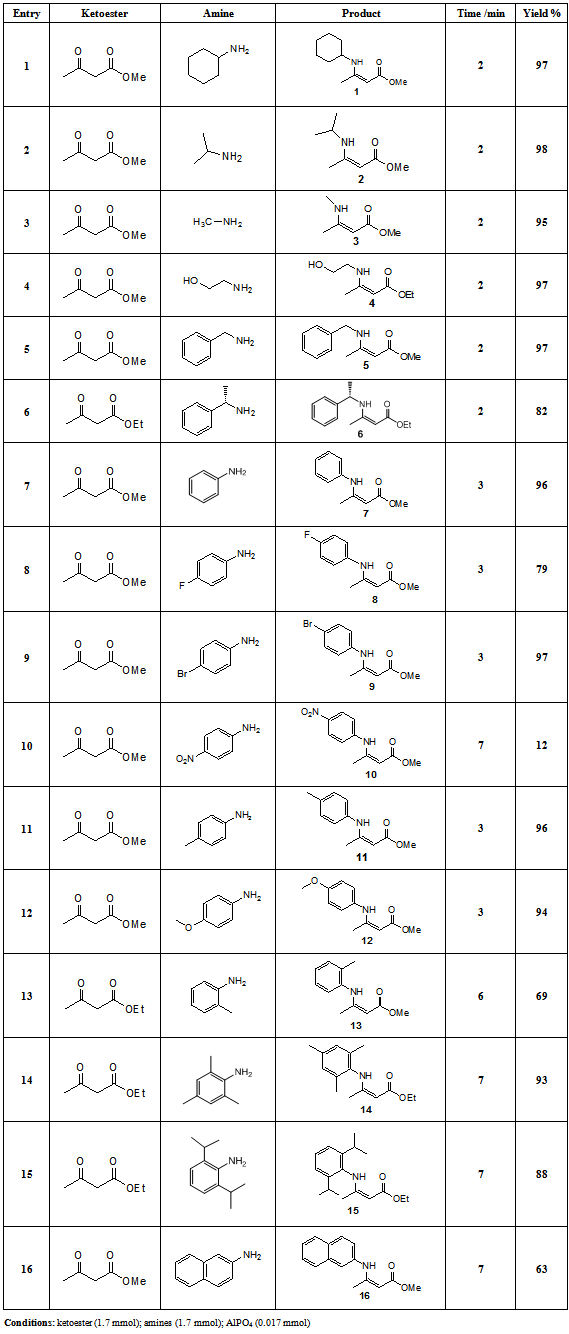
-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Chemistry
p-ISSN: 2165-8749 e-ISSN: 2165-8781
2012; 2(5): 271-276
doi: 10.5923/j.chemistry.20120205.05
Aluminum Phosphate Catalyzed Free Solvent Preparation of β-enamino Esters
Mohamed Anouar Harrad , Brahim Boualy , Larbi El Firdoussi , Mustapha Ait Ali
Université Cadi Ayyad Facuté des Sciences Semlalia, Equipe de Chimie de Coordination et Catalyse, Departement de Chimie BP 2390, 40001, Marrakech, Morocco
Correspondence to: Mustapha Ait Ali , Université Cadi Ayyad Facuté des Sciences Semlalia, Equipe de Chimie de Coordination et Catalyse, Departement de Chimie BP 2390, 40001, Marrakech, Morocco.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
Aluminum phosphate (AlPO4) efficiently catalyzed the condensation of 1,3-ketoesters with primary amines under free solvent conditions. The catalyst has been prepared and characterized by powder XRD and FT-IR studies. The AlPO4 synthesis was performed in water at room temperature from AlCl3 and H3PO4 in the presence of ammonia solution. Resultant material showed good catalytic efficiency in condensation of 1,3-ketoesters with primary amines using a domestic microwave oven with no solvent. The reaction was complete in 7 min to afford β-enamino esters in a high yield and high selectivity.
Keywords: β-enamino Esters, Microwave, Solvent Free Conditions, AlPO4
Cite this paper: Mohamed Anouar Harrad , Brahim Boualy , Larbi El Firdoussi , Mustapha Ait Ali , "Aluminum Phosphate Catalyzed Free Solvent Preparation of β-enamino Esters", American Journal of Chemistry, Vol. 2 No. 5, 2012, pp. 271-276. doi: 10.5923/j.chemistry.20120205.05.
Article Outline
1. Introduction
- β-enaminones are important intermediates in the synthesis of natural products[1] and heterocyclic compounds[2,3] . Anticonvulsive activity of several secondary enaminones has been reported[4]. A number of methods have been described for the preparation of β-enamino carbonylic compounds[5-8], such as condensation of amines with β-dicarbonylic compounds in aromatic solvents with azeotropic water removal[9] or Raney-Ni hydrogenation of isoxazoles[6]. However, these methods require high pressure and temperature[10]. The use of low boiling amines is particularly problematic. Consequently, various modified synthesis methods have been reported, in particular the addition of amide enolates[11]. Improved procedures have been reported which use protic acids such as PTSA (para-toluene sulfonic acid)[12], and Lewis acids such as BF3-OEt2[13], Mg(ClO4)2[14], Bi(OTf)3[15], Sc(OTf)3[16].As part of our work on the synthesis and reactivity of β-enaminoesters in heterogeneous media[17-19], we report here a simple and fast procedure for the synthesis of these compounds under solvent-free conditions using AlPO4 as catalyst and a domestic microwave oven. Aluminum phosphate is of great interest in environmental, technological fields[20-27] and catalytic reactions[21-27]. It can be easily prepared by means of a sol–gel protocol[28-35]. The prepared AlPO4 Materials have high density of Brönsted acid sites making them particularly suitable as heterogeneous catalysts for different organic synthesis reactions[34-35]. The use of microwave irradiation in the presence of catalysts or mineral-supported reagents, under solvent-free conditions[36], provides a simple chemical process with special attributes such as enhanced reaction rates, higher yields, greater selectivity and ease of manipulation[37].
2. Experimental
2.1. Instruments
- NMR studies were performed on a Bruker Advance 300 spectrometer in CDCl3. chemicals shifts are given in ppm relative to external TMS and coupling constant (J) in Hz. Infrared spectra (IR) were obtained on a Bruker-TENSOR 27 spectrometer instrument. X-ray diffraction patterns (XRD) were obtained with a Philips X’Pert MPD diffractometer using Cu Ka radiation (k = 1.54178 Å). Mass spectra were recorded on a GC-MS Varian star 3400 CX. Optical rotation was measured at room temperature using an ATAGO polax-D polarimeter . Microwave irradiations were carried out in a domestic microwave oven Model AVM510/WP/WH (700 W). The products’ physical and spectroscopic data were compared with those reported in the literature.
2.2. Synthesis of AlPO4
- In a 100 mL three-neck round bottom flask, AlCl3.6H2O (7.5 mmol), and 4 mL of H3PO4 (37 %) are introduced dropwise with stirring at room temperature[28-35]. After 30 min, 2.3 mL of ammonia solution (24 %) was added dropwise to precipitate aluminum phosphate at a pH of 9.0. The precipitate was filtered and washed with distilled water. The resulting product was recrystallized in methanol. The white solid was filtered, washed with Methanol and dried at 120℃ over night.
2.3. Procedure of Catalytic Studies
- In a typical experiment 1.7 mmol of ketoester, 1.7 mmol of amines and 0.17 mmol of AlPO4 were used, the heterogeneous mixture was transferred to a microwave oven at 60 W for the time indicated in Table 1. At the end of the reaction, 10 mL of distilled water were added to the residue and extracted with diethyl ether (3 × 25 mL). The organic layer was dried over Na2SO4, and the solvent was removed under vacuum. Pure β-enamioesters was obtained by column chromatography over silica gel using hexane/ethyl acetate as eluent. All isolated pure products were fully characterized by 1H, 13C NMR and Mass spectra compared with the known compounds[17].
3. Results and Discussion
3.1. Catalyst Characterization
- Structural properties of the prepared AlPO4 were characterized using FT-IR and XRD analysis.
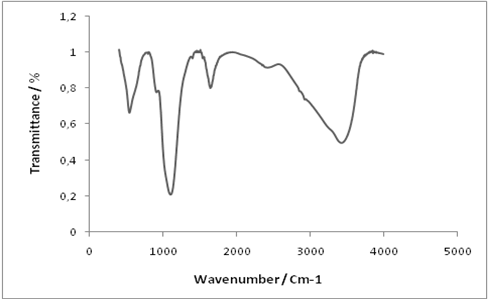 | Figure 1. FT-IR spectra of prepared AlPO4 |
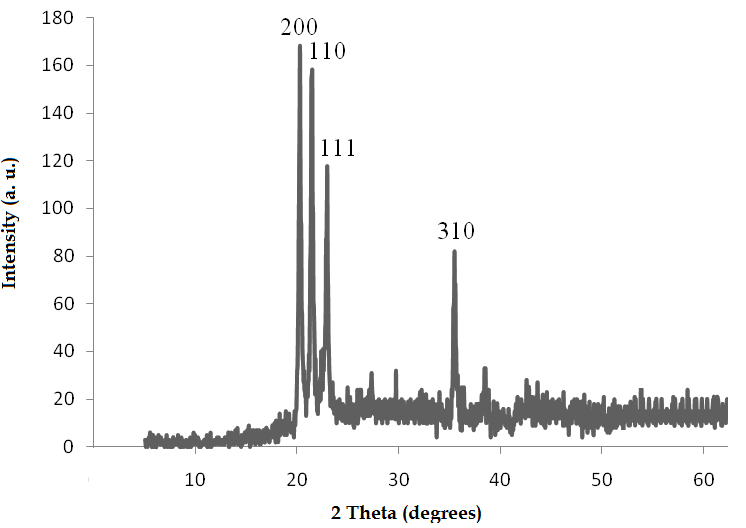 | Figure 2. The XRD patterns of obtained AlPO4 |
3.2. Catalyst Activity Studies
- In a typical experiment, equimolar ratio of ketoester and primary amine were mixed in the presence of a catalytic amount of AlPO4 (10 mol%) without solvent to obtain the corresponding enaminoester in good yields (Table 1). The reaction was completed within the indicated time under microwave irradiations using a domestic microwave oven. No by-products were obtained (scheme 1).
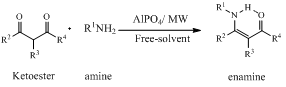 | Scheme 1. Condensation of Ketoesters with primary amines |
|
4. Conclusions
- Microwave irradiation in solvent-free conditions of primary amines and ketoesters using AlPO4 as catalyst leads to the formation of β-enaminoesters compounds in good to excellent yields. The reaction employs a simple catalytic system resulting in shorter reaction time than the conventional procedure. The advantages of the novel and facile methodology are precluding volatile, clean process, economic and environmental procedure.
ACKNOWLEDGMENTS
- We thank Pr. A. Lachgar (Wake Forest University Winston-Salem, USA) for his interest to this work and for his helpful discussion.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-Text HTML
Full-Text HTML