature d.c. electrical conductivity of the PE/PThNP/s-AgNP composites films are shown in Fig. 5. For these composites, a desired amount of nanoparticles was dispersed in PE solution state. As the content of PThNP/s-AgNP in the composites was increased from 1 to 5 wt. %, the conductivity increased from 10-2 to 1 Scm-1 at room temperature (Table 1). The observed increase in electrical conductivity can be associated with the synergetic effect of the polythiophone and silver nanoparticles. Since loading of silver nanoparticle alone brought about only a limited increase in the electrical conductivity i.e. 10-5 to 10-3 Scm-1 at 30°C. Moreover the loading of silver nanoparticle alone showed lower electrical conductivity values than the PThNP/s-AgNP loading. The effect of temperature on the conductivity of these composite was also studied. At 80°C, the conductivity of all the composites was found to increase [23-25].
 | Figure 5. Electrical conductivity vs. nanoparticle content in the composite films |
|
4. Conclusions
- Nano-sized polythiophene nanoparticles were synthesized in aqueous media using H2O2 and CuSO4.5H2O. Silver nanoparticles were also prepared using sol-gel route. Later composite nanoparticles of PTh and Ag were prepared and loaded in polyethylene matrix. Influence of PThNP/s-AgNP inclusion on the properties of composite was studied by FTIR, SEM and conductivity measurement. The synthesized nanoparticles have spherical shapes with relatively uniform size and good dispersion in PE matrix. The electrical conductivity was measured by a four-probe method using composite films. The electrical conductivity of film was increased with increasing the content of conducting nanoparticles. Furthermore, small increase in the electrical conductivity with increasing the temperature was observed.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML